Lead (Pb) – Definition, Preparation, Properties, Uses, Compounds, Reactivity
Lead, a heavy metal with significant historical and contemporary importance, is a topic of great relevance in various educational fields. Its properties, uses, and health implications offer a rich subject for teachers to explore in the classroom. This guide provides educators with practical examples and applications of Lead, making it easier to convey its complex characteristics to students. From discussing its role in batteries to examining its environmental impact, this guide is designed to enrich educational discussions about Lead, fostering a deeper understanding among learners.
What is Lead?
Lead is a dense, soft, and malleable metal with the symbol Pb (from the Latin plumbum) and atomic number 82. Historically used in a range of applications, Lead is known for its low melting point, high density, and resistance to corrosion. However, its use has decreased due to health concerns, as Lead exposure can be harmful. In the educational context, Lead serves as an important example of the intersection between chemistry, history, and environmental science, offering a multifaceted topic for teachers to delve into with their students.
Lead Formula
- Formula: Pb
- Composition: A single lead atom.
- Bond Type: Lead forms covalent or ionic bonds in compounds, utilizing its four valence electrons.
- Molecular Structure: Soft, dense metal with a bluish-white color when freshly cut, tarnishing to a dull grayish color when exposed to air.
- Electron Configuration: 82 electrons, with the configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 5d¹⁰ 6s² 6p².
- Significance: Historically used in pipes, paints, and fuels; now recognized for its toxicity and environmental impact.
- Role in Chemistry: Lead compounds are studied for their chemical, physical, and toxicological properties.
Atomic Structure of Lead
Lead, symbolized as Pb, is a chemical element with fascinating properties and extensive uses. Its atomic structure offers insight into its unique characteristics. Here’s a point-wise breakdown of lead’s atomic structure:
1. Proton Count: Each lead atom contains 82 protons in its nucleus. This proton count is responsible for its chemical properties and its position as element 82 on the periodic table.
2. Neutron Variability: Lead has varying numbers of neutrons across its isotopes, which affects its atomic mass. The most common isotopes of lead are Pb-204, Pb-206, Pb-207, and Pb-208, with neutron counts that contribute to the overall stability of each isotope.
3. Electron Configuration: Lead’s electron configuration is [Xe] 4f14 5d10 6s2 6p2, indicating it has electrons in several energy levels. This configuration is crucial for understanding lead’s chemical behavior and bonding characteristics.
4. Metallic Bonding: In solid lead, atoms are bound together in a metallic lattice structure. This structure involves a ‘sea’ of delocalized electrons that are not associated with any one atom but move freely throughout the metal. This feature is key to lead’s conductivity and malleability.
5. Atomic Mass: The atomic mass of lead varies with its isotopes, with a standard atomic weight of about 207.2 u. This variation is due to the different numbers of neutrons in each isotope.
6. Chemical and Physical Properties: The atomic structure contributes to lead’s dense, soft nature, and its low melting point. These properties, combined with its resistance to corrosion, make lead suitable for a wide range of applications.
7. Applications of Lead :Due to its atomic structure and resultant physical properties, lead is extensively used in batteries, radiation shielding, and as an additive in alloys. Its ability to absorb vibration also makes it useful in soundproofing applications.
