30+ Conduction Examples
Conduction is a fundamental heat transfer method where heat moves through materials or between objects in direct contact. This guide thoroughly explores conduction, offering clear examples and explanations. It’s an invaluable resource for educators, simplifying this complex concept for students. By examining everyday items and industrial processes, this guide highlights conduction’s crucial role in daily life and technological applications, demonstrating its significance in both natural and engineered systems.
What is Conduction? – Definition
Conduction is the process of direct heat transfer within a material or between materials in contact, without any movement of the material itself. It’s an important concept in thermodynamics and physical science, essential for understanding how heat moves through solids. Clear comprehension of conduction is vital for students studying physics and engineering, as it plays a key role in various applications and phenomena.
What is the Best Example of Conduction?
An excellent example of conduction is the heating of a cooking pan on a stove. When the pan is placed on a hot stove, heat conducts from the stove to the pan, evenly distributing it across the pan’s surface. This direct transfer of heat from the heat source to the pan and then to the food illustrates conduction’s efficiency and practicality in everyday use.
Conduction Formula
One equation for conduction calculates heat transfer per unit of time from thermal conductivity, area, thickness of the material, and the temperature difference between two regions:
Q = [K * A * (Thot – Tcold)] / d
Where,
Q is heat transfer per unit time
K is the coefficient of thermal conductivity of the substance
A is the area of heat transfer
Thot is the temperature of the hot region
Tcold is the temperature of the cold region
d is the thickness of the body
Conduction Unit
The primary unit used to measure thermal conductivity in conduction is watts per meter Kelvin (W/m·K). This unit quantifies how effectively a material conducts heat, playing a crucial role in material science and engineering. Understanding thermal conductivity is key in selecting materials for insulation, heating elements, and various industrial applications, ensuring efficient energy use and safety.
22 Conduction Examples
Conduction, a vital process in heat transfer, is evident in numerous everyday activities and scientific phenomena. This compilation of 22 unique examples illustrates the widespread application of conduction, providing a tangible understanding of this concept. Each example showcases how conduction facilitates the transfer of heat or electricity, making it a fundamental aspect of both natural processes and human-made technologies. Ideal for educational purposes, these instances help to clarify and contextualize conduction for students and teachers alike.
- Ironing Clothes: The heat from the iron transfers to the clothes through conduction.
- Cooking on a Gas Stove: Heat conducts from the flame through the pot or pan to the food.
- Ice Melting in Hand: Heat from the hand conducts to the ice, causing it to melt.
- Warming Feet on a Hot Water Bottle: Heat transfers from the water through the bottle to the feet.
- Metal Spoon in Hot Soup: The spoon heats up as heat conducts from the soup.
- Touching a Cold Window: Heat transfers from the hand to the colder glass.
- Walking Barefoot on Hot Sand: Heat from the sand conducts to the feet.
- Computer CPUs: Heat generated conducts to a heat sink to keep the CPU cool.
- Car Engine Cooling: Heat from the engine conducts to the coolant and then to the air.
- Charging Smartphones: Electrical conduction in the charger transfers energy to the phone battery.
- Lighting a Match: Heat conducts from the friction point to ignite the match head.
- Hot Coffee in a Mug: Heat from the coffee conducts to the mug.
- Handling a Hot Pot with a Wet Cloth: The heat conducts rapidly through the wet cloth.
- Underfloor Heating: Heat conducts from the heated floor to the room.
- Melting Butter in a Pan: Butter melts as heat conducts from the pan.
- Using a Heating Pad: Electrical energy is conducted and converted to heat.
- Baking in an Oven: Heat conducts from the oven walls to the baking food.
- Hot Asphalt Roads in Summer: Heat conducts to the surface, making it hot to touch.
- Metal Bridge Expansion in Sunlight: Heat from the sun conducts through the bridge, causing it to expand.
- Electric Kettles: Electrical energy is conducted to heat water.
- Ice Skating: Friction heat conducts to the ice, slightly melting it for smoother movement.
