24+ Physical Change Examples
Have you ever watched water boil as heat reaches the boiling point? Some people believe that boiling water is a chemical change due to heat application in the water. But this is a common misconception, as boiling water is an example of a physical change.
1. Physical Change Sample
2. Physical Change Format
3. Physical vs. Chemical Change
4. Physical Change Draft
5. Physical Changes of Matter
6. Basic Physical Change
7. Physical Changes Are Dramatic
8. Standard Physical Change
9. Value of Physical Change
10. Physical Change PDF
11. Physical Change Worksheet
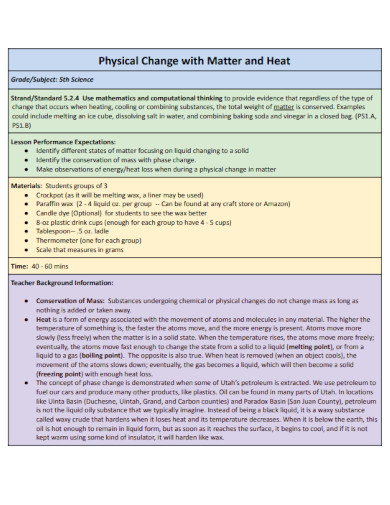
12. Physical Change with Matter and Heat
13. Physical vs. Chemical Change Example
14. Physical Processes and Climate Change
15. Physical Health Plan Change Request Form
16. Physical Changes and Concerns
17. Physical Change Lab
18. Sample Physical Change Example
19. Understanding Physical Change PDF
20. Physical Change in PDF Example
21. Physical Change in the Digestive System
22. Draft Physical Change
23. Physical Change General Chemistry
24. About Physical Change
25. Physical Change vs Chemical Change Lab
What Is a Physical Change
Physical change is often a reversible physical alteration of a substance or object. This will manifest in the changing size, form, color, and shape. There are also uncommon changes that intersect with chemical changes these are odor, solubility, fluidity, boiling, and freezing point.
How to Discern a Physical Change
Water boiling is a physical change of matter as it shifts the liquid state of matter to that of the gaseous state. Another basic physical change example is the bend of steel sheets, as this action doesn’t change the chemical composition as stress is applied to the sheet to change its form. Just note that a change in the physical property of matter doesn’t necessarily denote a physical change; as chemical changes can also change the properties of an object.
1.) Check the Method Applied to the Object
First, you must carefully appraise what method was used to create a change in the object. It is more likely that a physical method like bending, breaking, hammering, etc. will cause a physical change. This is usually the case as long as nothing new is added to the object as physical force is added to it.
2.) Observe any Changes in the Color
After checking the method applied to the object, you will now need to observe changes in the color of the object. A chemical change often causes a change in the shade of an object, but in some cases like adding new paint to a car, causes a physical change in color.
3.) Measure the Size of the Object
Any changes in the size of the object may indicate either a physical or a chemical change. Again you will know it is a physical change when it is reversible. An example of physical change would be the stretching of heated metal, at any point in time you can reverse the stretching of the metal by applying heat to and reforming the metal back to its original size.
4.) Examine for Any Change of Shape
Physical changes can often lead to a change in the object’s shape, like the shaping of clay. You can adjust the shape and size of a piece of clay, but its chemical composition stays the same. This is an excellent sample of physical change.
FAQs
Physical vs chemical change; what is the difference between physical and chemical change?
A chemical change is the perceived irreversible change of an object often the result of the change of chemical composition. The chemical change definition is something that is juxtaposed with the physical change definition. As physical changes entail reversible shifts or changes perceived on an object.
Are all chemical changes irreversible?
Yes, all chemical changes are irreversible, as one cannot reverse the change of another object’s chemical composition. An example of a chemical change is that of the baking of bread, several ingredients are mixed to create the dough of the bread. You cannot unmix the bread to obtain the ingredients you used in making said bread.
How do I reverse the physical change of water vapor, when I boil my water?
Water vapor is the product of water evaporating into the atmosphere. You can easily reverse the shift of water vapor by adding a piece of cloth over the vessel. This cloth will catch the vapor and condensate these vapors into their liquid state.
It is important to know what changes are physical or chemical, because of how they can affect our lives. Any wrong treatment of a chemical change may cause irreversible damage to one’s life and livelihood.