Glucose (C₆H₁₂O₆) – Definition, Structure, Preparation, Uses, Benefits
Glucose is a simple sugar that is crucial to the energy processes in living organisms, including humans. It’s a type of carbohydrate found naturally in fruits and other foods, and it plays a vital role in providing cells with energy. Chemically, glucose is a covalent compound, meaning it’s made up of atoms sharing electrons, forming strong bonds that hold the molecule together. This simple sugar is essential for many biological functions and is a fundamental topic in the study of chemistry and biology.
What is Glucose?
Glucose is a type of sugar that is a key source of energy for the cells of our bodies. It comes from the food we eat, especially carbohydrates like bread, fruits, and vegetables. Once inside the body, glucose travels through the bloodstream to our cells, where it is used to produce energy. This energy is vital for our bodies to perform various functions, from running and jumping to thinking and even breathing. Glucose is not just important for humans but for all living organisms. In simple terms, think of glucose as the fuel that powers the engine of life.
Chemical Names and Formulas
| Property | Value |
|---|---|
| Formula | C₆H₁₂O₆ |
| Name | D-(+)-glucose |
| IUPAC Name | 6-(hydroxymethyl)oxane-2, 3, 4, 5-tetrol |
| Alternate Names | dextrose, dextrosol, D-glucose, glucose, grape sugar, sugar, grape |
Structure Of Glucose (C₆H₁₂O₆)
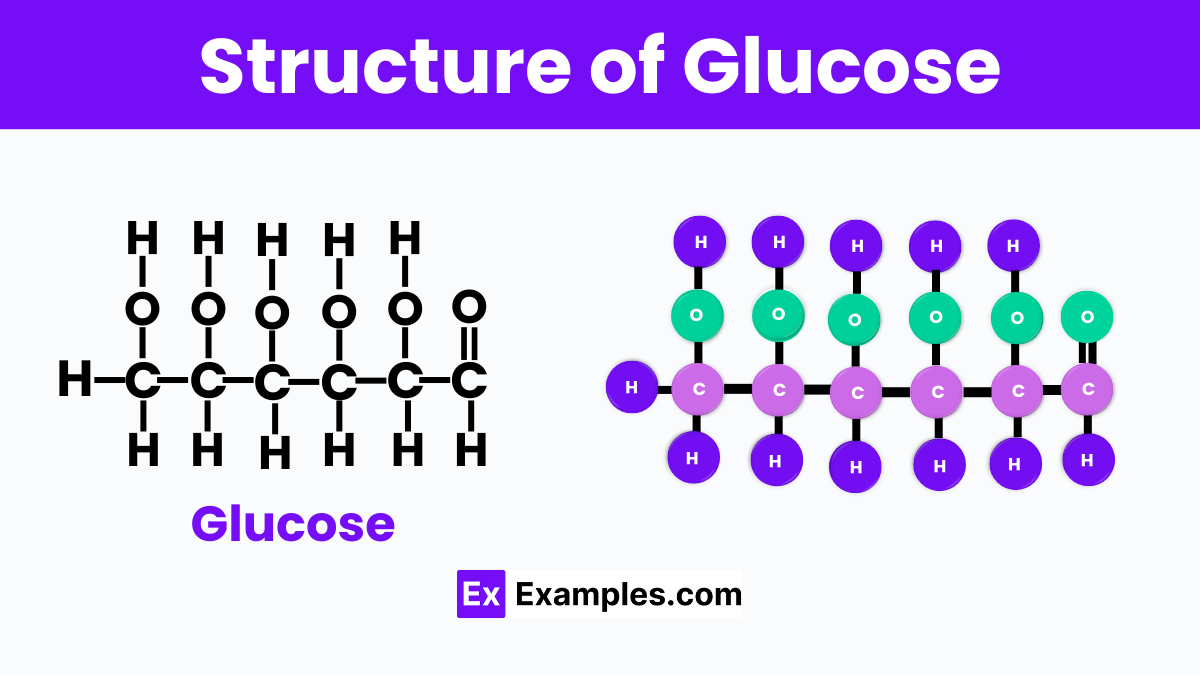
The structure of glucose, which has the chemical formula C₆H₁₂O₆. It is like a tiny building block made up of carbon (C), hydrogen (H), and oxygen (O) atoms. In this structure, six carbon atoms form a chain, with hydrogen and oxygen atoms attached to them in a specific pattern. This arrangement gives glucose its unique properties and enables it to provide energy to living organisms. The atoms in glucose are held together by covalent bonds, which means they share electrons to stay connected. This structure is not just a random assembly; it’s precisely organized to make glucose soluble in water, making it easy for our bodies to transport and use it wherever it’s needed.
Preparation Of Glucose (C₆H₁₂O₆)
Hydrolysis of Starch
The hydrolysis of starch involves breaking down complex carbohydrates into simpler sugars, primarily glucose, by adding water. This reaction can be catalyzed by acids or enzymes. The equation for the acid-catalyzed hydrolysis of starch into glucose is represented as:
In this process, starch (polymer of glucose) is treated with water in the presence of an acid at high temperature. The acid acts as a catalyst to break the glycosidic bonds between the glucose units in starch, resulting in the formation of glucose molecules. This method is widely used in the food industry for the production of glucose syrup from corn or potato starch.
