24+ Ionic Bond Examples
Everything around us is a melting pot of various atomic-level chemical bonds that create a working system. These bonds can be electrically or neutrally charged based on the characteristics taken on by the atom.
1. Ionic Bond Example
2. Ionic Bond Sample
3. Formation of Ionic Bond
4. Ionic Bond Model
5. Ionic Bond Lesson
6. Nail Test for Ionic Bonding
7. The Nature of the Ionic Bond
8. Ionic Bond Quiz
9. Electrovalent Ionic Bond
10. Ionic Bond Types
11. Energy and Ionic Bond
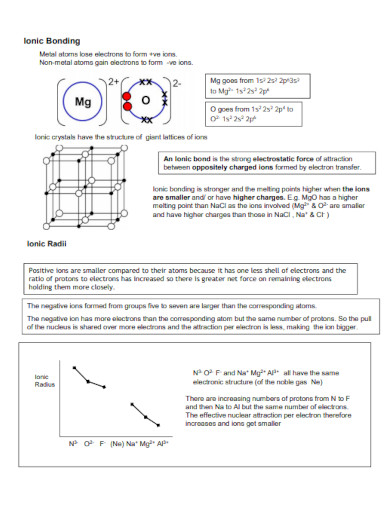
12. Ionic Bond
13. Ionic Bond Molecules
14. Ionic Bond Chapter
15. Ionic Bond Format
16. Ionic Bond Structure
17. Ionic Bond with Introduction
18. Ionic Bond Template
19. Short Ionic Bond Explanation
20. Ionic Bond Model Example
21. Standard Ionic Bond
22. Ionic Bond Worksheet
23. Printable Ionic Bond
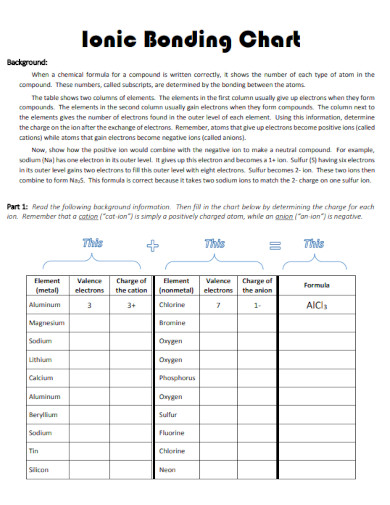
24. Ionic Bond Chart
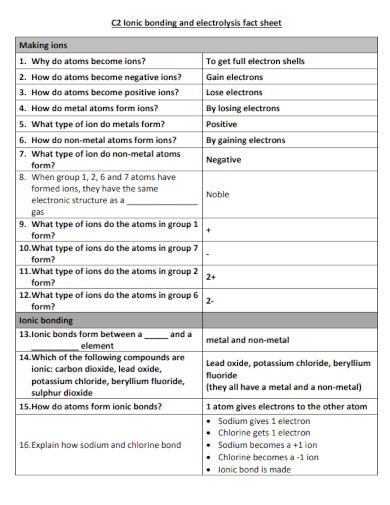
25. Ionic Bond Fact Sheet
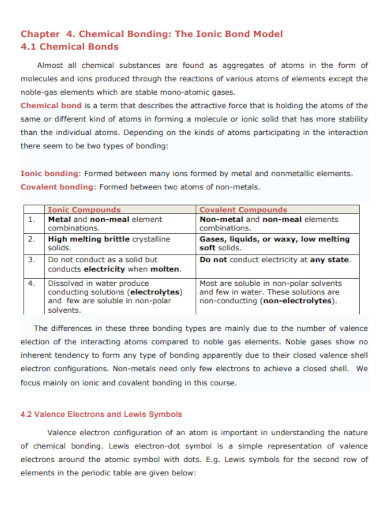
What Is an Ionic Bond
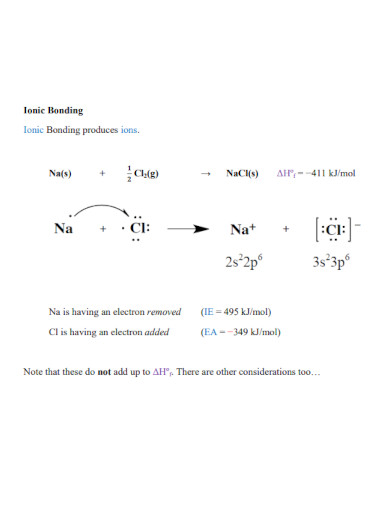
Ionic bonds are the chemical connection between two atoms. An ionic bond is formed when a specific atom transfers its electrons to another atom, forming an ionic bond. This in turn will create a product called an ionic compound that has plenty of identifiable traits.
