Zinc Sulfate – Definition, Structure, Preparation, Properties, Functions, Uses, Benefits, Side Effects
Zinc sulfate is a white crystalline salt that plays an important role in chemistry, especially in the study of compounds and reactions. It forms when zinc reacts with sulfuric acid, resulting in a compound that dissolves easily in water. Zinc sulfate is often used in various applications, including as a dietary supplement, in fertilizers to supply essential zinc to plants, and in medicine to treat zinc deficiencies. Understanding zinc sulfate helps to grasp the broader concepts of how different salts and compounds interact in chemical reactions.
What is Zinc Sulfate?
Chemical Names and Formulas
| Property | Value |
|---|---|
| Formula | ZnSO₄ |
| Hill Formula | O₄SZn |
| Name | Zinc Sulfate |
Structure of Zinc Sulfate
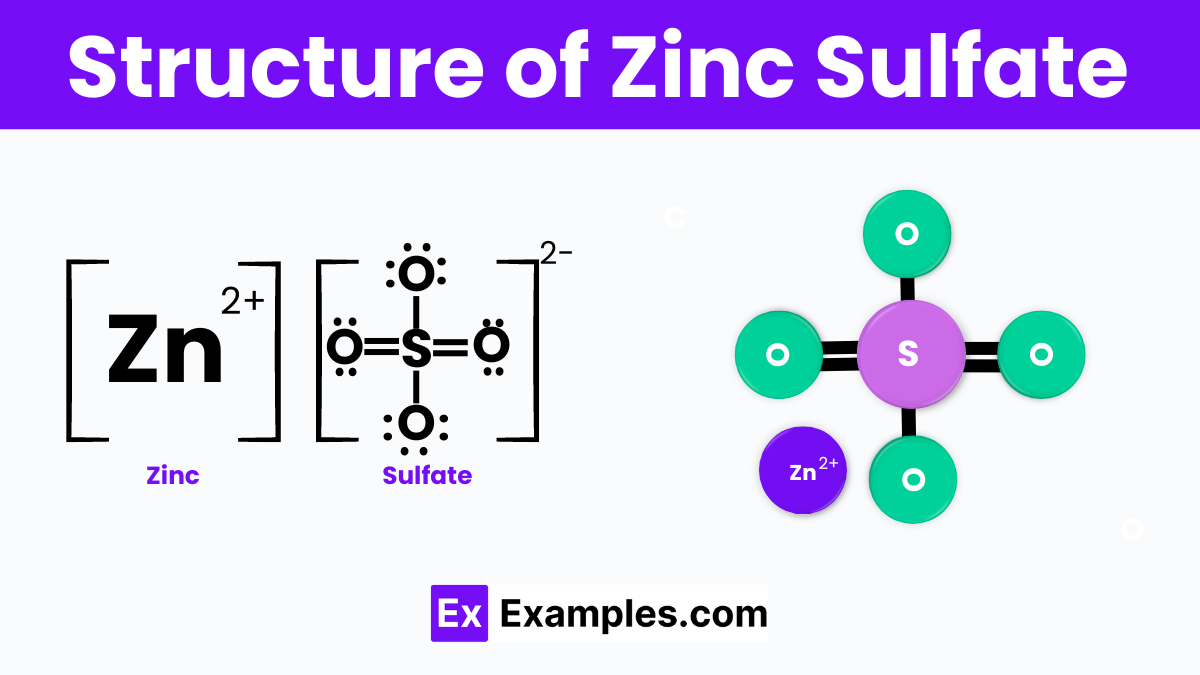
The structure of zinc sulfate consists of one zinc ion (Zn²⁺) and one sulfate ion (SO₄²⁻). In this structure, the zinc ion is bonded to the oxygen atoms of the sulfate ion. The sulfate ion has a tetrahedral shape, with the sulfur atom in the center and four oxygen atoms surrounding it. This arrangement forms a crystalline structure, where each zinc ion coordinates with oxygen atoms from nearby sulfate ions, creating a repeating lattice pattern. This structure allows zinc sulfate to dissolve easily in water, making it useful in various applications.
Preparation of Zinc Sulfate
To prepare zinc sulfate, you can react zinc with sulfuric acid. First, take a piece of zinc metal and add it to a container of dilute sulfuric acid. The zinc reacts with the sulfuric acid to produce zinc sulfate and hydrogen gas. You will notice bubbles forming as the hydrogen gas is released.
The chemical equation for this reaction is:
After the reaction, you can filter the solution to remove any unreacted zinc. Then, evaporate the water from the solution to obtain zinc sulfate crystals. This method is straightforward and is commonly used in laboratories to produce zinc sulfate.