Properties of Lead
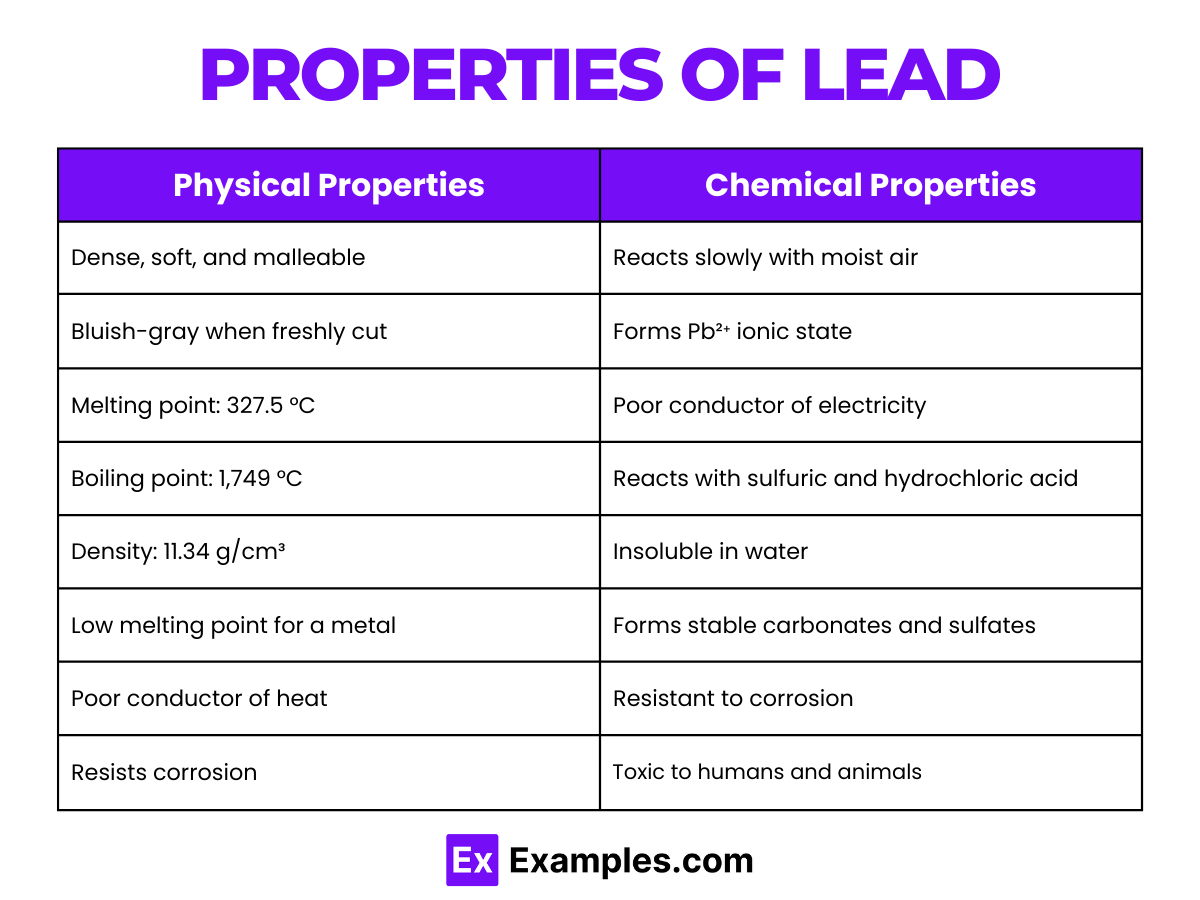
Physical Properties of Lead
| Property | Description |
|---|---|
| Appearance | Silvery, bluish-gray metal |
| Atomic Mass | Average atomic mass of approximately 207.2 u |
| Density | Approximately 11.34 g/cm³ at room temperature |
| Melting Point | 327.46°C (621.43°F) |
| Boiling Point | 1749°C (3180°F) |
| Electrical Conductivity | Poor conductor of electricity compared to other metals |
| Thermal Conductivity | Relatively low; about 35 W/(m·K) |
| Malleability and Ductility | Highly malleable and ductile; can be shaped into various forms |
| Crystal Structure | Face-centered cubic (fcc) |
Chemical Properties of Lead
Lead exhibits a range of chemical properties that are crucial for its applications and interactions with other substances. Below are some of the key chemical properties of lead:
Reactivity with Oxygen
Lead reacts with oxygen to form lead oxide. Upon exposure to air, lead forms a protective layer of oxide that inhibits further corrosion. The reaction can be represented as: 2Pb + O₂→2PbO
Reaction with Acids: Lead is relatively resistant to corrosion in sulfuric and hydrochloric acid due to the formation of a protective layer.
