Osmium (Os) – Definition, Preparation, Properties, Uses, Compounds, Reactivity
Embark on an exploration of Osmium, the heaviest and densest naturally occurring element known for its remarkable hardness and luster. This comprehensive guide unveils Osmium’s definition, delves into its myriad uses from strengthening alloys to precision instruments, and uncovers the fascinating chemistry behind its compounds. As a pivotal material in science and industry, Osmium’s unique properties not only challenge our understanding of metallic behavior but also offer innovative solutions to modern technological demands.
What is Osmium?
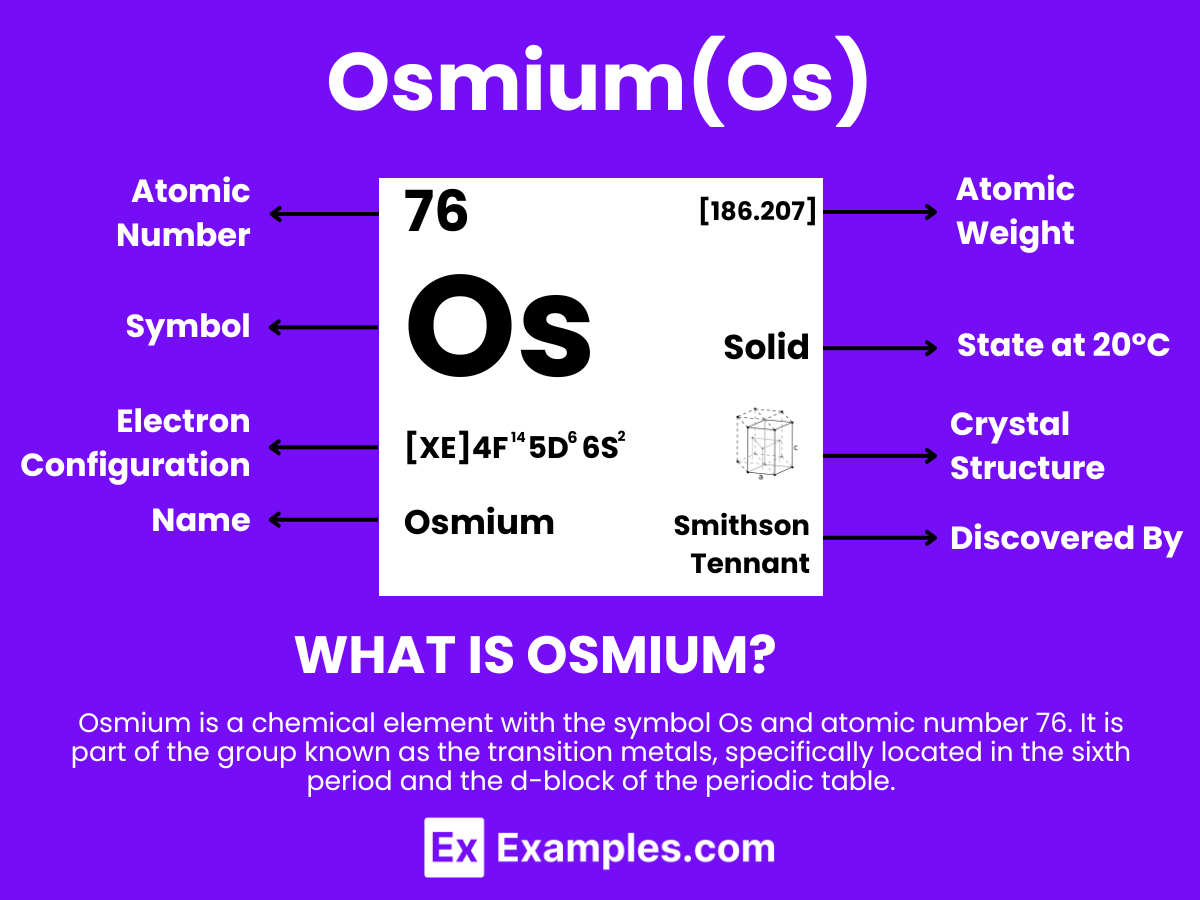
Osmium is a metallic element with the chemical symbol Os and atomic number 76. It is extracted from ores containing platinum metals, where it occurs in low concentrations. Osmium is one of the densest elements in the Earth’s crust and has one of the highest melting points of all elements, making it highly valued for its durability and resistance to wear and corrosion in extreme environments. The discovery of osmium was significant in the field of chemistry for its unique properties among the platinum group metals in the periodic table. Its exceptional density and hardness, along with its catalytic properties, make it crucial in applications requiring materials that can withstand extreme pressure and chemical reactions, such as in electrical contacts.
