Units Of Energy – Definition, List of Units, Conversion Chart
In physics, we often discuss different quantities like energy, time, and work. Each of these quantities needs a standard unit of measurement to help us describe and understand them properly. For example, when we say someone weighs 36 kilograms (kg) and lives 1200 kilometers (km) away, “kg” and “km” are the units used to describe weight and distance, respectively. Similarly, we use Kelvin to measure temperature.
What is the Unit of Energy?
SI Unit of Energy
The Joule (symbol: J) is the official unit of energy in the International System of Units (SI). It is named after the English physicist James Prescott Joule. One joule represents the amount of energy transferred when applying a force of one newton over a distance of one meter.
Common Use: Joules are used universally in all fields of science and engineering to measure energy, work, or heat. For example, in electrical terms, one joule is the energy expended by passing an electric current of one ampere through a resistance of one ohm for one second.
CGS Unit of Energy
The erg is the unit of energy in the centimeter-gram-second (CGS) system of units. It is a smaller unit compared to the joule. One erg is defined as the amount of energy done by a force of one dyne exerted for a distance of one centimeter.
Common Use: Though largely replaced by the joule in many fields due to the international adoption of the SI system, the erg is still used in some contexts within physics, especially in electromagnetism and astronomy.
Units of Energy in MKS System
In the MKS (meter-kilogram-second) system, which is a precursor to the modern SI system, the unit of energy is also the joule. The MKS system uses meters, kilograms, and seconds as its basic units of length, mass, and time, respectively. This makes the joule’s definition in MKS the same as in SI.
Common Use: The MKS system was widely used before the official adoption of the SI system and its use of the joule paved the way for its inclusion in the SI system. Today, joules are universally recognized and used across scientific and engineering disciplines.
List of Energy Units
| Energy Unit | Symbol |
|---|---|
| Joule | J |
| Kilowatt-hour | kWh |
| Calorie | cal |
| Kilocalorie | kcal |
| Electronvolt | eV |
| British Thermal Unit | BTU |
| Foot-pound | ft-lb |
Joule (J)
The Joule is the SI unit of energy named after James Prescott Joule. It measures the amount of energy expended when a force of one newton is applied over a distance of one meter. It’s used universally in science and engineering to quantify energy, work, or heat.
Kilowatt-hour (kWh)
This unit is widely used in electrical applications, particularly for measuring electrical energy usage in homes and businesses. It represents the energy produced or consumed by a power of one kilowatt over a period of one hour.
Calorie (cal)
A calorie is a unit of energy that originally was defined as the amount of heat needed to raise the temperature of one gram of water by one degree Celsius at atmospheric pressure. It’s commonly used in chemistry and the food and beverage industry to specify the energy content.
Kilocalorie (kcal)
Also known as a food calorie, kilocalories are used to express the energy content in foods and are equivalent to 1000 small calories. In dietary contexts, these are the “calories” that are counted to determine energy intake.
Electronvolt (eV)
An electronvolt is a unit of energy equal to approximately 1.602×10−191.602×10−19 joules. It is commonly used in atomic, nuclear, and particle physics to describe the energy levels of electrons and other subatomic particles.
British Thermal Unit (BTU)
The BTU is used primarily in the United States for heating and cooling systems and defines the amount of energy needed to raise the temperature of one pound of water by one degree Fahrenheit. It is still common in industrial and commercial heating and cooling.
Foot-pound (ft-lb)
This unit measures energy in terms of mechanical work done when a force of one pound-force is exerted along one foot of displacement. It is mainly used in the United States and remains popular in mechanical engineering and physics for measuring rotational forces.