Physical Properties of Glucose (C₆H₁₂O₆)
| Property | Description |
|---|---|
| Molecular Weight | 180.16 g/mol |
| Physical State | Solid at room temperature |
| Color | White |
| Taste | Sweet |
| Solubility | Highly soluble in water, sparingly soluble in ethanol, insoluble in ether |
| Melting Point | 146 °C (α-D-glucose) |
| Boiling Point | Decomposes upon heating before boiling |
| Density | 1.54 g/cm³ (solid) |
| Optical Rotation | +52.7° (D-glucose, in water) |
| Specific Rotation | α_D^20 = +52.5 to +53.0° (in water) |
Chemical Properties of Glucose (C₆H₁₂O₆)
Formation of Glucosides
Glucose reacts with alcohols in the presence of an acid catalyst to form glucosides, a type of glycosidic bond.
Equation: C₆H₁₂O₆ + ROH → C₆H₁₁OR + H₂O (R = alkyl group)
Oxidation
Glucose can be oxidized to gluconic acid by mild oxidizing agents like bromine water.
Equation: C₆H₁₂O₆ + Br₂ + H₂O → C₆H₁₂O₇ + 2HBr
Reduction
It can be reduced to sorbitol (a sugar alcohol) in the presence of hydrogen and a catalyst.
Equation: C₆H₁₂O₆ + H₂ → C₆H₁₄O₆
Fermentation
- Under anaerobic conditions, glucose is fermented by yeast to produce ethanol and carbon dioxide.
- Equation: C₆H₁₂O₆ → 2C₂H₅OH + 2CO₂
Formation of Osazone
- When glucose is treated with phenylhydrazine, it forms a yellow crystalline compound called glucosazone.
- Equation: 3C₆H₁₂O₆ + 3PhNHNH₂ → (C₆H₁₀O₄)(NHNHPh)₂ + 3H₂O + 3PhNH₂ (Ph = Phenyl)
Isomerization
- In the presence of an alkali, glucose can isomerize into fructose, a process used in the production of high-fructose corn syrup.
- Equation: C₆H₁₂O₆ (glucose) ⇌ C₆H₁₂O₆ (fructose)
Tollens’ Test
- Glucose reduces Tollens’ reagent (ammoniacal silver nitrate solution) to metallic silver.
- No specific equation but indicative of reducing sugars.
Fehling’s Test
- Glucose also reduces Fehling’s solution to a red precipitate of copper(I) oxide.
- Again, no specific equation but demonstrates the reducing nature of glucose.
Glucose (C₆H₁₂O₆) Chemical Compound Information
Chemical Identifiers
| Property | Value |
|---|---|
| CAS Registry Number | 50-99-7 |
| Beilstein Number | 1724615 |
| PubChem Compound ID | 206 |
| PubChem Substance ID | 3521 |
| SMILES Identifier | C(C1C(C(C(C(O1)O)O)O)O)O |
| InChI Identifier | InChI=1/C6H12O6/c7-1-2-3(8)4(9)5(10)6(11)12-2/h2-11H, 1H2 |
| InChI Key | WQZGKKKJIJFFOK-UHFFFAOYAC |
| EU Number | 200-075-1 |
| RTECS Number | LZ6600000 |
| NSC Number | 406891 |
NFPA Label
| Property | Value |
|---|---|
| NFPA Health Rating | 0 |
| NFPA Fire Rating | 0 |
| NFPA Reactivity Rating | 0 |
Uses of Glucose (C₆H₁₂O₆)
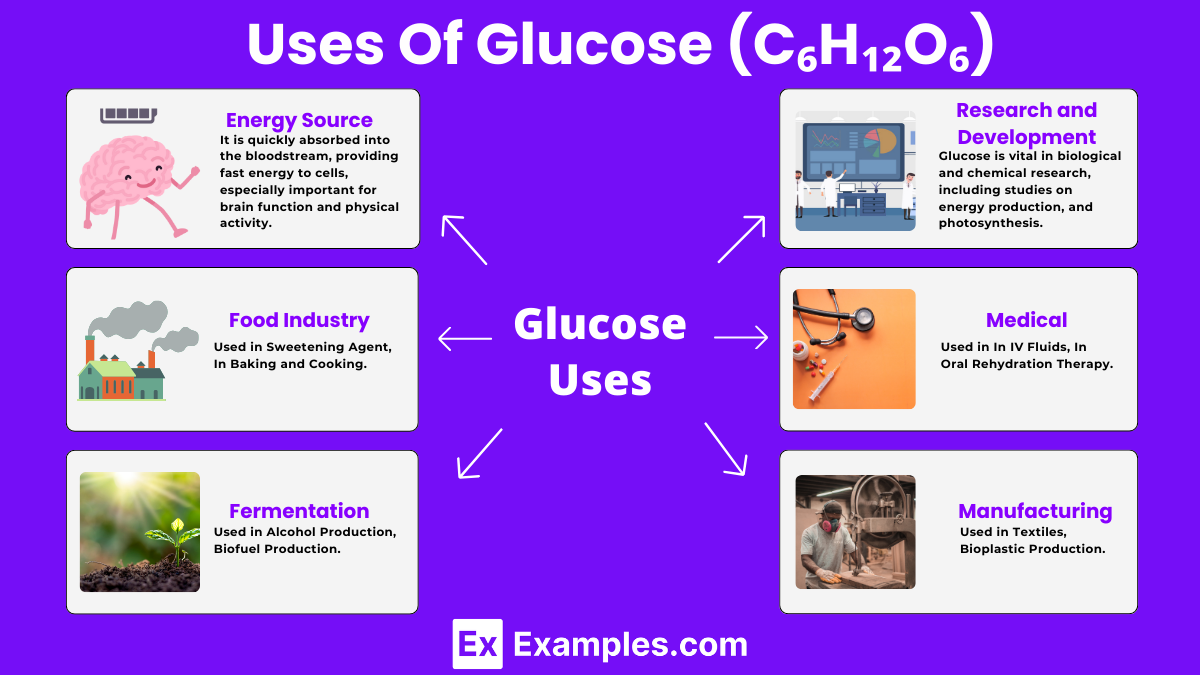
Energy Source
- In Humans and Animals: Glucose is the main energy source for the body’s cells. It is quickly absorbed into the bloodstream, providing fast energy to cells, especially important for brain function and physical activity.
