Toluene – Definition, Structure, Preparation, Properties, Uses, Benefits, Side Effects
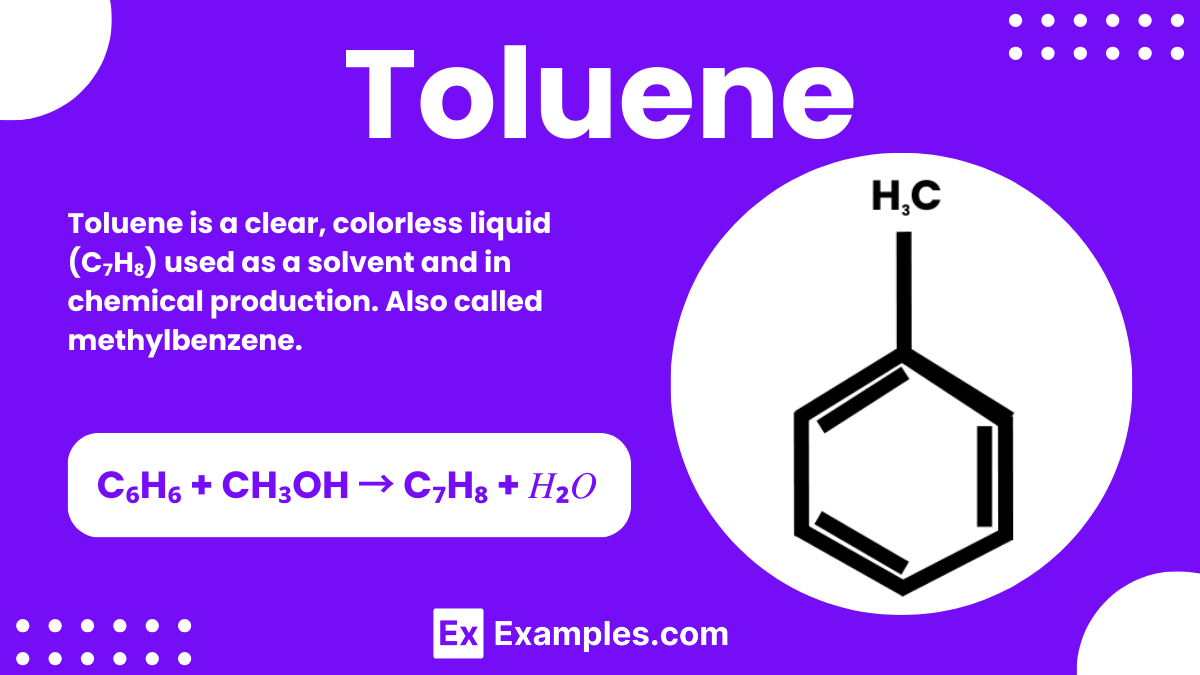
Toluene is a clear, colorless liquid commonly used in chemistry as a solvent and an important starting material for making other compounds. It has a sweet, pungent smell and is found in paint thinners, nail polish removers, and some types of glue. Chemists often work with toluene because it can dissolve many substances without reacting with them. Being a hydrocarbon, it consists of a benzene ring with a single methyl group attached, making it a key compound in organic chemistry.
What is Toluene?