- Thermal Conductivity Experiments in Labs: Demonstrating how different materials conduct heat.
Conduction Examples In Everyday Life
Conduction is not just a scientific phenomenon but a part of our daily experiences. This section highlights five unique examples of conduction in everyday life, emphasizing its practical presence in common activities. These instances demonstrate how conduction is integral to routine tasks, making it easier for educators to explain this concept in a relatable, understandable way. From household chores to personal care, conduction plays a vital role in our day-to-day activities.
Examples:
- Handling a Hot Cup of Tea: Heat conducts from the hot tea to the cup and then to your hands.
- Using a Metal Ladle in Cooking: The ladle heats up when it’s in a pot of boiling soup due to conduction.
- Resting on Leather Car Seats in Summer: The seats absorb heat from the sun, becoming hot to touch.
- Wearing Metal Jewelry in the Sun: The jewelry conducts heat from the sun and warms up against the skin.
- Ironing Board Getting Warm: Heat from the iron transfers to the board through conduction during ironing.
Conduction Examples In Real Life
Conduction in real life is evident in various forms, impacting our daily activities and industrial processes. This section offers five distinct examples, showcasing the practical applications of conduction in real-life scenarios. These examples provide an insight into how conduction is a fundamental part of many systems and processes, proving to be an invaluable teaching tool for illustrating the concept’s relevance in the real world.
Examples:
- Warming Hands by a Radiator: Heat from the radiator conducts to your hands when you hold them close.
- Melting Chocolate in a Pan: The pan conducts heat from the stove to the chocolate, causing it to melt.
- Wearing Insulated Gloves in Winter: The gloves prevent body heat from conducting away, keeping hands warm.
- Working on Laptops: Heat conducts from the internal components to the laptop’s body, often making it warm.
- Cooking Eggs on a Hotplate: The hotplate conducts heat directly to the frying pan, cooking the eggs.
Types of Conduction
Conduction, a critical concept in physics, manifests in various types including ionic, electric, and thermal conduction. Each type has unique mechanisms and applications, making conduction a versatile phenomenon in different fields.
1. Ionic Conduction
Occurs in electrolytes and involves the movement of ions. An example is the conduction of electricity through an electrolyte solution in a battery, where ions move to transmit electrical energy.
2. Electric Conduction
Involves the flow of electrons through a conductor, like metals. Electric wires in household appliances are classic examples, where electrons move through the wire to transmit electricity.
3. Thermal Conduction
Refers to the transfer of heat through a material. A common example is a metal rod heated at one end, where heat travels through the rod to the cooler end.
Process of Conduction
Conduction is the process of heat transfer through direct contact of atoms and molecules within a material or between materials. It’s a fundamental concept in thermodynamics, playing a crucial role in various everyday phenomena and industrial applications. This guide outlines the steps involved in the conduction process, providing a clear, step-by-step understanding of how conduction occurs. Through these steps, the mechanism by which heat or electricity moves through materials becomes more comprehensible, particularly in the context of teaching and learning physics.
Steps of the Conduction Process:
- Initiation of Heat Source: Conduction begins when one part of an object or material is exposed to a heat source.
- Vibration of Atoms: The heat causes atoms or molecules at the heated end to vibrate more vigorously.
- Transfer of Kinetic Energy: These vibrations transfer kinetic energy to adjacent atoms or molecules, which start to vibrate as well.
- Continued Energy Transfer: The process continues along the material, with energy being passed from one particle to the next.
- Heat Distribution: Gradually, this transfer of kinetic energy leads to a uniform distribution of heat across the material.
- Equilibrium State: Eventually, the entire material reaches thermal equilibrium, where heat is evenly distributed, and no further conduction occurs unless there is a change in temperature.
Understanding these steps of conduction is essential in fields like material science, engineering, and environmental studies, where heat transfer plays a key role in system design and functionality.