Conversion of Energy Units
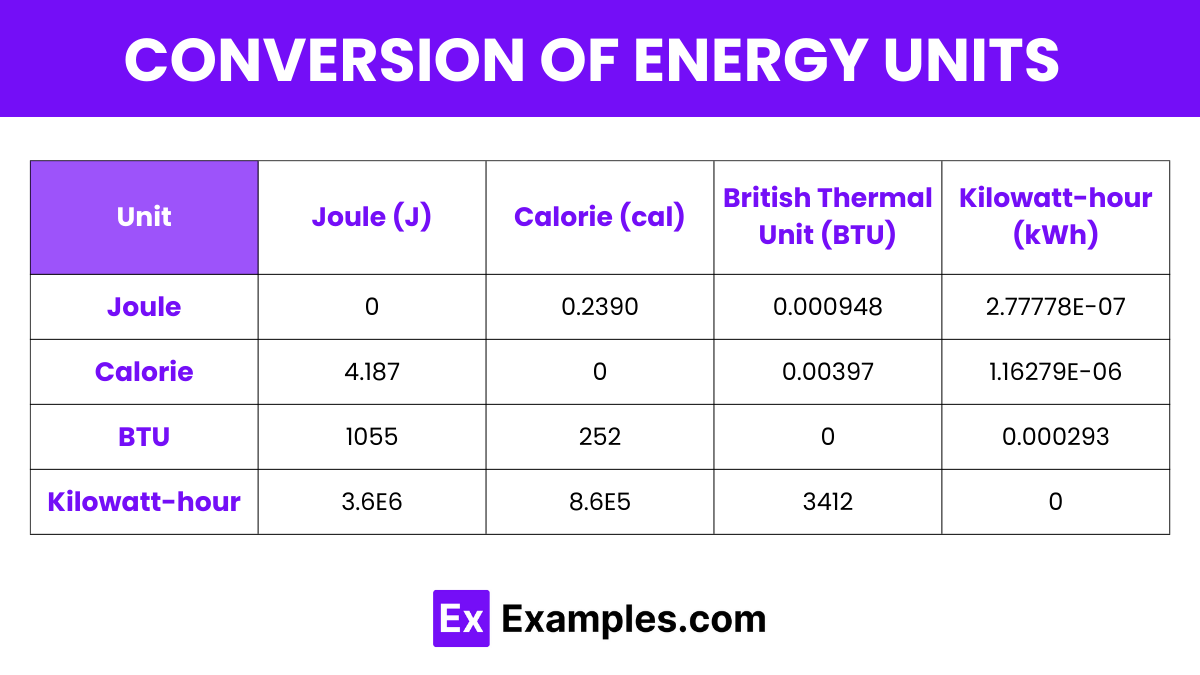
| Unit | Joule (J) | Calorie (cal) | British Thermal Unit (BTU) | Kilowatt-hour (kWh) |
|---|---|---|---|---|
| Joule | 0 | 0.2390 | 0.000948 | 2.77778E-07 |
| Calorie | 4.187 | 0 | 0.00397 | 1.16279E-06 |
| BTU | 1055 | 252 | 0 | 0.000293 |
| Kilowatt-hour | 3.6E6 | 8.6E5 | 3412 | 0 |
1. Joule to Calorie
- Conversion: 1 calorie = 4.187 joules.
- Example: To convert 100 joules to calories, divide by 4.187.
- 100 J/4.187 = 23.88 cal
2. Joule to British Thermal Unit (BTU)
- Conversion: 1 BTU = 1055 joules.
- Example: To convert 500 joules to BTUs, divide by 1055.
- 500 J/1055 ≈ 0.474 BTU
3. Joule to Kilowatt-hour
- Conversion: 1 kilowatt-hour = 3.6 x 10⁶ joules.
- Example: To convert 18000 joules to kilowatt-hours, divide by 3.6 x 10⁶.
- 18000 J/3.6×10⁶ ≈ 0.005 kWh
4. Calorie to Joule
- Conversion: 1 joule = 0.2390 calories.
- Example: To convert 50 calories to joules, multiply by 4.187.
- 50 cal×4.187 = 209.35 J
5. Calorie to British Thermal Unit (BTU)
- Conversion: 1 calorie = 0.00397 BTUs.
- Example: To convert 100 calories to BTUs, multiply by 0.00397.
- 100 cal×0.00397 = 0.397 BTU
6. Calorie to Kilowatt-hour
- Conversion: 1 kilowatt-hour = 8.6E5 calories.
- Example: To convert 1000 calories to kilowatt-hours, divide by 8.6E5.
- 1000 cal/8.6×105 ≈ 0.00116 kWh
7. British Thermal Unit to Joule
- Conversion: 1 joule = 0.000948 BTUs.
- Example: To convert 2 BTUs to joules, multiply by 1055.
- 2 BTU×1055 = 2110 J
8. British Thermal Unit to Calorie
- Conversion: 1 BTU = 252 calories.
- Example: To convert 5 BTUs to calories, multiply by 252.
- 5 BTU×252 = 1260 cal
9. British Thermal Unit to Kilowatt-hour
- Conversion: 1 kilowatt-hour = 3412 BTUs.
- Example: To convert 10 BTUs to kilowatt-hours, divide by 3412.
- 10 BTU/3412 ≈ 0.00293 kWh
10. Kilowatt-hour to Joule
- Conversion: 1 joule = 2.77778E-07 kilowatt-hours.
- Example: To convert 1 kilowatt-hour to joules, multiply by 3.6 x 10⁶.
- 1 kWh×3.6×10⁶ = 3.6×10⁶ J
11. Kilowatt-hour to Calorie
- Conversion: 1 calorie = 1.16279E-06 kilowatt-hours.
- Example: To convert 0.5 kilowatt-hours to calories, multiply by 8.6E5.
- 0.5 kWh×8.6×105 = 430000 cal
12. Kilowatt-hour to British Thermal Unit
- Conversion: 1 kilowatt-hour = 3412 BTUs.
- Example: To convert 0.1 kilowatt-hour to BTUs, multiply by 3412.
- 0.1 kWh×3412 = 341.2 BTU