However, it dissolves in nitric acid, forming lead nitrate: Pb + 4HNO₃→Pb(NO₃)₂+2NO₂+2H₂O
Reaction with Sulfur: Lead reacts with sulfur to form lead sulfide, a reaction that is of particular importance in the processing of lead ores: Pb + S→PbS
Solubility: Lead compounds exhibit varied solubility in water. Lead(II) chloride (PbCl2) is sparingly soluble, while lead(II) acetate (Pb(C₂H₃O₂)₂) is soluble, making it toxic when it leaches into water supplies.
Oxidation States: Lead primarily exhibits two oxidation states: +2 (lead(II)) and +4 (lead(IV)). Lead(II) compounds are more stable and common, such as PbO, PbS, and PbCO₃. Lead(IV) compounds are strong oxidizing agents and less stable, such as PbO₂.
Environmental Impact: Lead compounds can have significant environmental and health impacts. For example, lead(II) acetate is toxic, and lead oxides can contribute to air and water pollution, affecting human health and the ecosystem.
Chemical Compounds of Lead
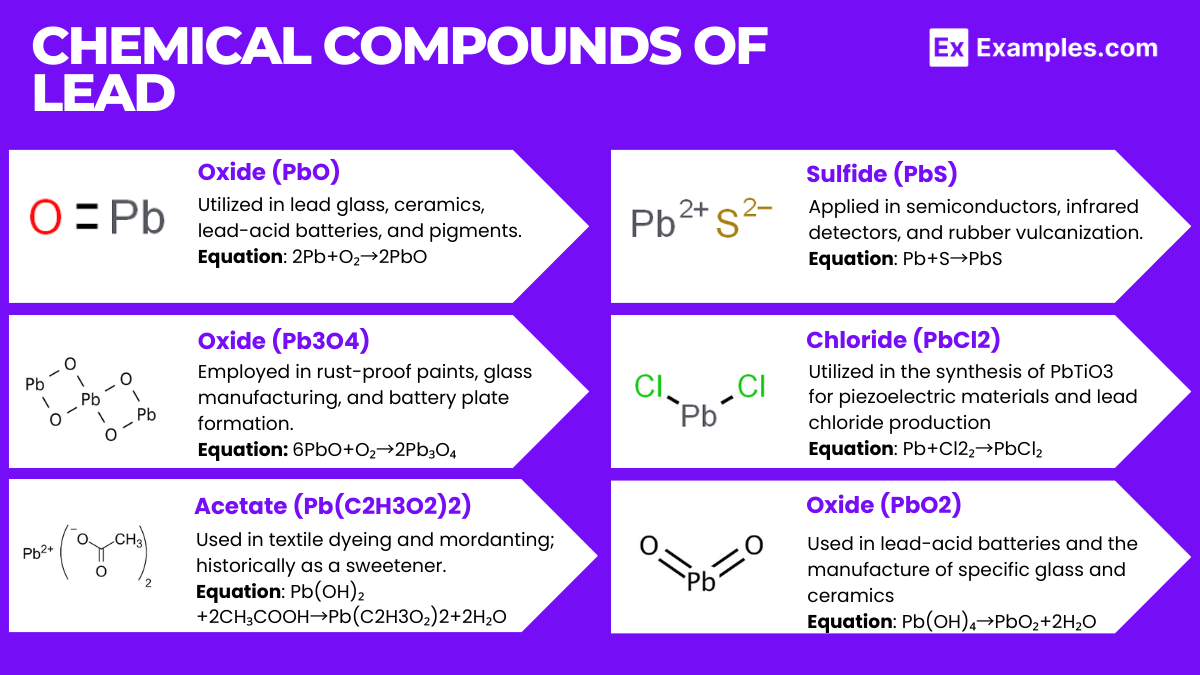
Lead(II) Oxide (PbO)
- Description: Lead(II) oxide exists in two polymorphs: litharge (red) and massicot (yellow). It is insoluble in water but soluble in acetic acid and alkaline solution.