Characteristics of Conduction
Conduction is a fundamental mode of heat transfer characterized by the direct transfer of thermal energy through a material without any physical movement of the substance itself. This process relies on the interaction and energy exchange between particles within a material. Understanding the key characteristics of conduction is essential in fields like physics, engineering, and materials science, where heat transfer plays a crucial role in system designs and operations.
Key Characteristics:
- Particle Interaction: Involves the transfer of energy between adjacent atoms or molecules within a material.
- Direct Contact Required: Occurs within a material or between materials in direct physical contact.
- Predominant in Solids: Most effective in solids, especially metals, due to closely packed atoms allowing efficient energy transfer.
- Temperature Gradient: Requires a temperature difference, with heat flowing from a hotter region to a cooler one.
- No Mass Transport: Unlike convection, conduction involves no movement of mass from one place to another.
- Thermal Conductivity: Varies among materials, with metals generally having higher conductivity than insulators.
- Rate of Conduction: Dependent on the material’s thermal conductivity, cross-sectional area, temperature difference, and thickness.
Heat Transfer through Conduction
Heat transfer through conduction can be explained in a series of steps, illustrating how thermal energy moves through materials.
- Heat Introduction: Heat is introduced to one part of the material.
- Molecular Vibration: This heat causes the molecules at the heated part to vibrate more vigorously.
- Heat Transfer: These vibrations are passed along to neighboring molecules.
- Thermal Equilibrium: The process continues until there is uniform heat distribution or thermal equilibrium.
How to Solve a Conduction Solution
The formula of conduction is written as Q = (-K)[(A)(Dx/Dt)]. This will allow the person to figure out the heat flow in a given system, which will technically allow the person to understand how fast the heat source can transfer thermal energy to another object.
Step 1: Write Down the Formula
Begin by writing down the equation for conduction (heat transfer) on your physical notepad or note-taking software. The writing down of the equation will provide you with a working outline that will provide you with the necessary structure and information to create the equation.
Step 2: List Out the Given Numbers
After you have written down the equation, you must list out the given numbers in the question and try to isolate them from each other. This will not only help organize the information but will also allow you to conduct individual conversions for each of the numbers.
Step 3: Substitute the Numbers to the Formula
After you have listed out and isolated the numbers, you must substitute the given numbers into the formula. Q represents the heat flow through the two objects in watts (W), K represents the constant thermal conductivity of the body (W/mk), A represents the surface area of the system (cm), dt represents the temperature difference in the solution (C or K), and dx represents the thickness of the body in the flow (cm).
Step 4: Use the Equation to Search for the Missing Number
After you have substituted the formula you will end up with an equation with a missing value that you will need to search for. Ensure that the missing value is on the left side of the equation, and observe the proper conversion of the equation.
Step 5: Ensure the Correct Format for The Answer
The answer should be written in the correct format based on the thing it represents in the formula. If the question requires a different unit of measurement then adjust and convert accordingly.