Physical Properties of Zinc Sulfate
| Property | Description |
|---|---|
| Appearance | Colorless, crystalline solid |
| Solubility | Dissolves easily in water |
| Molecular Formula | ZnSO₄ |
| Molecular Weight | 161.47 g/mol |
| Melting Point | 680°C (decomposes) |
| Density | 3.54 g/cm³ |
| Taste | Astringent, metallic |
| Odor | Odorless |
Chemical Properties of Zinc Sulfate
Reaction with Water
Zinc sulfate dissolves easily in water, forming an aqueous solution. This property makes it useful in various applications where a liquid form is needed.
ZnSO₄ (s) → ZnSO₄ (aq)
Reaction with Bases
Zinc sulfate reacts with bases like sodium hydroxide (NaOH) to form zinc hydroxide (Zn(OH)₂) and sodium sulfate (Na₂SO₄). This reaction is an example of a double displacement reaction.
ZnSO₄ (aq) + 2NaOH (aq) → Zn(OH)₂ (s) + Na₂SO₄(aq)
Reaction with Carbonates
Zinc sulfate reacts with carbonates like sodium carbonate (Na₂CO₃) to form zinc carbonate (ZnCO₃) and sodium sulfate. This reaction produces a precipitate of zinc carbonate.
ZnSO₄ (aq) + Na₂CO₃ (aq) → ZnCO₃ (s) + Na₂SO₄ (aq)
Zinc Sulfate Chemical Compound Information
Chemical Identifiers
| Property | Value |
|---|---|
| CAS Registry Number | 7733-02-0 |
| PubChem Compound ID | 24424 |
| PubChem Substance ID | 24859179 |
| SMILES Identifier | [O-]S(=O)(=O)[O-].[Zn+2] |
| InChI Identifier | InChI=1/H2O4S.Zn/c1-5(2,3)4;/h(H2,1,2,3,4);/q;+2/p-2/fO4S.Zn/q-2;m |
| MDL Number | MFCD00011302 |
Function of Zinc Sulfate
Agricultural Use
Zinc sulfate corrects zinc deficiency in crops. Farmers apply it to soil or leaves, helping plants grow healthier and produce more.
Medical Use
Doctors use zinc sulfate as a dietary supplement to treat zinc deficiency in humans. It helps boost the immune system and improve overall health.
Industrial Use
Industries use zinc sulfate in manufacturing processes. It acts as a mordant in dyeing, a preservative for leather, and a component in various chemical products.
Water Treatment
Water treatment facilities use zinc sulfate to remove impurities. It helps clarify water by precipitating unwanted particles.
Uses of Zinc Sulfate
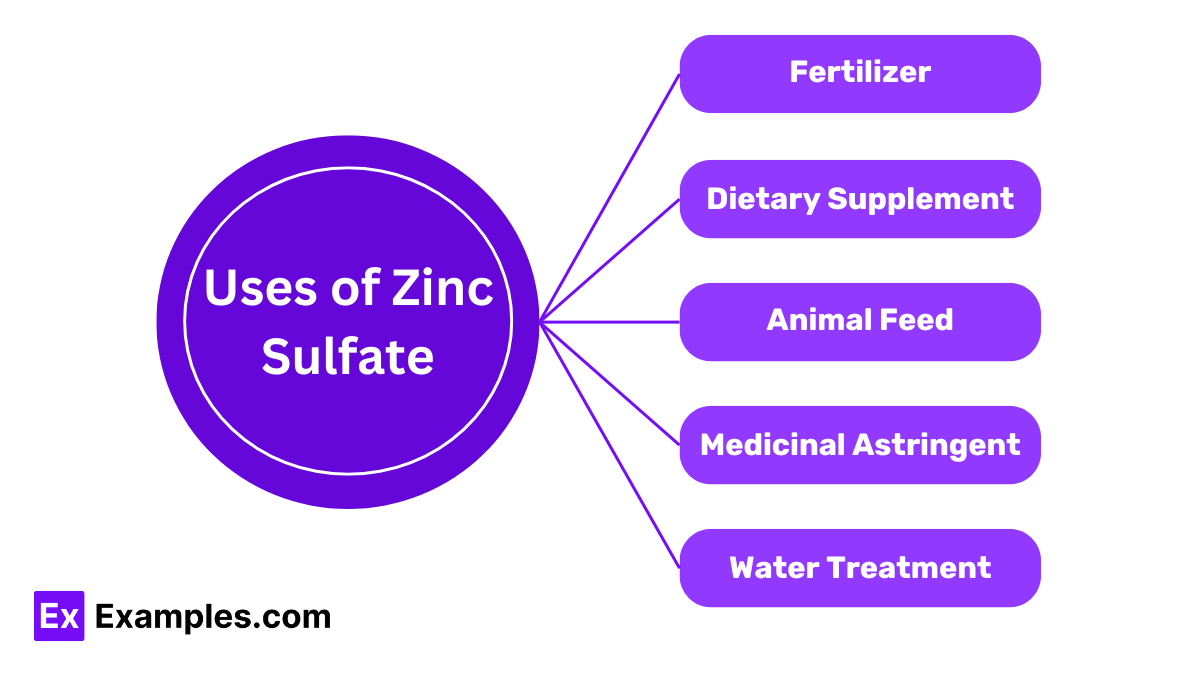
Fertilizer
Farmers use zinc sulfate as a fertilizer to correct zinc deficiencies in soil, promoting healthy plant growth and higher crop yields.