Osmium Formula
The elemental formula for Osmium is simply “Os”. This concise representation embodies a complex element characterized by its unmatched density and stability.
- Formula: Os
- Composition: Pure Osmium is a dense, bluish-white metal in its elemental form.
- Bond Type: Osmium forms a variety of compounds through both covalent and ionic bonds, showcasing its versatility in chemical reactions.
- Molecular Structure: Osmium metal crystallizes in a hexagonal close-packed structure, indicative of its high density and hardness.
- Compounds: Notable Osmium compounds include Osmium tetroxide (OsO₄), used in staining biological specimens and in organic synthesis, and Osmium carbide (OsC), known for its extreme hardness.
Atomic Structure of Osmium
Delving into the atomic structure of Osmium reveals why it’s the densest element on the periodic table:
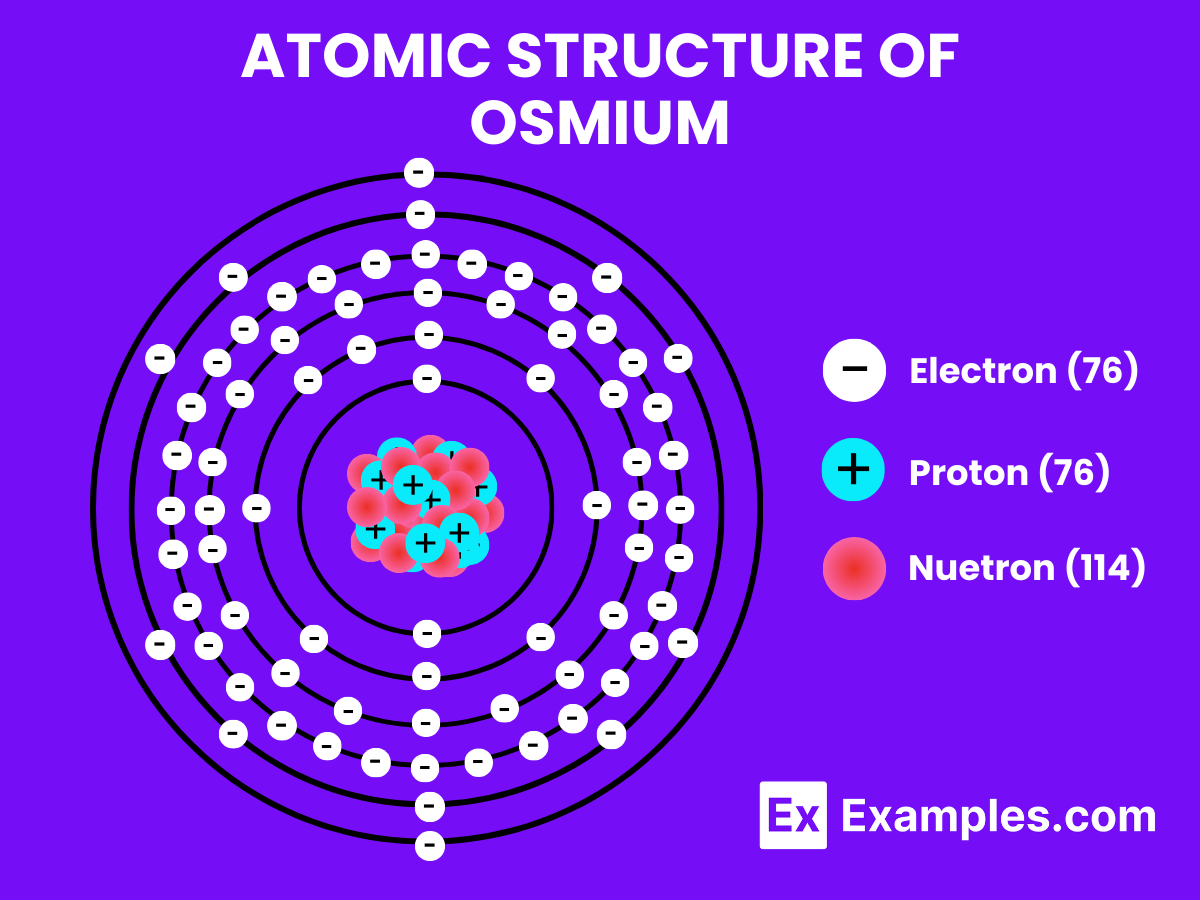
- Atomic Level: Each Osmium atom contains 76 protons, placing it as atomic number 76. The electron configuration of Osmium is [Xe]4f¹⁴5d⁶6s², highlighting a filled 4f orbital and a partially filled 5d orbital, which contributes to its chemical and physical properties.
- Molecular Formation: Osmium does not easily form molecules due to its low reactivity; however, it can engage in complex formation, particularly with oxygen, to create compounds like Osmium tetroxide (OsO₄).
