Copper (Cu) – Definition, Preparation, Properties, Uses, Compounds, Reactivity
Discover the versatile world of Copper, a fundamental metal pivotal in various industries, including electrical, construction, and jewelry. This comprehensive guide delves into Copper’s properties, applications, and historical significance, offering insightful examples of its utility and importance. Uncover the benefits of Copper, from its superior conductivity to its antimicrobial features, and explore how this durable, recyclable metal continues to shape technological advancements and sustainable solutions. Join us as we navigate the multifaceted applications of Copper, revealing its enduring relevance in modern society.
What is Copper?
Copper is a chemical element with the symbol Cu and atomic number 29. It is a transition metal known for its reddish-brown appearance and excellent conductivity. Copper does not tarnish easily when exposed to air, making it highly resistant to corrosion and perfect for electrical wiring. Historically, Copper has been used in coinage and alloys such as brass and bronze, and today, it finds applications in electrical equipment, plumbing, and renewable energy technologies. For teachers, Copper serves as an outstanding example to discuss the properties of elements, their position in the periodic table, and their extensive applications in daily life.
Copper Formula
Formula: Cu
Composition: A single copper atom.
Bond Type: Copper forms metallic bonds in its metallic state and can form covalent or ionic bonds in its compounds, with one or two valence electrons in the outer shell.
Molecular Structure: Exists mainly in one form as metallic copper, which is highly conductive and malleable.
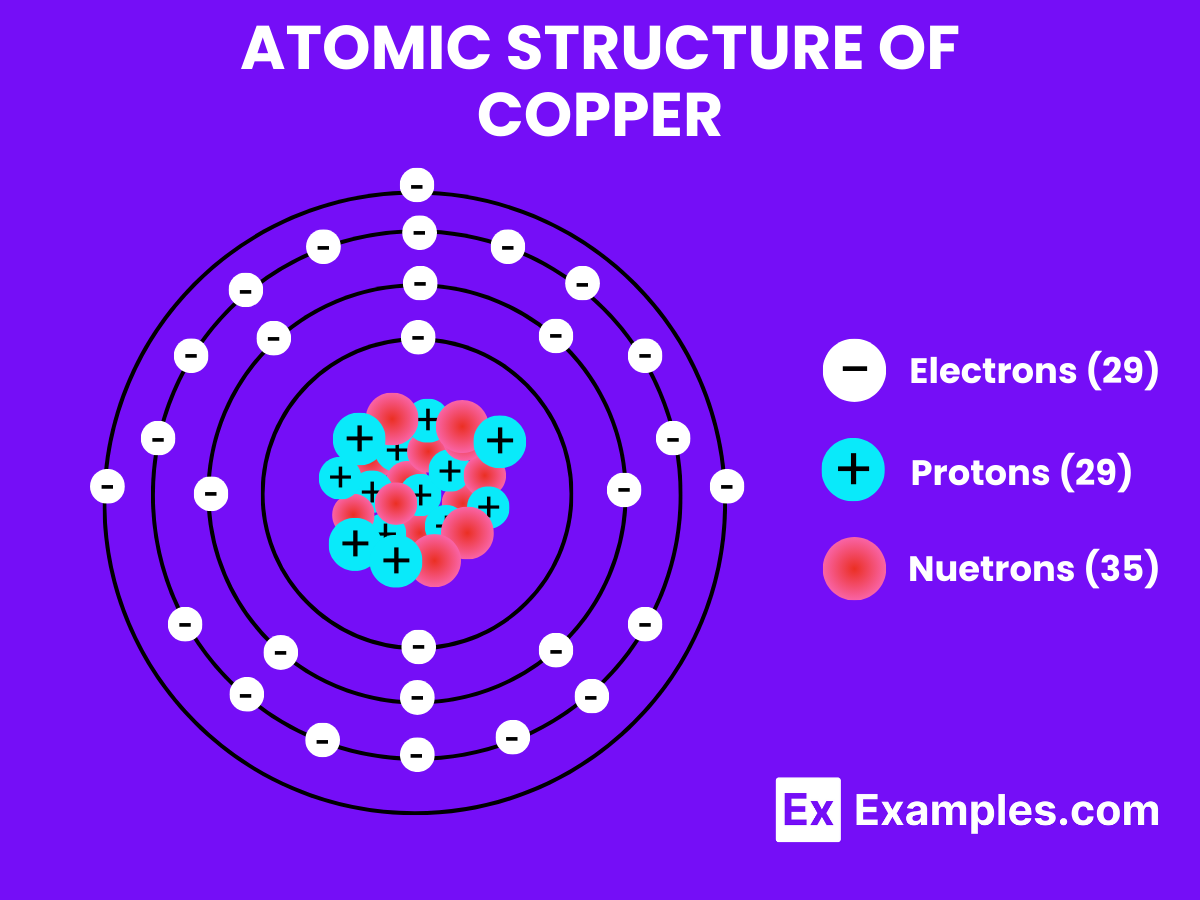
Electron Configuration: 29 electrons, with the configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s¹.
