Density – Examples, Formula, Applications, Importance
Density is a fundamental concept in chemistry, representing the mass of a substance per unit volume. It is crucial in understanding material properties and behavior. The density formula is expressed as p = M/V where ( P ) is density, ( m ) is mass, and ( V ) is volume. In electrochemistry, charge density is important, defined as the electric charge per unit area or volume, calculated using the charge density formula σ = Q/A or P = Q/V
What Is Density?
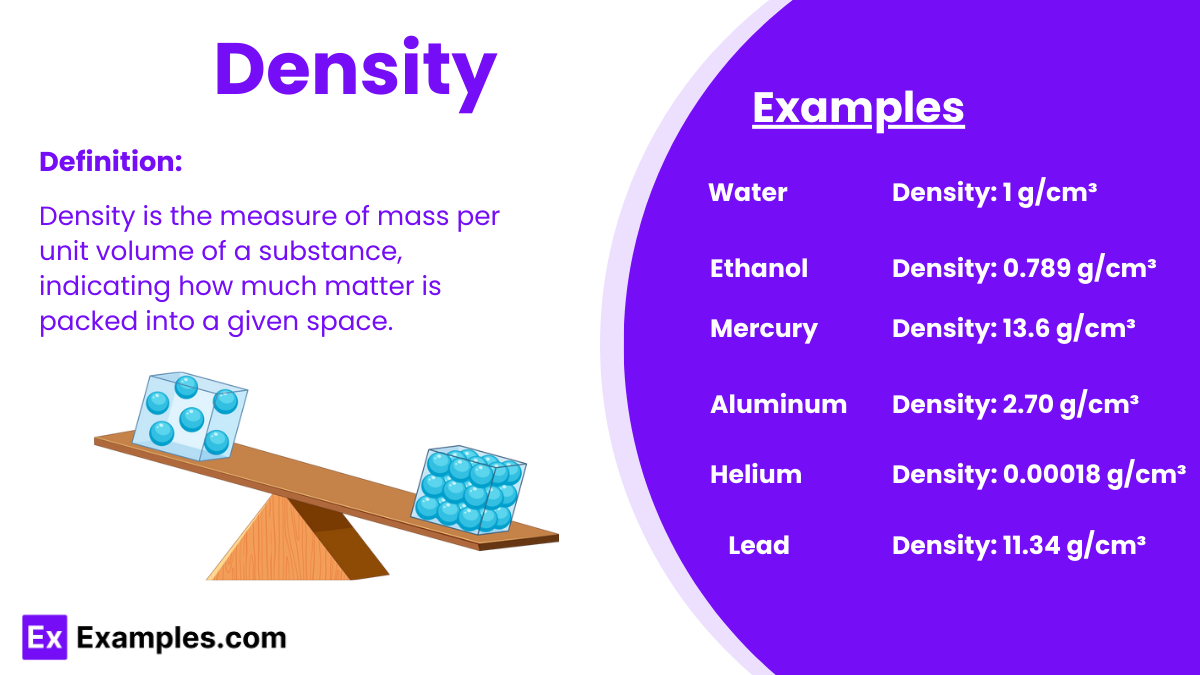
Density is a measure of mass per unit volume of a substance. It is calculated by dividing an object’s mass by its volume and is typically expressed in units of grams per cubic centimeter (g/cm³) or kilograms per cubic meter (kg/m³).
Density Examples

1. Water
- Density: 1 g/cm³
- Water is used as a standard for measuring the density of other substances.
2. Air
- Density: 0.0012 g/cm³
- The low density of air makes it suitable for respiration and weather phenomena.
3. Iron
- Density: 7.87 g/cm³
- Iron high density makes it ideal for construction and manufacturing.
4. Gold
- Density: 19.32 g/cm³
- Gold high density contributes to its use in jewelry and electronics.
5. Oil
- Density: 0.92 g/cm³
- Oil is less dense than water, causing it to float and separate in mixtures.
6. Lead
- Density: 11.34 g/cm³
- Lead density makes it suitable for radiation shielding and batteries.
7. Ethanol
- Density: 0.789 g/cm³
- The density of ethanol affects its behavior in alcoholic beverages and fuel.
8. Mercury
- Density: 13.6 g/cm³
- Mercury density and liquid state at room temperature make it useful in thermometers and barometers.