- Stability and Behavior: The compact atomic structure contributes to Osmium’s incredible density and hardness. Under standard conditions, it remains solid, showcasing remarkable stability against wear and corrosion.
This comprehensive overview of Osmium, from its fundamental formula and atomic structure to its practical applications, highlights the element’s significant role across various scientific and industrial domains.
Properties of Osmium
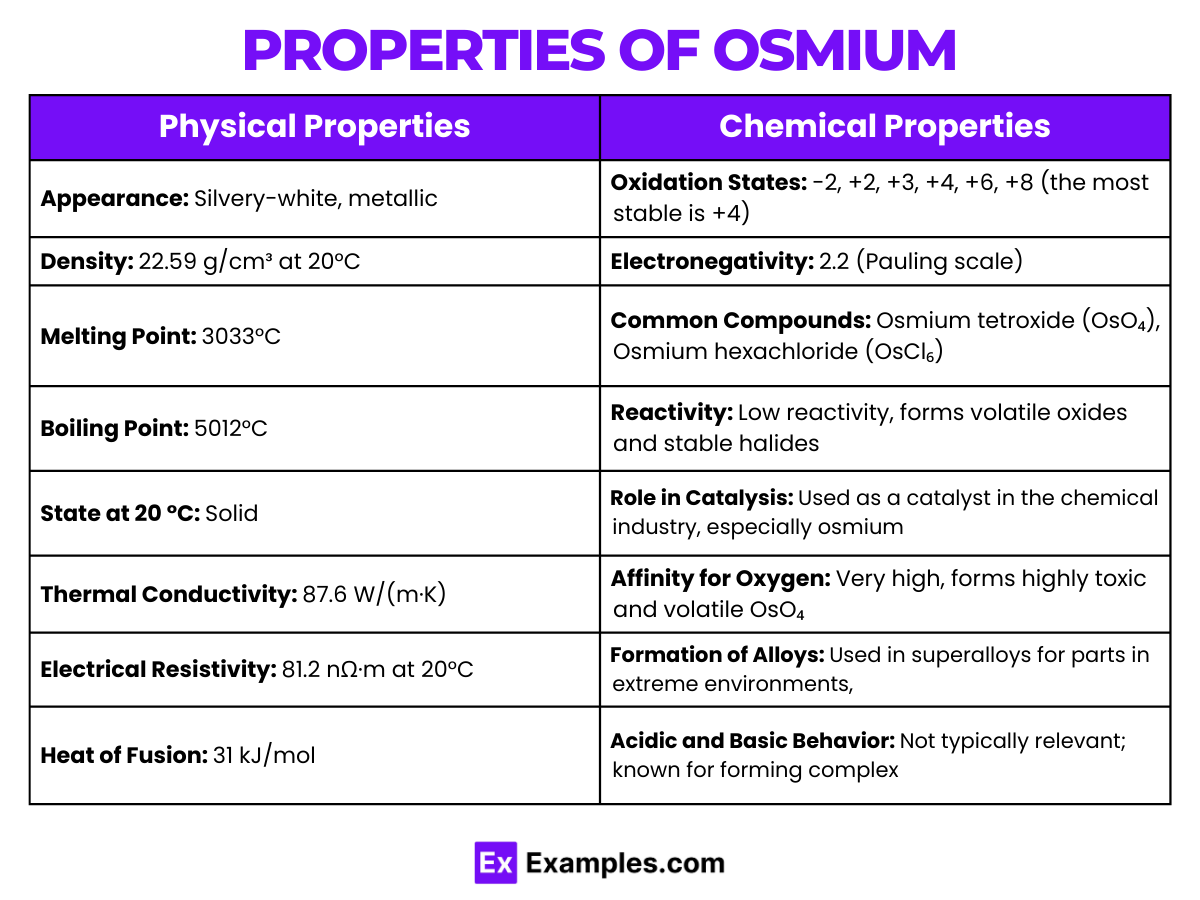
Physical Properties of Osmium
| Physical Property | Description |
|---|---|
| Density | 22.59 g/cm³ at 20 °C, the highest among all elements |
| Melting Point | 3033°C (5491°F) |
| Boiling Point | 5012°C (9054°F) |
| State at 20 °C | Solid |
| Appearance | Bluish-white, lustrous metal |
| Crystal Structure | Hexagonal close-packed (hcp) |
| Thermal Conductivity | 87.6 W/(m·K) (at 300 K) |
| Electrical Conductivity | Lower than most metals but sufficient for some applications |
Chemical Properties of Osmium
Osmium, with the symbol Os and atomic number 76, is a hard, brittle, bluish-white transition metal in the platinum group, known for its high density and remarkable stability. It exhibits a wide range of oxidation states, from -2 to +8, with +4 and +8 being the most common.
- Oxidation States and Compounds:
- Osmium tetroxide (OsO₄) is one of its most well-known compounds, illustrating the +8 oxidation state. This volatile compound is used in staining biological specimens and in organic synthesis.
