Bronze (Copper and Tin) – Definition, Structure, Preparation, Uses, Side Effects
Bronze is a durable metallic compound with a long history in various cultures, valued for its strength and beauty. It’s crafted through a chemical process that combines copper and tin, along with small amounts of other metals like aluminum, manganese, nickel, or zinc to create different types of bronze with unique properties. This blend makes bronze harder than pure copper, giving it a wide range of applications from making coins and medals to constructing statues and musical instruments. In chemistry, bronze is fascinating because it demonstrates how combining elements can create a material with new characteristics, including resistance to corrosion and a distinctive, golden-brown color.
What is Bronze?
Structure of Bronze
Bronze is an alloy, which means it is made by melting and mixing two or more elements together. In the case of bronze, the primary elements are copper and tin. Copper, the main component, gives bronze its strength and ductility, allowing it to be easily shaped and molded. Tin, added in smaller amounts, enhances the hardness and melting point of the alloy, making it more resistant to wear and tear. The structure of bronze can vary depending on the exact ratio of copper to tin, leading to different types of bronze suited for various applications. Some bronzes may also include small amounts of other elements like zinc, lead, or phosphorus to alter its properties, such as improving its ability to cast or increasing its resistance to corrosion.
Preparation of Bronze
The preparation of bronze involves melting copper and tin together. First, copper is heated until it melts. The melting point of copper is about 1,984 degrees Fahrenheit (1,085 degrees Celsius). Once the copper is in its liquid form, tin is added to the molten copper. Tin melts at a lower temperature, around 449 degrees Fahrenheit (231 degrees Celsius), so it easily mixes with the molten copper. The typical ratio is about 88% copper to 12% tin, but this can vary depending on the desired properties of the bronze.
The chemical equation for the reaction, simplifying the process, looks something like this:
This equation represents solid copper (Cu (s)Cu (s)) and solid tin (Sn (s)Sn (s)) being heated and combined to form a solid bronze alloy (CuSn (s)CuSn (s)). In reality, the process involves the metals in their molten (liquid) state, and the resulting alloy’s composition can vary. After the metals are combined and stirred to ensure a uniform mixture, the molten bronze is poured into molds and allowed to cool and solidify into the desired shapes
Physical Properties of Bronze
| Property | Description |
|---|---|
| Color | Bronze has a characteristic golden to reddish-brown color, which can vary depending on the specific copper-to-tin ratio. |
| Density | It typically has a density around 7.4 to 8.9 grams per cubic centimeter, making it heavier than pure copper. |
| Melting Point | Bronze melts at approximately 1,742°F (950°C), which is lower than pure copper but varies with composition. |
| Hardness | It is harder than copper due to the presence of tin, making it resistant to wear and suitable for tools and machinery. |
| Conductivity | While bronze conducts electricity and heat, it does so less efficiently than copper, due to tin’s inclusion. |
| Malleability | Despite being hard, bronze is malleable and can be shaped into various forms, though it is less so than copper. |
| Corrosion Resistance | Bronze is highly resistant to corrosion, especially to seawater, making it ideal for maritime applications. |
| Patina Formation | Over time, bronze develops a green patina when exposed to air, which protects it from further corrosion. |
Chemical Properties Of Bronze
Resistance to Corrosion
Bronze resists corrosion due to the formation of a protective oxide layer on its surface. This is especially effective against salt water, making bronze valuable for maritime uses.
Patination
Bronze develops a protective green or brown patina over time, which is a layer of copper carbonate. This occurs through the reaction:
2Cu + CO₂ + O₂ + H₂O → Cu₂(OH)₂CO₃
This layer shields the bronze from further corrosion.
Reactivity
Bronze shows low reactivity, making it stable and durable, especially in conditions promoting oxidation.