- Chemical Equation: 2Pb+O₂→2PbO
- Uses: PbO is used in making lead glass and ceramics, in lead acid batteries as the positive plate, and in the production of pigments.
Lead(II,IV) Oxide (Pb₃O₄)
- Description: Also known as red lead or minium, this compound is a bright red or orange crystal. It is a mixed oxide that can be thought of as lead(II) oxide with lead(IV) oxide.
- Chemical Equation: 6PbO+O₂→2Pb₃O₄
- Uses: Red lead is used in rust-proof primer paints, in the manufacture of glass, and in lead acid batteries for the formation of plates.
Lead(II) Acetate (Pb(C₂H₃O₂)₂)
- Description: Known as sugar of lead, this compound forms colorless crystals or white powders and is soluble in water.
- Chemical Equation: Pb(OH)₂+2CH₃COOH→Pb(C₂H₃O₂)₂+2H₂O (Formation from lead(II) hydroxide and acetic acid)
- Uses: It’s used in the textile industry for mordanting and dyeing, and historically as a sweetener and preservative, although its use is now limited due to toxicity.
Lead(II) Sulfide (PbS)
- Description: A black, insoluble solid that occurs naturally as the mineral galena.
- Chemical Equation: Pb+S→PbS (Direct combination of lead and sulfur)
- Uses: PbS is used in semiconductors, infrared detectors, and in the vulcanization of rubber.
Lead(II) Chloride (PbCl₂)
- Description: A white insoluble solid that becomes soluble in hot water, forming a colorless solution.
- Chemical Equation: 2Pb+Cl₂→PbCl₂ (Direct reaction between lead and chlorine)
- Uses: It’s used in the synthesis of lead(IV) chloride and as a precursor for the production of lead titanate (PbTiO₃) used in piezoelectric materials.
Lead(IV) Oxide (PbO₂)
- Description: A dark-brown solid that is insoluble in water and alkali.
- Chemical Equation: Pb(OH)₄→PbO₂+2H₂O (Dehydration of lead(IV) hydroxide)
- Uses: PbO₂ is used in lead acid batteries as the active material in the positive plate and in the production of certain types of glass and ceramic glazes.
Lead(II) Carbonate (PbCO₃)
- Description: Lead(II) carbonate is a white, insoluble powder known historically as cerussite when found in its mineral form. It is sensitive to light and reacts with acids to release carbon dioxide.
- Chemical Equation: Pb²⁺+CO₃²⁻→PbCO₃ (Formation from lead(II) ions and carbonate ions)
- Uses: Historically, lead(II) carbonate was used as a white pigment in paints (known as “white lead”) and in the making of ceramics. Due to its toxicity and environmental concerns, its use has significantly declined and is now regulated.
Lead(II) Nitrate (Pb(NO₃)₂)
- Description: A white, soluble crystalline solid, lead(II) nitrate dissolves in water to give a colorless solution. It is one of the few lead compounds that is water-soluble.
- Chemical Equation: Pb+4HNO₃→Pb(NO₃)₂+2NO₂+2H₂O (Reaction of lead with nitric acid producing lead(II) nitrate, nitrogen dioxide, and water)
- Uses: Lead(II) nitrate is used in the production of matches, fireworks, and special explosives where it acts as an oxidizing agent. It also serves as a heat stabilizer in nylon and polyesters, and as a mordant in dyeing processes
Isotopes of Lead
Lead has four stable isotopes, which are crucial for various scientific and practical applications. These isotopes differ in the number of neutrons in their nuclei, giving each a unique atomic mass. The stable isotopes of lead include:
- Lead-204 (Pb-204): The least abundant of the stable lead isotopes, with a natural abundance of approximately 1.4%.