- In its +4 state, Osmium forms stable compounds like osmium dioxide (OsO₂).
- Reactivity: Despite its general inertness, Osmium reacts with oxygen at room temperature to form osmium tetroxide. It also reacts with halogens to form various halides.
Thermodynamic Properties of Osmium
| Property | Value |
|---|---|
| Melting Point | 3033 °C |
| Boiling Point | 5012 °C |
| Heat of Fusion | 31 kJ/mol |
| Heat of Vaporization | 378 kJ/mol |
| Specific Heat Capacity | 130 J/(kg·K) |
Material Properties of Osmium
| Property | Value |
|---|---|
| Density | 22.59 g/cm³ |
| Mohs Hardness | 7 |
| Vickers Hardness | 3920 MPa |
| Bulk Modulus | 462 GPa |
| Tensile Strength | Not readily available |
Electromagnetic Properties of Osmium
| Property | Value |
|---|---|
| Electrical Resistivity | 81.2 nΩ·m |
| Thermal Conductivity | 87.6 W/(m·K) |
| Magnetic Susceptibility | -0.0002 (dimensionless) |
Nuclear Properties of Osmium
| Property | Value |
|---|---|
| Atomic Number | 76 |
| Atomic Mass | 190.23 u |
| Isotopes | 184, 186, 187, 188, 189, 190, 192 |
| Stable Isotopes | 184, 187, 188, 189, 190, 192 |
Preparation of Osmium
Osmium, with its unique and extreme properties, is both rare and challenging to prepare in pure form. Here are five key points regarding the preparation process of osmium:
- Extraction from Platinum Ores: Osmium is primarily obtained from platinum ores where it exists in small quantities.