9. Aluminum
- Density: 2.70 g/cm³
- Aluminum moderate density is perfect for lightweight construction materials.
10. Helium
- Density: 0.00018 g/cm³
- The extremely low density of helium allows its use in balloons and airships.
Density Formula
Density(ρ) = Mass(m)/Volume(V)
- Density (P): This represents the density of the substance and is typically measured in kilograms per cubic meter (kg/m³) or grams per cubic centimeter (g/cm³).
- Mass (m): This is the total amount of matter contained in the object, measured in kilograms (kg) or grams (g).
- Volume (V): This is the space that the object occupies, measured in cubic meters (m³) or cubic centimeters (cm³).
How Is Density Calculated?
1. Measure the Mass
Determine the mass (( m )) of the object using a balance or scale. Ensure the mass is measured in grams (g) or kilograms (kg).
2. Measure the Volume
Determine the volume (( V )) of the object. For regular shapes, use mathematical formulas (e.g., ( V = l x w x h ) for a rectangular prism). For irregular shapes, use water displacement in a graduated cylinder to find the volume in cubic centimeters (cm³) or cubic meters (m³).
3. Convert Units if Necessary
Ensure that the mass and volume are in compatible units. For example, if mass is in grams, volume should be in cubic centimeters (cm³). If mass is in kilograms, volume should be in cubic meters (m³).
4. Apply the Density Formula
Use the density formula:
[ p = m/v ]
Where:
- ( p ) is density
- ( m ) is mass
- ( V ) is volume
5. Calculate the Density
Divide the mass by the volume to obtain the density. Ensure the result is in the appropriate units (e.g., g/cm³ or kg/m³).
Units of Density
The units of density indicate how much mass is contained within a given volume. Common units include kilograms per cubic meter (kg/m³) in the International System of Units (SI) and grams per cubic centimeter (g/cm³) in the metric system. Other units like pounds per cubic foot (lb/ft³) are also used in specific contexts.
Density of Water
The density of water is a critical reference value in science and engineering. At standard temperature and pressure (STP), the density of water is:
- 1 gram per cubic centimeter (g/cm³)
- 1000 kilograms per cubic meter (kg/m³)
This value is essential for various calculations and experiments, serving as a benchmark for comparing the densities of other substances.
Applications of Density in Real Life
- Archimedes’ Principle: Density determines buoyancy, essential in shipbuilding and designing flotation devices.
- Oil and Water Separation: Density differences separate oil from water during oil spills.
- Cooking and Baking: Density affects ingredient layering and texture in cooking.
- Meteorology: Air density variations influence weather patterns and climate.
- Material Selection in Engineering: Engineers use density to balance strength and weight in structures.
- Gold Purity Testing: Density verifies gold purity and detects impurities.
- Hydrometers: Hydrometers measure liquid density, determining substance concentration.
- Medical Imaging: Density differences in tissues aid in medical imaging techniques.
Important Densities to Remember
| Substance | Density (g/cm³) | Aspects |
|---|---|---|
| Water | 1.00 | Basis for comparison, essential for life |
| Air | 0.0012 | Varies with temperature and pressure, low density |
| Gold | 19.32 | High density, valuable, used in jewelry and finance |
| Iron | 7.87 | Common metal, used in construction and manufacturing |
| Aluminum | 2.70 | Lightweight, used in aerospace and packaging |
| Mercury | 13.53 | Only liquid metal at room temperature, dense |
| Ice | 0.92 | Less dense than water, floats, important in climates |
| Granite | 2.75 | Common rock, used in construction |
| Lead | 11.34 | Dense, used in batteries and radiation shielding |
| Ethanol | 0.79 | Alcohol, less dense than water, used in fuels |
How is density calculated?
Density is calculated by dividing mass by volume.
What are the units of density?
Density is commonly expressed in g/cm³ or kg/m³.
Why is density important?
Density helps identify substances and determine their properties.
What is the density of water?
The density of water is 1.00 g/cm³.
Does temperature affect density?
Yes, density typically decreases as temperature increases.
What is the density of air?
The density of air is approximately 0.0012 g/cm³.
How does pressure affect density?
Density increases with increasing pressure.
What is the density of gold?
The density of gold is 19.32 g/cm³.
Is ice more or less dense than water?
Ice is less dense than water.
What is the density of aluminum?
The density of aluminum is 2.70 g/cm³.