- Lead-206 (Pb-206): This isotope results from the decay of uranium-238 and is found in nature with a relative abundance of about 24.1%.
- Lead-207 (Pb-207): Formed from the radioactive decay of uranium-235, it has a natural abundance of around 22.1%.
- Lead-208 (Pb-208): The heaviest stable isotope and the end product of the thorium decay series, it is the most abundant, comprising approximately 52.4% of natural lead
Uses of Lead
Lead is utilized in various applications, benefiting from its physical and chemical properties. Some of the primary uses include:
- Batteries: Lead-acid batteries are widely used in automobiles and other vehicles for starting, lighting, and ignition purposes.
- Radiation Shielding: Due to its high density, lead is effective at absorbing radiation, making it a preferred material for protective shielding around X-ray, gamma-ray, and other radiation sources.
- Cable Sheathing: Lead’s resistance to corrosion makes it ideal for protecting cables from moisture and chemical degradation.
- Construction Materials: Lead is used in roofing, pipes, and as a pigment in paints, though its use in paint has significantly decreased due to health concerns.
- Ammunition: Lead is a common material for bullets and shot due to its high density and malleability.
- Solders and Alloys: Its low melting point and malleability make lead useful in solders, fusible alloys, and as a component in brass and bronze.
- Lead Crystal Glassware: The addition of lead oxide to glass increases its refractive index, making lead glass desirable for optical quality and decorative items
Commercial Production of Lead
The commercial production of lead primarily involves the processing of lead ore, with the two most common ores being galena (lead sulfide, PbS) and, to a lesser extent, cerussite (lead carbonate, PbCO3). The production process consists of several steps designed to extract and purify lead:
1. Mining and Ore Concentration
- Lead is usually mined through either open-pit or underground mining techniques. The ore is then concentrated, often by froth flotation, to increase the lead content before smelting.
2. Smelting
- Concentrated lead ore is roasted in air to convert lead sulfide to lead oxide (PbO), releasing sulfur dioxide (SO2) gas. Then, the lead oxide is reduced to metallic lead, typically in a blast furnace or a rotary furnace, using coke as the reducing agent.
3. Refining
- The crude lead obtained from smelting contains impurities like silver, zinc, copper, and bismuth. Refining processes are employed to remove these impurities. One common method is the Parkes process, which involves adding zinc to the molten lead. Since zinc is more reactive toward oxygen, it bonds with the silver and other metals, which can then be skimmed off the surface.
4. Alloying
- Finally, the refined lead may be alloyed with other metals to achieve specific properties required for various applications, such as increased hardness or resistance to corrosion.
This production process yields lead that is ready for commercial use in batteries, radiation shielding, and other applications
Health Effects of Lead
Lead exposure can have severe health effects, particularly on young children and pregnant women. Even at low levels, lead exposure can cause a range of health issues:
1. Neurological Impact
- Lead exposure is particularly harmful to the developing brains of children, causing reduced IQ, behavioral problems, and learning difficulties. In adults, high levels of exposure can lead to memory loss and reduced cognitive function.
2. Kidney Damage
- Chronic lead exposure can lead to kidney damage and decreased kidney function in both adults and children.
3. Blood System Effects
- Lead interferes with the production of hemoglobin, the molecule in red blood cells that carries oxygen throughout the body. This can lead to anemia.
4. Reproductive Problems
- Both men and women can experience reproductive issues as a result of lead exposure, including reduced fertility and harm to the fetus in pregnant women.
5. Cardiovascular Effects
- For adults, increased blood lead levels are associated with hypertension and an increased risk of heart disease
Lead, symbolized as Pb, is a versatile metal with significant industrial applications, from batteries to radiation shielding. Despite its utility, lead poses serious health risks, including neurological damage and cardiovascular issues. Responsible use, recycling, and minimizing exposure are crucial to harnessing lead’s benefits while protecting public health and the environment.