Significance: Essential for electrical wiring, plumbing, and as a component in various alloys like brass and bronze.
Role in Chemistry: Key in the study of electrical conductivity and the formation of alloys, as well as in bioinorganic chemistry due to its biological importance.
Atomic Structure of Copper
- Crystal Structure: Copper has a face-centered cubic (FCC) lattice structure. In this arrangement, each copper atom is at the center of a cube and is surrounded by eight other atoms at the corners, with closest interactions with the four atoms in the face centers of the cube.
- Coordination Number: The coordination number in copper’s FCC structure is 12. This means each copper atom is directly surrounded by and touching 12 other copper atoms, facilitating its high ductility and malleability.
- Properties Enabled by Structure: The FCC lattice structure allows copper to be highly ductile and malleable. Layers of atoms can slide over each other easily, which is essential for processes like drawing copper into wires or hammering it into sheets.
- Electron Configuration: Copper’s electron configuration is [Ar] 3d¹⁰ 4s¹. It has a single electron in the 4s orbital, which can move freely between atoms, contributing to copper’s excellent electrical and thermal conductivity.
- Significance in Applications: The structural and electronic properties of copper make it ideal for electrical wiring and components, thanks to its superior electrical conductivity. Additionally, it is valuable in making alloys such as brass and bronze, enhancing strength, corrosion resistance, and thermal conductivity in these materials.
Properties of Copper
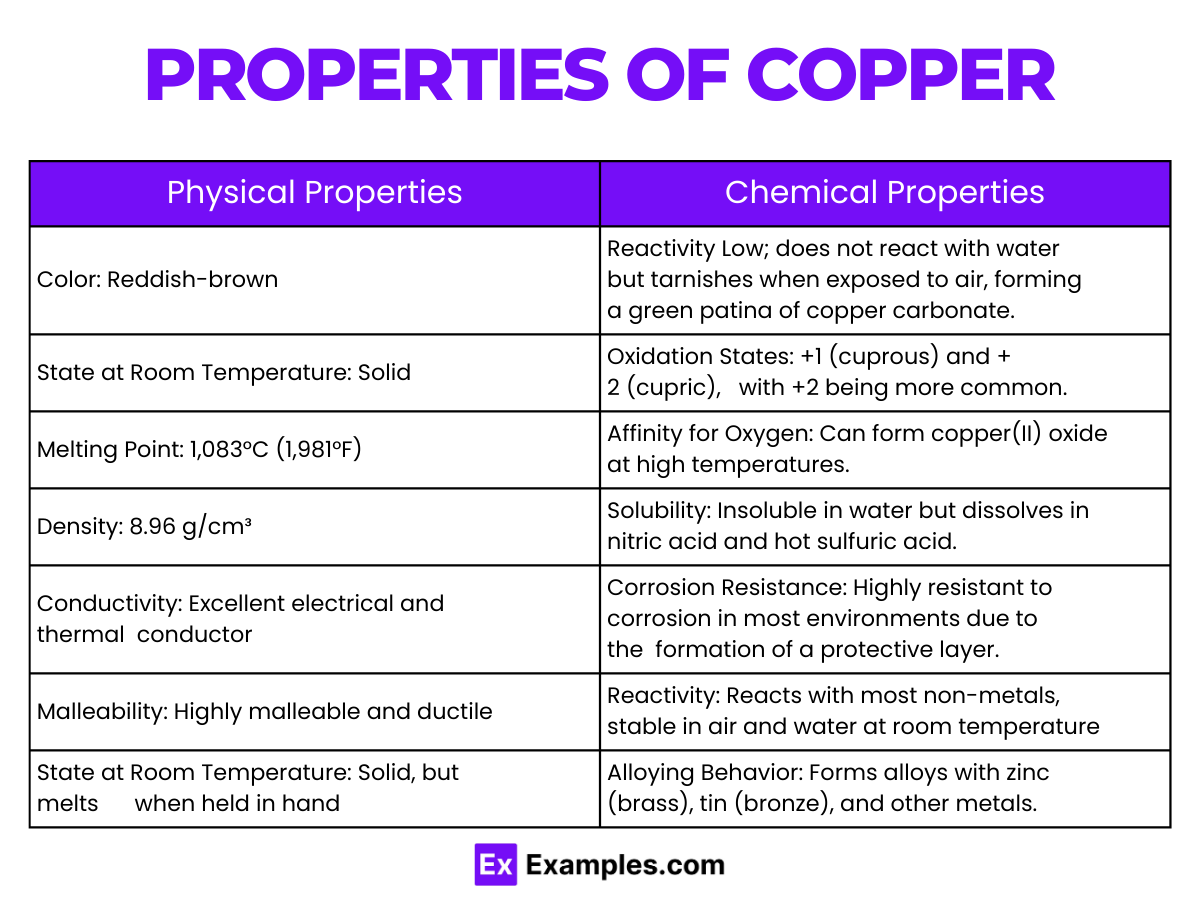
Physical Properties of Copper
| Property | Value |
|---|---|
| Appearance | Reddish-brown, metallic luster |
| Density | 8.96 g/cm³ |
| Melting Point | 1084.62°C (1984.32°F) |
| Boiling Point | 2562°C (4643.6°F) |
| State at Room Temperature | Solid |
| Thermal Conductivity | 401 W/(m·K) |
| Electrical Conductivity | 5.96 × 10⁷ S/m (at 20°C) |
| Heat Capacity | 0.385 J/(g·K) |
Chemical Properties of Copper
Copper, with the chemical symbol Cu and atomic number 29, showcases a variety of chemical properties that contribute to its wide range of applications:
- Oxidation States: Copper primarily exhibits two oxidation states, +1 (cuprous) and +2 (cupric), with +2 being the more stable and common form.
- Reaction with Oxygen: At room temperature, copper reacts slowly with atmospheric oxygen, forming a layer of brown-black copper oxide on its surface.
- Equation: 2Cu+O₂→2CuO
- Reaction with Acids: Copper is not reactive with non-oxidizing acids but reacts with oxidizing acids, such as nitric acid and sulfuric acid, to produce copper salts.
- Equation with Nitric Acid: Cu+4HNO₃→Cu(NO₃)₂+2NO₂+2H₂O
- Conductivity: Copper is known for its excellent electrical conductivity, second only to silver, making it a prime choice for electrical wiring and components.
- Alloy Formation: Copper forms alloys readily with other metals, such as bronze and brass (copper and zinc), which are harder, stronger, and more corrosion-resistant than pure copper.
- Corrosion Resistance: Besides its slow oxidation in air, copper forms a green layer of copper carbonate, known as patina, when exposed for long periods, which protects the metal from further corrosion.
Thermodynamic Properties of Copper
| Property | Value |
|---|---|
| Melting Point | 1,083°C (1,981°F) |
| Boiling Point | 2,567°C (4,653°F) |
| Specific Heat Capacity | 0.385 J/g·K |
| Thermal Conductivity | 401 W/m·K |
| Heat of Fusion | 13.26 kJ/mol |
| Heat of Vaporization | 300.4 kJ/mol |
Material Properties of Copper
| Property | Value |
|---|---|
| Density | 8.96 g/cm³ |
| Young’s Modulus | 110-128 GPa |
| Tensile Strength | 210-220 MPa |
| Hardness (Brinell) | 874 MPa |
| Elastic Modulus | 117 GPa |
| Poisson’s Ratio | 0.34 |
Electromagnetic Properties of Copper
| Property | Value |
|---|---|
| Electrical Conductivity | 5.96×10⁷ S/m |
| Electrical Resistivity | 1.68×10⁻⁸ Ω·m |
| Magnetic Susceptibility | -9.6×10⁻⁶ cm³/mol (diamagnetic) |
Nuclear Properties of Copper
| Property | Value |
|---|---|
| Natural Isotopes | Cu-63 (69.17%), Cu-65 (30.83%) |
| Neutron Cross Section | 3.78 barns (for Cu-63) |
| Neutron Mass Absorption | 0.0 |
Preparation of Copper
The preparation of copper involves several key steps to extract and refine the metal from its ores, primarily from chalcopyrite (CuFeS₂), the most abundant copper ore. The process encompasses:
- Mining: Copper ore is extracted from open pit or underground mines, depending on the depth of the ore.