Animal Feed
Veterinarians add zinc sulfate to animal feed to ensure livestock receive essential nutrients, improving their health and growth.
Dietary Supplement
People take zinc sulfate as a dietary supplement to treat zinc deficiency, boosting the immune system and overall health.
Medicinal Astringent
Doctors use zinc sulfate as an astringent in medical treatments. It helps reduce skin irritation and inflammation.
Water Treatment
Water treatment plants use zinc sulfate to purify water. It helps remove impurities, making the water safe for consumption.
Dyeing and Printing
Textile industries use zinc sulfate as a mordant in dyeing and printing fabrics. It helps fix dyes to the fabric, ensuring vibrant and lasting colors.
Benefits of Zinc Sulfate
- Promotes Plant Growth: Zinc sulfate provides essential nutrients to plants, helping them grow stronger and healthier.
- Enhances Animal Health: Adding zinc sulfate to animal feed improves livestock health and growth by preventing zinc deficiency.
- Boosts Immune System: Taking zinc sulfate supplements helps strengthen the immune system, reducing the risk of infections.
- Improves Skin Condition: Using zinc sulfate as an astringent helps treat skin irritations and reduce inflammation.
- Purifies Water: Zinc sulfate helps remove impurities from water, making it safer for drinking and other uses.
- Enhances Textile Quality: Using zinc sulfate in dyeing and printing processes ensures that fabrics have vibrant and long-lasting colors.
Side Effects of Zinc Sulfate
- Nausea: Taking zinc sulfate can sometimes cause an upset stomach and feelings of nausea.
- Vomiting: Some individuals may experience vomiting after consuming zinc sulfate supplements.
- Diarrhea: Zinc sulfate can lead to gastrointestinal disturbances, including diarrhea.
- Stomach Pain: Users might experience abdominal discomfort or cramping.
- Metallic Taste: Taking zinc sulfate may leave a metallic taste in the mouth.
- Headache: Some people may develop headaches as a side effect of zinc sulfate.
What is zinc sulfate good for?
Zinc sulfate is good for treating zinc deficiency, promoting plant growth, enhancing animal health, purifying water, and improving skin conditions as an astringent.
Is it safe to take zinc sulfate every day?
Taking zinc sulfate daily is safe in recommended doses, but excessive intake can cause adverse effects. Always follow healthcare advice.
Why would a patient take zinc sulfate?
A patient takes zinc sulfate to treat zinc deficiency, boost the immune system, and improve overall health.
Is zinc sulfate soluble in water?
Yes, zinc sulfate is highly soluble in water, making it useful for various applications.
Who should not take zinc sulfate?
People with allergies to zinc, certain medical conditions, or those on specific medications should avoid zinc sulfate. Consult a doctor first.
What are the disadvantages of zinc sulfate?
Disadvantages of zinc sulfate include nausea, vomiting, diarrhea, stomach pain, and potential interactions with medications.
Is zinc sulfate anti-inflammatory?
Yes, zinc sulfate has anti-inflammatory properties, which help reduce skin irritation and inflammation.
What to avoid when taking zinc supplements?
Avoid taking zinc supplements with high-fiber foods, calcium, iron, or antibiotics as they can interfere with zinc absorption.
Is zinc sulfate ionic or covalent?
Zinc sulfate is an ionic compound, consisting of zinc ions (Zn²⁺) and sulfate ions (SO₄²⁻).
What is the pH of zinc sulfate?
The pH of a zinc sulfate solution is typically acidic, around 4-5.



