Laws of Atomic and Molecular Physics – Definition, History, Rydberg Formula, Applications
Atomic and Molecular Physics is a fascinating branch of physics that delves into the laws governing the behavior of atoms and molecules. These fundamental particles are the building blocks of all matter, and understanding their interactions is crucial for grasping how the universe works at a microscopic level. The laws of physics applied in this field help explain phenomena ranging from the structure of the periodic table to the principles of quantum mechanics. By studying atomic and molecular physics, we gain insights into the forces that bind electrons to nuclei and how energy levels affect chemical reactions and physical properties of materials. This area of physics not only enriches our knowledge of the natural world but also drives innovations in technology, medicine, and environmental science.
What are Laws of Atomic and Molecular Physics ?
History of Laws of Atomic and Molecular Physics
The history of the laws of atomic and molecular physics begins in the early 20th century. When scientists started to uncover the mysteries of atomic structures. In 1911, Ernest Rutherford proposed the nuclear model of the atom. He revealed that atoms have a dense nucleus surrounded by orbiting electrons. This model set the stage for Niels Bohr in 1913. He introduce the Bohr model. He proposed that electrons travel in specific orbits and that the energy levels of these orbits are quantized.
Building on these foundational ideas, quantum mechanics emerged in the 1920s with pivotal contributions from Werner Heisenberg and Erwin Schrödinger. They developed theories that mathematically described the behavior of electrons in atoms. The Pauli Exclusion Principle, introduced by Wolfgang Pauli in 1925, further explained electron arrangements in atoms. During the mid-20th century, the development of quantum field theory and advancements in molecular physics, including the understanding of chemical bonds and molecular structures. Through VSEPR theory and orbital hybridization, enriched our understanding of molecular interactions. These discoveries collectively form the core of modern atomic and molecular physics, profoundly impacting technology, chemistry, and material sciences.
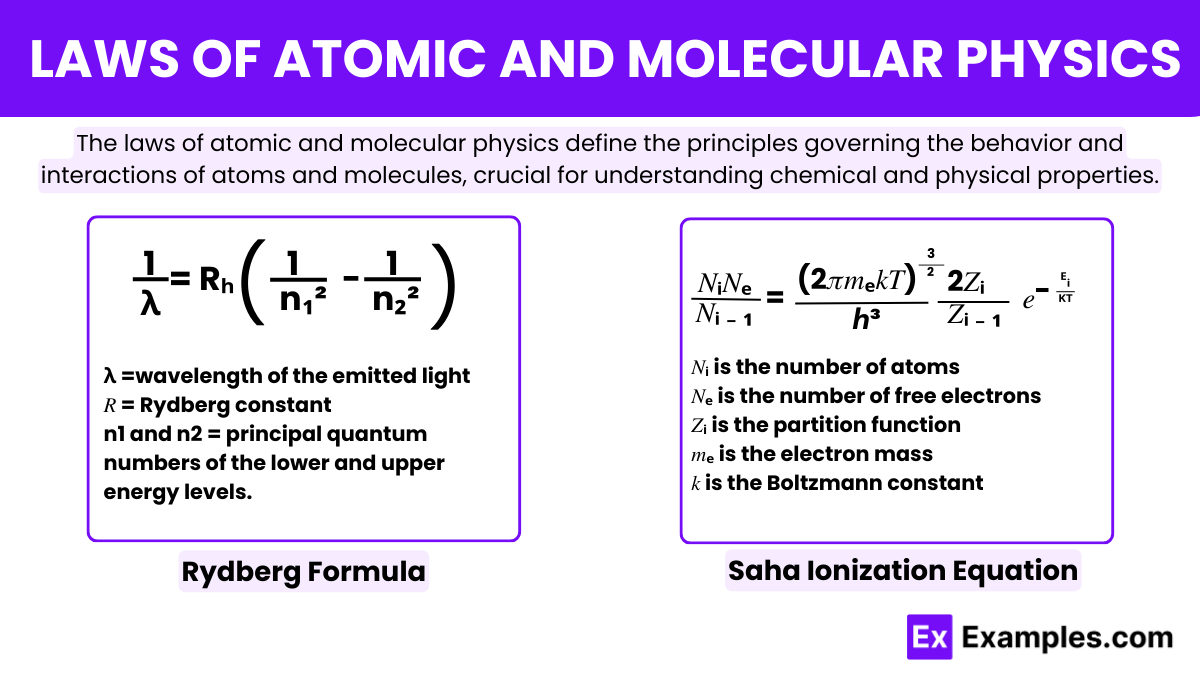
Rydberg Formula
The Rydberg formula is a fundamental equation used to predict the wavelengths of spectral lines in many chemical elements, particularly hydrogen. Proposed by the Swedish physicist Johannes Rydberg in 1888. This formula calculates the wavelengths of the electromagnetic radiation emitted when an electron in an atom transitions between energy levels. The formula is expressed as
- λ is the wavelength of the emitted light
- 𝑅 is the Rydberg constant
- n1 and n2 are the principal quantum numbers of the lower and upper energy levels.