How to Differentiate an Ionic Compound from a Covalent Compound
There are two main types of compounds that are created when chemists form said compounds through the chemical mixing of atoms. These two types of compounds are ionic and covalent compounds. The type of bond the compound uses dictates what type of compound is made. If you are still confused about the concept of the ionic compound or bond, then you are free to read and peruse any of the ionic bond examples, samples, and pdfs on the list above.
1.) Check if the Compound has Ionic Properties
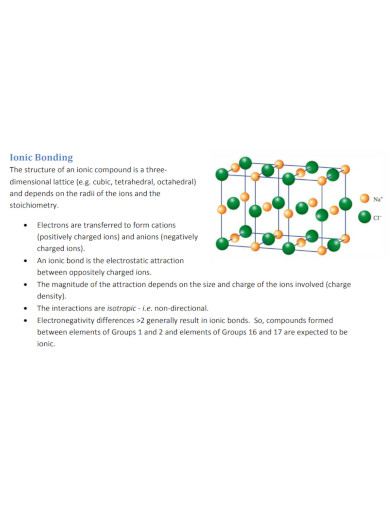
Begin by checking if the compound is exhibiting any of the shared properties of an ionic compound. Most ionic compounds are crystalline in appearance and have high melting and freezing points. These compounds are soluble in water and will often release an electrical charge when mixed with water.
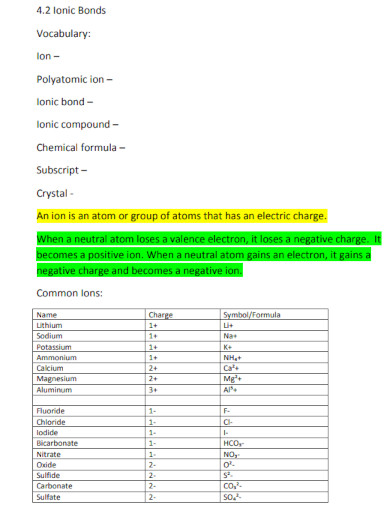
2.) Identify the Ions of the Compound
All ionic compounds are formed and share an ionic bond. This means that the atoms composing the compound will be a combination of a negatively charged ion, an anion, and a positively charged ion called a cation.
3.) Check if the Compound is Electrically or Neutrally Charged
The main difference between ionic and covalent compounds is the electrical charges these two compounds hold. Water is an example of a covalent compound and is neutrally charged without the interference of outside forces. While salt, an ionic compound releases a small electrical charge when it is mixed with water due to its ionic composition.
FAQs
Ionic bond vs Covalent bond; what are the differences between ionic and covalent bonds?
There are two types of chemical bonds in chemistry these two types come in the form of ionic and covalent bonds. An ionic bond is a chemical bond between two or more atoms that transfer energy from one to the other. The resulting atoms and compounds in the ionic chemical bond will be electrically charged. A covalent bond is a chemical bond where two or more atoms share their energy with each other. The resulting atoms and compounds of the covalent bond are not electrically charged and are considered neutrally charged. These are the main differences between both ionic and covalent bonds.
What are typical real-life examples of ionic bonds or compounds?
There are plenty of ionic compounds we can find in our everyday lives, this is because most ionic compounds are soluble in water. NaCl or sodium chloride, also known as table salt, is a commonly used ionic compound. This is because Na or sodium atoms transfer their electrons to the Cl or chlorine atom to form NaCl. Another example of an ionic compound is NaHCO3 or sodium bicarbonate also known as baking soda. All ionic compounds are a product of the combination of a cation and an anion.
Why is it essential for researchers or chemists to understand the concept of ionic bonds?
The understanding of ionic compounds allows chemists to predict and ensure the synthesis or combination of both atoms results in an ionic compound. Not only will this allow chemists to predict the creation of an ionic compound it will also let the chemist edit or take advantage of the ionic properties or characteristics of the compound to create the desired product or compound.
Ionic bonds are chemical bonds that indicate the successful transfer of one energy to another forming ionic compounds. These ionic compounds have multitudes of uses due to their property of being very miscible and soluble in water. In conclusion, ionic bonds make up a portion of the things we see and use in our everyday lives.