- Concentration: The ore is crushed and ground, followed by froth flotation to separate copper minerals from other substances in the ore, producing copper concentrate.
- Roasting: Copper concentrate is then roasted in the presence of air to convert sulfides into oxides: 2CuFeS₂+O₂→Cu₂S+2FeS+SO₂
- Smelting: The roasted concentrate is smelted in a furnace, where it reacts with silica to form a slag of iron silicates, and copper matte, a mixture of copper sulfide and iron sulfide:
- Conversion: Copper matte is then processed in a converter furnace, where it is blown with air or oxygen to remove sulfur and iron, leaving behind blister copper (roughly 98% copper): 2Cu₂S+3O₂→2Cu₂O+2SO₂
Cu₂O+Cu₂S→4Cu+SO₂ - Electrorefining: Blister copper is further purified through electrorefining, where it is made the anode in an electrolytic cell, with copper sulfate as the electrolyte. Pure copper plates out onto the cathode, achieving purity levels of 99.99%
Chemical Compounds of Copper
- Copper(II) Oxide (CuO)
- Description: A black powder that is insoluble in water but soluble in ammonium chloride solution. It is used as a pigment in ceramics to produce blue, green, and red colors and in the manufacture of rayon fabric.
- Equation:
- Copper(I) Oxide (Cu2O)
- Description: A red to brownish powder, used as a fungicide and in antifouling paints for boats to prevent algae growth. It also serves as a significant product in the Cuprous oxide semiconductor industry.
- Equation:
- Copper(II) Sulfate (CuSO4)
- Description: Also known as blue vitriol, it is a blue crystalline solid used in agriculture as a fungicide, in water treatment as an algaecide, and in the chemical industry for the production of other copper compounds.
- Equation:
- Copper(II) Chloride (CuCl2)
- Description: A yellowish-brown solid that dissolves in water to give a blue-green solution, used in wood preservation, copper electroplating baths, and as a catalyst in organic synthesis.
- Equation: 2
- Copper(I) Chloride (CuCl)
- Description: A white powder that is used in the manufacturing of pigments, in the production of organic dyes, and as a catalyst in various chemical reactions.
- Equation:
- Copper(II) Nitrate (Cu(NO3)2)
- Description: A blue crystalline solid that dissolves in water to form a blue solution, used in ceramics to produce blue and green glazes, as a catalyst in organic reactions, and in textile dyeing.
- Equation:
Isotopes of Copper
| Isotope | Natural Abundance | Half-Life | Mode of Decay |
|---|---|---|---|
| Cu-63 | 69.15% | Stable | N/A |
| Cu-65 | 30.85% | Stable | N/A |
| Cu-64 | Trace | 12.7 hours | β+ decay to Ni-64, β- decay to Zn-64, Electron capture |
| Cu-67 | Synthetic | 61.83 hours | β- decay to Zn-67 |
Copper has two stable isotopes, Cu-63 and Cu-65, which are found naturally in the environment. The other isotopes, like Cu-64 and Cu-67, are radioactive and produced synthetically in laboratories. These isotopes have applications in medicine and scientific research.
Uses of Copper
Copper is a versatile metal with a wide range of applications, thanks to its excellent electrical and thermal conductivity, corrosion resistance, and malleability. Some of the most significant uses of copper include:
- Electrical Wiring and Components: Due to its outstanding electrical conductivity, copper is widely used in electrical wiring, motors, and other electrical devices.
- Plumbing and Roofing: Copper’s durability and resistance to corrosion make it ideal for plumbing pipes, fittings, and roofing materials.