- Separation from Other Metals: After extraction, the mixed metals are treated with aqua regia (a mixture of nitric and hydrochloric acid) to dissolve them.
- Formation of Osmium Tetroxide: The solid residue containing osmium is then exposed to chlorine gas in the presence of sodium or potassium chlorate at high temperatures.
- Reduction to Metallic Osmium: The osmium tetroxide is then carefully reduced to metallic osmium. This can be done through several methods, including heating with hydrogen gas or using a more reactive metal like sodium as a reducing agent.
- Purification and Consolidation: The final step involves the purification and consolidation of osmium. Any remaining impurities are removed, and the pure osmium is consolidated into a usable form.
Chemical Properties of Osmium
Osmium Tetroxide
Osmium tetroxide is one of the most significant compounds of osmium,
Equation: Os+4O₂→2OsO₄
Osmium Dioxide
Osmium dioxide is another oxide of osmium, showcasing the metal’s ability to exhibit different oxidation states.
Equation: Os+O₂→OsO₂
Osmium Osmate
Potassium osmate is the product of the aqueous reaction of osmium tetroxide with potassium hydroxide.
Equation: K₂[OsO₂(OH)₄]
Osmium Hexachloride
Osmium hexachloride emphasizes osmium’s ability to form halides, contributing to its comprehensive halogen chemistry.
Equation: Os+3Cl₂→OsCl₆
Osmium Chloride
Osmium chloride is a complex ion demonstrating osmium’s ability to form coordination compounds with ammonia.
Equation:Os+6NH₃+3Cl₂
Osmium Oxide Hydrate
Osmium oxide hydrate is a hydrated form of osmium tetroxide.
Equation: OsO₄+nH₂O
Isotopes of Osmium
| Isotope | Half-life | Natural Abundance |
|---|---|---|
| Os-184 | Stable | 0.02% |
| Os-187 | Stable | 1.6% |
| Os-188 | Stable | 13.3% |
| Os-189 | Stable | 16.1% |
| Os-190 | Stable | 26.4% |
| Os-192 | Stable | 41.0% |
Uses of Osmium
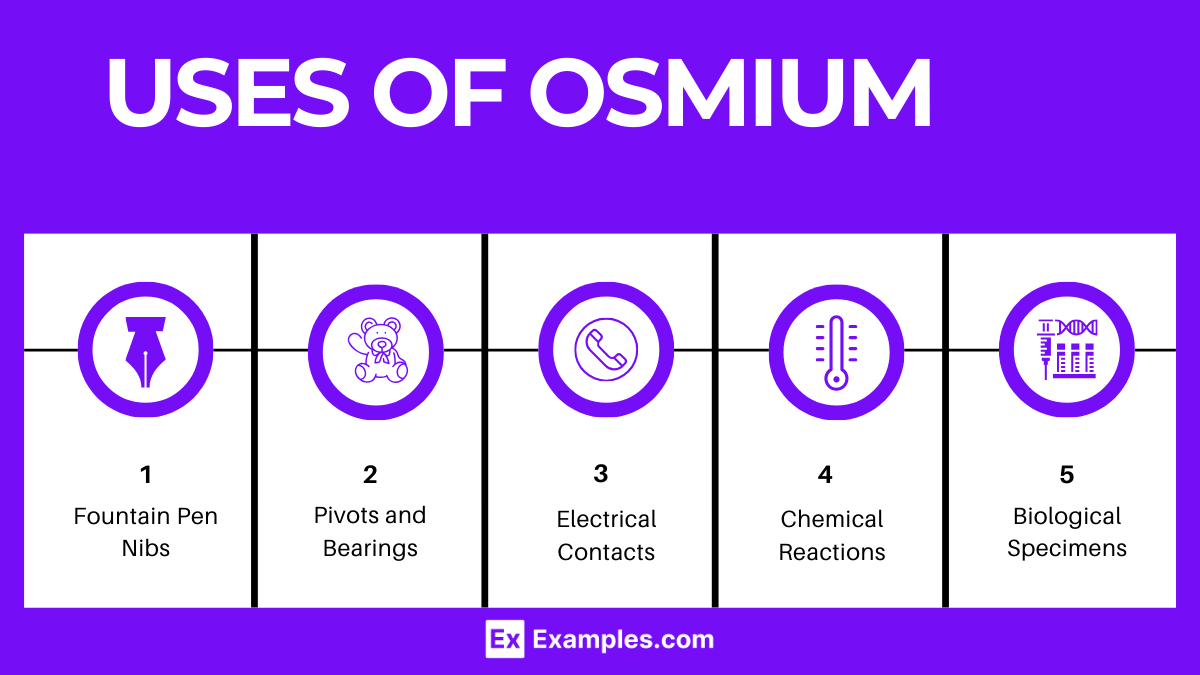
- Electrical Contacts: Osmium is used in electrical contacts for its excellent durability and resistance to wear, ensuring reliable performance over time.