The Rydberg formula was initially derived for hydrogen and provided an empirical way to describe the spectral patterns observed in hydrogen’s emission spectra. These patterns, known as the Balmer series, Lyman series, and other related series, were critical in the development of atomic theory. Each series corresponds to electrons falling to lower energy levels from higher ones, emitting light at specific wavelengths.
Importantly, the Rydberg formula’s adaptability extends to other elements with single-electron atoms under similar conditions, highlighting its utility in the broader context of quantum mechanics. It underscores the quantized nature of electron transitions, a cornerstone concept in quantum physics, influencing further development in theoretical and experimental physics.
Saha Ionization Equation
It was developed by Indian Astrophysicist Meghnad Saha in 1920. It plays a crucial role in astrophysics by describing how the ionization state of the gases in stars depends on the temperature and pressure. This equation links the temperature and pressure of a gas to the ratio of the number densities of ions and electrons to neutral atoms. It allows scientists to understand the physical conditions of stars and the ionization of elements at various temperatures and pressures found in stellar atmospheres.
The equation itself is derived from the principles of Thermodynamics and quantum mechanics, specifically through the application of the Boltzmann distribution and the concept of ionization potential. It states that the degree of ionization is dependent on the temperature to such an extent that higher temperatures result in higher degrees of ionization. This relationship is crucial for interpreting the spectral lines of stars and thus determining their chemical composition, temperature, and evolutionary stage.
By applying the Saha Ionization Equation, astrophysicists can accurately model stellar atmospheres and predict the spectral emission lines of stars. Which are vital for the classification of star types and the study of stellar evolution. This equation has significantly enhanced our understanding of the univers. By demonstrating the intimate connection between microscopic atomic processes and the macroscopic properties of astronomical objects.
Application of Laws of Atomic and Molecular Physics
- Material Science: Researchers use these laws to design and synthesize new materials with specific properties, such as Superconductors and Nanomaterials.
- Medicine: In radiotherapy, principles of atomic physics guide the use of radiation to target and destroy cancer cells effectively.
- Electronics: Semiconductor technology relies on understanding electron behavior in materials, paving the way for advancements in computers and mobile devices.
- Chemistry: These laws assist chemists in predicting molecular structures and the outcomes of chemical reactions, crucial for pharmaceutical development.
- Environmental Science: By analyzing molecular interactions, scientists develop better catalysts that help in reducing pollutants and improving air quality.
- Astronomy: Atomic and molecular physics underpin the analysis of light from stars and galaxies. By helping astronomers determine their composition and distances.
- Energy Production: The principles are crucial in nuclear energy generation. The atomic reactions are harnessed to produce large amounts of power.
FAQs
What are the 4 atomic laws?
The four atomic laws are the Law of Conservation of Mass, the Law of Definite Proportions, Dalton’s Law, and Avogadro’s Law.
What are the laws of atomic theory?
Atomic Theory’s laws comprise Dalton’s Postulates. That elements consist of atoms, atoms of one element are identical, atoms rearrange in reactions, and compounds have atom ratios.
What is the difference between atomic physics and molecular physics?
Atomic physics studies individual atoms and their constituents. While Molecular physics focuses on molecular structures, bonds, and dynamics involving multiple atoms.