Chemical Names and Formulas
| Property | Value |
|---|---|
| Formula | C₆H₅CH₃ |
| Hill Formula | C₇H₈ |
| Name | Toluene |
| IUPAC Name | Methylbenzene |
| Alternate Names | Meth Acid E, Methane Phenyl-, Methylbenzene, Methylbenzol, Phenylmethane, Toluol |
Structure of Toluene
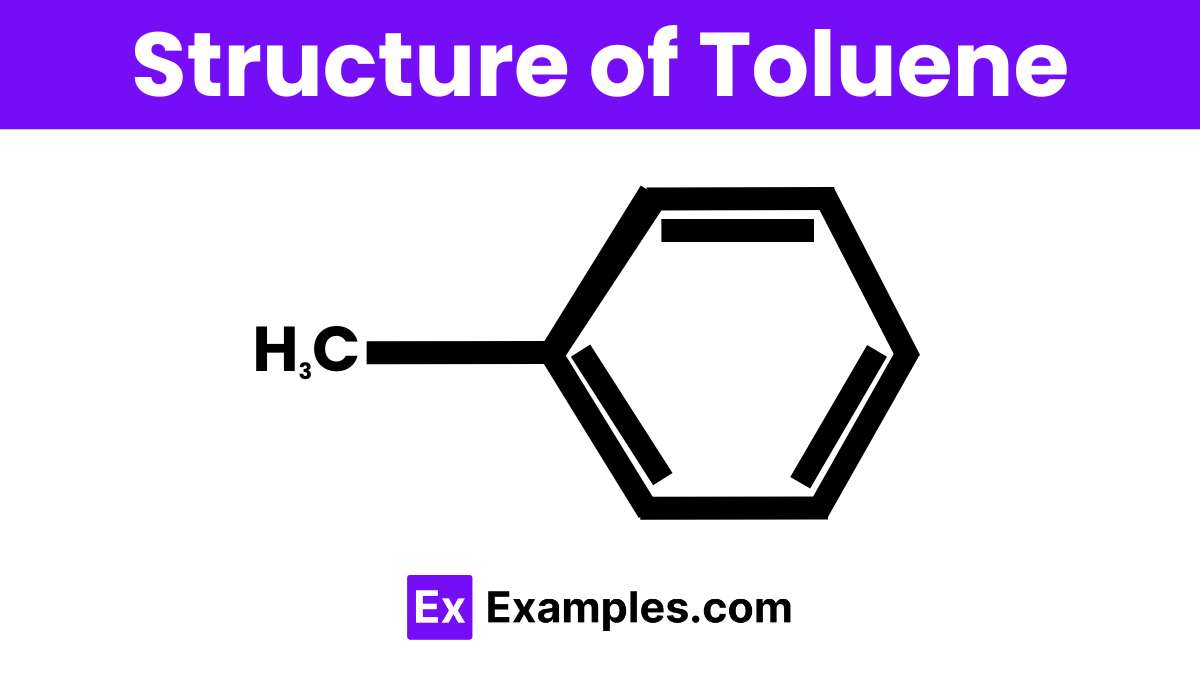
Toluene has a simple and distinctive structure consisting of a benzene ring bonded to a single methyl group (CH₃). This means that six carbon atoms form a hexagonal ring with alternating double bonds, while one carbon of this ring is attached to a group of three hydrogen atoms. This structure gives toluene its chemical properties and makes it an important compound in various industrial applications.
Preparation of Toluene
To prepare toluene, we primarily use the catalytic reforming of petroleum naphtha. In this process, you heat and pressurize naphtha in the presence of a catalyst, usually platinum or rhenium, which rearranges the hydrocarbons to produce toluene. The reaction is efficient and commonly used in the petrochemical industry to obtain toluene from crude oil derivatives.
Another method involves the methylation of benzene. By reacting benzene (C₆H₆) with methanol (CH₃OH) in the presence of a catalyst, toluene (C₇H₈) is produced along with water. The chemical equation for this reaction is:
These methods ensure the commercial availability of toluene for various applications in industries like manufacturing and pharmaceuticals.
Physical Properties of Toluene
| Property | Description |
|---|---|
| Appearance | Clear, colorless liquid |
| Odor | Distinctive, sweet smell |
| Molecular Weight | 92.14 g/mol |
| Melting Point | -95°C (-139°F) |
| Boiling Point | 110.6°C (231.1°F) |
| Density | 0.87 g/cm³ |
| Solubility in Water | Slightly soluble |
| Vapor Pressure | 28.4 mmHg at 25°C (77°F) |
Chemical Properties of Toluene
Combustion
- Toluene burns in the presence of oxygen to produce carbon dioxide and water. This reaction releases energy and is used in fuel applications.
- Equation: 𝐶7𝐻8+9𝑂2→7𝐶𝑂2+4𝐻2𝑂
Halogenation
- In the presence of a catalyst, toluene reacts with chlorine or bromine to produce benzyl chloride or benzyl bromide. This reaction involves the substitution of a hydrogen atom by a halogen atom.
- Equation: 𝐶7𝐻8+𝐶𝑙2→𝐶6𝐻5𝐶𝐻2𝐶𝑙+𝐻𝐶𝑙
Nitration
- Toluene reacts with a mixture of concentrated nitric acid and sulfuric acid to form nitrotoluene. This reaction introduces a nitro group (-NO₂) into the benzene ring.
- Equation: 𝐶7𝐻8+𝐻𝑁𝑂3→𝐶7𝐻7𝑁𝑂2+𝐻2𝑂
Sulfonation
- Toluene reacts with concentrated sulfuric acid to form toluenesulfonic acid. This reaction introduces a sulfonic acid group (-SO₃H) into the benzene ring.