Thermal Expansion
The thermal expansion of bronze is characterized by its expansion when heated and contraction when cooled, at rates determined by its specific composition
Uses of Bronze
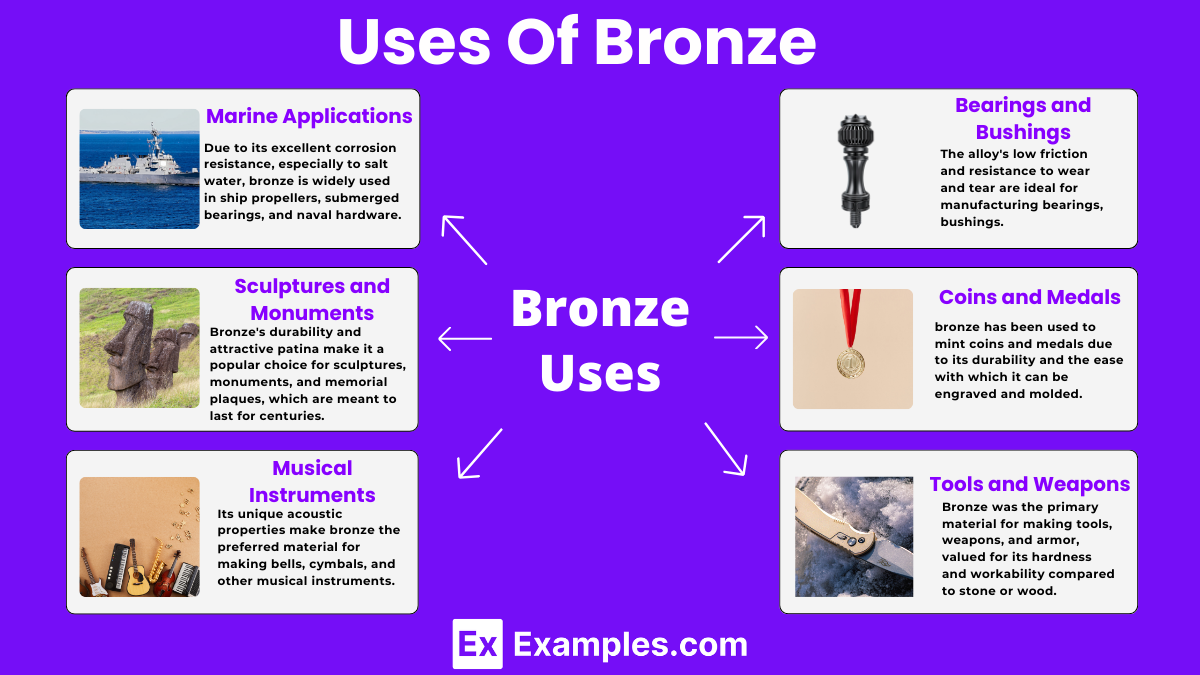
Marine Applications
Due to its excellent corrosion resistance, especially to salt water, bronze is widely used in ship propellers, submerged bearings, and naval hardware.
Sculptures and Monuments
Bronze’s durability and attractive patina make it a popular choice for sculptures, monuments, and memorial plaques, which are meant to last for centuries.
Musical Instruments
Its unique acoustic properties make bronze the preferred material for making bells, cymbals, and other musical instruments, providing a rich, resonant sound.
Bearings and Bushings
The alloy’s low friction and resistance to wear and tear are ideal for manufacturing bearings, bushings, and similar components in machinery and vehicles.
Coins and Medals
Historically, bronze has been used to mint coins and medals due to its durability and the ease with which it can be engraved and molded.
Tools and Weapons
In ancient times, bronze was the primary material for making tools, weapons, and armor, valued for its hardness and workability compared to stone or wood.
Benefits of Bronze
Durability
Bronze is highly durable, able to withstand wear, pressure, and damage, making it suitable for long-lasting applications.
Corrosion Resistance
It exhibits excellent resistance to corrosion, especially against environmental conditions like salt water, reducing maintenance needs.
Aesthetic Appeal
Bronze develops a beautiful patina over time, adding to its aesthetic appeal, which is especially valued in art and architecture.
Antimicrobial Properties
Bronze surfaces have inherent antimicrobial properties, making them beneficial for reducing the spread of germs and bacteria.
Versatility
The alloy can be easily cast into complex shapes and sizes, offering great versatility in manufacturing and artistic creation.
Acoustic Properties
Bronze is favored for musical instruments due to its superior acoustic properties, producing a clear, resonant sound.
Side Effects of Bronze
- Patina Development: Over time, bronze develops a greenish patina. While often considered desirable for art, this change might not be wanted in other applications.
- Cost: Compared to other metals like aluminum or steel, bronze can be more expensive due to the cost of copper and tin.
- Weight: Bronze is heavier than some alternative materials, which might limit its use in applications where weight is a critical factor.
- Limited Electrical Conductivity: While still conductive, bronze has lower electrical conductivity than pure copper, making it less suitable for electrical applications.
- Maintenance for Outdoor Use: In outdoor applications, especially in highly corrosive environments, bronze may require regular maintenance to preserve its appearance and structural integrity.
FAQ’S
Why is Bronze So Special?
Bronze, an alloy of copper and tin, is prized for its strength, corrosion resistance, and beautiful patina, making it ideal for art and marine uses.
Is Bronze Just Copper?
No, bronze is not just copper. It’s an alloy made primarily of copper and tin, which enhances its strength, durability, and corrosion resistance.
Why is Bronze No Longer Used?
Bronze is still used but less commonly for tools and weapons due to modern materials like steel and aluminum, which are stronger and cheaper.
Why is Bronze Expensive?
Bronze is expensive due to the cost of raw materials, copper and tin, and the process involved in alloying and casting it into usable forms.