Medical Use
- In IV Fluids: Glucose solutions are often used in intravenous (IV) fluids to provide nutrition and energy directly to patients who cannot eat or drink.
- In Oral Rehydration Therapy: It helps in treating dehydration, especially in cases of severe diarrhea, by aiding the absorption of electrolytes in the intestines.
Food Industry
- Sweetening Agent: Glucose syrup, derived from starch, is widely used in food products as a sweetener. It’s found in candies, soft drinks, and desserts.
- In Baking and Cooking: Glucose can control the crystallization of sugar, improving the texture and shelf life of baked goods and confectionery.
Fermentation
- Alcohol Production: Glucose serves as the primary feedstock for yeast in the fermentation process, which produces alcohol. This process is fundamental in making beer, wine, and spirits.
- Biofuel Production: In the production of bioethanol, glucose is fermented by microorganisms. Bioethanol is a renewable energy source that can be used as a fuel for vehicles.
Manufacturing
- In Textiles: Glucose is used in the textile industry for sizing and finishing fabrics, improving their strength and smoothness.
- Bioplastic Production: It’s a key ingredient in producing biodegradable plastics, offering an eco-friendly alternative to traditional petroleum-based plastics.
Research and Development
- In Laboratory Research: Glucose is vital in biological and chemical research, including studies on metabolism, energy production, and photosynthesis.
Functions Of Glucose
Blood Glucose Regulation
- Insulin and Glucagon: These hormones manage blood glucose levels, ensuring energy stability.
Energy Storage and Release
- Glycogenesis: Excess glucose is stored as glycogen in the liver and muscles.
- Glycogenolysis: Stored glycogen is converted back to glucose to meet energy demands.
Biological Precursor
- Synthesis: Glucose is a starting point for making other essential molecules, including some amino acids and fatty acids.
Health Implications
- Diabetes: Imbalances in glucose regulation can lead to diabetes, affecting various organs.
- Metabolic Disorders: Conditions affecting glucose metabolism highlight its importance in energy management.
Benefits of Glucose
Immediate Energy Source
Glucose is the primary fuel for the body’s cells, offering quick energy, particularly crucial for brain function and muscle activity.
Brain Function Support
It is essential for brain health, aiding cognitive functions like thinking, memory, and learning due to its role as the brain’s main energy source.
Blood Sugar Regulation
Glucose helps in regulating blood sugar levels, with the body’s insulin response facilitating its uptake by the cells, thus providing a steady energy supply.
Athletic Performance Enhancement
For athletes and those engaged in high-intensity workouts, glucose is vital for improving performance and endurance by fueling muscle contractions.
Metabolic Health
Glucose metabolism plays a critical role in maintaining metabolic balance, supporting weight management, and reducing the risk of metabolic syndrome.
Recovery Aid
It is key in post-exercise recovery, helping to replenish muscle and liver glycogen stores, which speeds up recovery and reduces fatigue.
Immune Function Support
Glucose provides energy to white blood cells, aiding in the rapid response to infections and promoting healing, thus supporting the immune system.
FAQ’S
What Does It Mean When Your Glucose Is High?
High glucose, or hyperglycemia, indicates excess sugar in the blood, often signaling diabetes or prediabetes, requiring medical attention.
What Is Normal Glucose Sugar Level?
A normal glucose level ranges between 70-99 mg/dL when fasting and less than 140 mg/dL two hours after eating.
How Can I Lower My Glucose Level?
Lower glucose by exercising regularly, eating a balanced diet, managing stress, staying hydrated, and following medical advice.
What Is Glucose vs Sugar?
Glucose is a simple sugar, a building block of carbohydrates. “Sugar” generally refers to table sugar, a disaccharide called sucrose.