- Equation: 𝐶7𝐻8+𝐻2𝑆𝑂4→𝐶7𝐻7𝑆𝑂3𝐻+𝐻2𝑂
Toluene Chemical Compound Information
Chemical Identifiers
| Property | Value |
|---|---|
| CAS Registry Number | 108-88-3 |
| Beilstein Number | 635760 |
| PubChem Compound ID | 1140 |
| PubChem Substance ID | 24850838 |
| SMILES Identifier | CC1=CC=CC=C1 |
| InChI Identifier | InChI=1/C7H8/c1-7-5-3-2-4-6-7/h2-6H, 1H3 |
| InChI Key | YXFVVABEGXRONW-UHFFFAOYAT |
| RTECS Number | XS5250000 |
| MDL Number | MFCD00008512 |
NFPA Label
| Property | Value |
|---|---|
| NFPA Health Rating | 2 |
| NFPA Fire Rating | 3 |
| NFPA Reactivity Rating | 0 |
Uses of Toluene
Solvent
Toluene acts as a solvent in paints, coatings, and adhesives. It effectively dissolves many organic compounds, making it ideal for these applications.
Fuel Additive
Toluene is used as a fuel additive to improve octane ratings in gasoline. This helps engines run more smoothly and efficiently.
Chemical Synthesis
Industries use toluene as a starting material in the synthesis of chemicals such as benzene, xylene, and TNT (trinitrotoluene). Its reactivity makes it valuable in producing these compounds.
Cleaning Agent
Toluene serves as a cleaning agent for machinery and electronic equipment. It removes grease and other residues effectively.
Pharmaceuticals
In the pharmaceutical industry, toluene is used as a solvent in the production of various medicines. Its purity and effectiveness make it suitable for this purpose.
Benefits of Toluene
- Effective Solvent: Toluene dissolves a wide range of organic compounds, making it valuable in producing paints, coatings, and adhesives.
- Improves Fuel Efficiency: When used as a fuel additive, toluene increases the octane rating of gasoline, helping engines run more efficiently.
- Versatile Chemical Precursor: Toluene serves as a starting material for synthesizing important chemicals like benzene, xylene, and TNT, which are crucial in various industries.
- Efficient Cleaning Agent: Toluene effectively removes grease and residues from machinery and electronic equipment, ensuring proper maintenance and functionality.
- Essential in Pharmaceuticals: Toluene acts as a solvent in the pharmaceutical industry, aiding in the production of medicines and ensuring their purity.
- Supports Industrial Manufacturing: Toluene’s properties make it an important component in manufacturing processes, contributing to the production of a wide range of products.
Side Effects of Toluene
- Respiratory Issues: Inhaling toluene vapors can cause throat irritation, coughing, and shortness of breath.
- Central Nervous System Effects: Exposure to toluene can lead to headaches, dizziness, and nausea. Prolonged exposure may result in neurological damage.
- Skin Irritation: Direct contact with toluene can cause skin irritation, redness, and dryness.
- Eye Irritation: Toluene vapors or direct contact can irritate the eyes, leading to redness and watering.
- Liver and Kidney Damage: Long-term exposure to toluene can harm the liver and kidneys, affecting their function.
- Developmental Effects: Pregnant women exposed to high levels of toluene may risk developmental problems for their unborn child.
Is toluene legal?
Yes, toluene is legal and widely used in industrial applications. However, regulations control its usage and exposure due to its potential health risks.
What happens if you drink toluene?
Drinking toluene can cause severe health issues, including stomach pain, nausea, dizziness, and central nervous system damage. It requires immediate medical attention.
What does toluene do to the human body?
Toluene can affect the central nervous system, causing symptoms like headaches, dizziness, and nausea. Long-term exposure may lead to more serious health issues, including organ damage.
How flammable is toluene?
Toluene is highly flammable. It can easily ignite and burn, making it essential to handle it with care and store it away from open flames and heat sources.
How poisonous is toluene?
Toluene is toxic if inhaled, ingested, or absorbed through the skin. Prolonged exposure can cause significant health problems, including liver and kidney damage, and requires careful handling.
What happens when toluene is mixed with water?
Toluene is only slightly soluble in water. When mixed, it forms a separate layer, with toluene floating on top due to its lower density.
Is toluene hallucinogenic?
People are exposed to toluene through inhalation of fumes, skin contact, or ingestion. Common sources include industrial solvents, paints, and gasoline.
Is toluene in cigarettes?
Yes, toluene is one of the many chemicals found in cigarette smoke. It contributes to the harmful effects of smoking on health.
Can you smell toluene?
Yes, you can smell toluene. It has a distinct, sweet, and pungent odor, which makes it noticeable even at low concentrations.