- Heat Exchangers and Cooking Utensils: Its excellent thermal conductivity makes copper a preferred choice for heat exchangers in HVAC systems and cookware.
- Alloys: Copper is used to make various alloys, including bronze (copper and tin) and brass (copper and zinc), which are stronger and more resistant to corrosion than pure copper.
- Electronics and Telecommunications: Copper is used in the manufacturing of electronic devices and components, including circuit boards, due to its conductivity.
- Renewable Energy: In solar panels, wind turbines, and other renewable energy systems, copper plays a critical role in the distribution and generation of electrical energy.
- Medicine: Copper has antimicrobial properties and is used in medical devices and as a supplement to prevent or treat copper deficiency.
- Art and Architecture: Copper and its alloys are used in sculptures, decorative art, and architectural elements for their aesthetic appeal and durability.
Copper Production and Reserves
| Country | Mine Production 2016 (Metric Tons)* | % of World Mine Production | Demonstrated Reserves 2016 (Metric Tons)* | % of World Demonstrated Reserves |
|---|---|---|---|---|
| Chile | 5,500,000 | 26.9% | 170,000,000 | 28.0% |
| Peru | 2,350,000 | 11.5% | 81,000,000 | 13.3% |
| China | 1,740,000 | 8.5% | 30,000,000 | 4.9% |
| United States | 1,410,000 | 6.9% | 35,000,000 | 5.7% |
| Congo (DRC) | 1,030,000 | 5.0% | 20,000,000 | 3.3% |
| Australia | 970,000 | 4.7% | 88,000,000 | 14.5% |
| Russia | 710,000 | 3.5% | 30,000,000 | 4.9% |
| Zambia | 708,000 | 3.4% | 20,000,000 | 3.3% |
| Canada | 696,000 | 3.4% | 11,000,000 | 1.8% |
| Mexico | 575,000 | 2.8% | 38,000,000 | 6.2% |
Production of Copper
The production of copper involves a series of processes that extract the metal from its ores and refine it to a usable purity level. The main steps include:
- Mining: Copper ore is extracted from the earth through open-pit or underground mining methods.
- Crushing and Grinding: The ore is then crushed and ground to liberate the copper minerals from the surrounding rock.
- Concentration: Through flotation, the ground ore is mixed with water to form a slurry, where copper minerals attach to bubbles and are skimmed off as a froth, concentrating the copper content.
- Smelting: The concentrate is smelted to produce matte copper (a mixture of copper, iron sulfides).
- Converter Process: The matte is further processed in a converter, where it is blown with air or pure oxygen to remove the iron and sulfur, producing blister copper (roughly 98-99% pure).
- Electrorefining: Blister copper undergoes electrorefining, where it is used as an anode in an electrolytic cell. Pure copper is deposited on the cathode, achieving a purity of 99.99%.
- Casting: The refined copper is then cast into various shapes, such as rods, billets, or cakes, for further processing or direct use.
Applications of Copper
Copper’s properties, such as high conductivity, ductility, and corrosion resistance, make it suitable for a wide range of applications:
- Electrical Conductors: Copper is the standard material for electrical wiring and cables due to its excellent electrical conductivity.
- Construction: It’s used in plumbing, roofing, and cladding because of its durability and resistance to corrosion.
- Heat Exchangers: Copper’s superior thermal conductivity makes it ideal for heat exchangers in HVAC systems and industrial processes.
- Electronics: Found in electronic devices and circuit boards, copper enables efficient operation of a wide range of technologies.
- Transportation: Copper is used in the automotive and aerospace industries for wiring, motors, and batteries.
- Renewable Energy: Essential in the construction of solar panels, wind turbines, and related infrastructure due to its conductive properties.
- Coinage: Many countries use copper alloys, such as bronze and brass, for making coins.
- Antimicrobial Applications: Copper surfaces are used in healthcare settings to reduce the transmission of pathogens.
This article provided a comprehensive exploration of copper, highlighting its extraction, refining process, isotopes, and wide-ranging applications. Copper’s remarkable properties make it indispensable in electrical wiring, construction, renewable energy, and more. Understanding copper’s production and multifaceted uses showcases its vital role in advancing technology and supporting modern infrastructure, emphasizing its significance in our daily lives and future developments.