FAQs
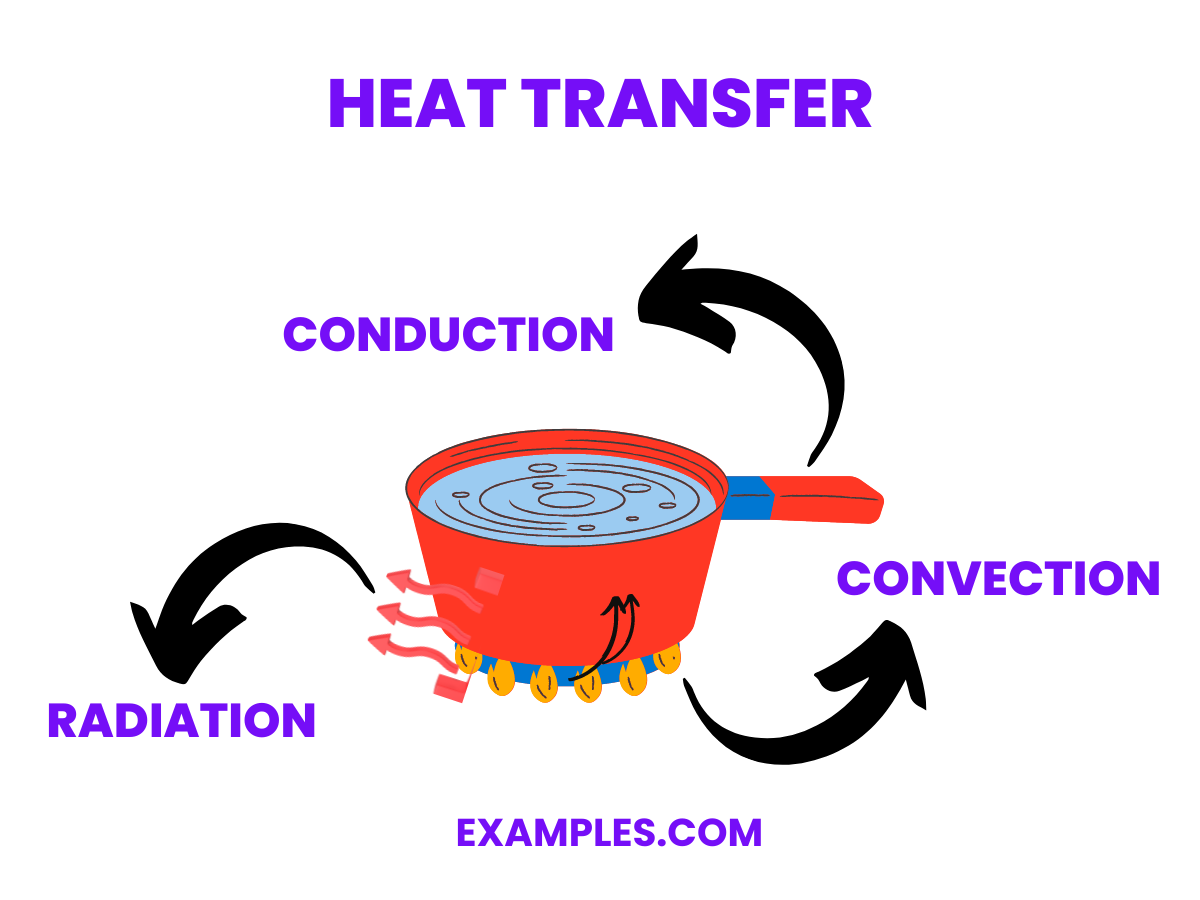
What is the difference between conduction & convection in heat transfer?
The main difference between these two methods of heat transfer is their main method of transferring heat from one place to another. Conduction is a method of transferring heat or thermal energy through direct contact with the heat source and the object. This is juxtaposed by convection which uses a combination of liquid and density as the primary heat source, which will heat up the object either through the submerging of a specific object in the heated liquid or through a piping system that will act as the heating system. This best illustrates the difference between the two types of heat transfer.
What are the 2 types of conduction?
Steady-state conduction is one of the types of conduction that indicates the constancy of the heat source and its contact with the heated object. Not only that but this type of conduction requires a specific type of wall that will not absorb the heat source’s thermal energy. This type of wall is called an adiabatic wall. The other type of energy is called transient conduction, which has a heat source that will not constantly emit the same amount of heat or thermal energy throughout the session. Most conduction heat sources fall in the category of transient conduction as a steady state requires a complicated and complex set-up.
What is the difference between conduction & induction?
Conduction, in terms of electricity generation and flow, is the generation or pushing of a current to a specific direction through the use of electrical fields. The flow of free charges in a system is guided by the presence or absence of an electric field in an electric conductor. Induction, on the other hand, is the movement or generation of electrical current through the presence of a magnetic field. The charged current will repulse or attract another uncharged current to flow through a single direction in an electric conductor.
Conduction refers to two scientific methods that span the transmission of heat and the flow of energy in a single direction inside a closed system. This method of heat transfer is one of the most frequently used methods of transferring thermal energy.