- Fountain Pen Nibs: Alloyed with iridium, osmium provides the hardness and wear resistance needed for high-quality fountain pen nibs, offering a smooth writing experience.
- Instrument Pivots and Bearings: Its resistance to wear makes osmium ideal for the small, precise pivots and bearings found in navigational and measuring instruments, enhancing their longevity and accuracy.
- Catalysis in Chemical Reactions: Osmium compounds, particularly osmium tetroxide (OsO4), serve as catalysts in organic synthesis processes, such as the dihydroxylation of alkenes, adding value in pharmaceutical and chemical research.
- Tetroxide for Staining Biological Specimens: In microscopy, osmium tetroxide is used as a staining agent, providing high contrast for electron microscopy studies of biological specimens.
- Phonograph Styli: The hardness of osmium makes it suitable for use in phonograph styli, where it can endure the wear of playing vinyl records, preserving sound quality.
- Clock and Watchmaking: Osmium’s wear resistance is exploited in high-end clock and watchmaking, especially in components subjected to friction and wear.
- Synthetic Diamonds: Osmium is used as a catalyst in the chemical vapor deposition process for synthetic diamond production, aiding in the creation of high-quality diamonds for industrial and gemological purposes.
- Specialty Alloys: Osmium is alloyed with other metals to produce specialty alloys that benefit from its density, stability, and strength.
Production of Osmium
- Source and Extraction: Osmium is primarily sourced as a byproduct from the processing of platinum and nickel ores.
- Distillation of Osmium Tetroxide: The extracted metals are then subjected to further processing to isolate osmium. This involves converting osmium into its volatile compound, osmium tetroxide (OsO4), through oxidation.
- Reduction to Metal: After distillation, osmium tetroxide is carefully reduced to pure osmium metal. This step requires precise control of temperature.
- Refining and Purification: The metallic osmium produced through reduction is then refined to remove any remaining impurities.
- Safety and Environmental Considerations: The production of osmium must be carefully managed due to the toxic nature of osmium tetroxide, which is highly volatile and dangerous if inhaled.
Applications of Osmium
Osmium’s unique properties, including its extreme density, hardness, and resistance to wear, make it valuable in a variety of specialized applications:
- Electrical Contacts and Filaments: Due to its low wear and stable electrical properties, osmium is used in electrical contacts and filaments where durability is essential.
- Fountain Pen Nibs: Alloyed with other platinum-group metals, osmium provides the necessary hardness and wear resistance for high-quality fountain pen nibs.
- Instrument Pivots and Bearings: The exceptional wear resistance of osmium makes it ideal for use in precision instruments, such as compasses and clocks, where it is used for pivots and bearings.
- Catalysis: Osmium compounds, especially osmium tetroxide, are used as catalysts in organic synthesis, including the dihydroxylation of alkenes.
- Chemical Research: Osmium’s chemical properties make it a subject of research, particularly in high-pressure physics and materials science.
Osmium, the densest naturally occurring element, plays a pivotal role in specialized industrial and scientific applications due to its unique combination of high density, hardness, and chemical stability. Though produced in minimal quantities, its use in electrical contacts, precision instruments, and catalysis underscores its invaluable contribution to advanced technology and scientific research.