Radioactive Decay Law – Examples, Derivation, Uses, FAQ’s
The Radioactive Decay Law describes the process by which an unstable atomic nucleus loses energy by emitting radiation. This law is a fundamental concept in nuclear physics and helps explain the behavior of radioactive substances.
What is Radioactive Decay Law?
The Radioactive Decay Law states that the rate at which a radioactive substance decays is directly proportional to the amount of the substance present. This relationship can be mathematically expressed using an exponential function. The Radioactive Decay Law provides a quantitative framework for understanding the behavior of radioactive materials over time.
Radioactive Decay
Radioactive decay is a natural process that plays a critical role in various scientific and practical fields. Understanding the different types of decay and their characteristics allows scientists and engineers to harness this phenomenon for a wide range of applications, from dating ancient objects to treating diseases and generating energy. This process results in the transformation of the original, unstable nucleus into a more stable one, often of a different element. The study of radioactive decay is crucial in fields such as nuclear physics, radiometric dating, medicine, and energy production. Radioactive isotopes are used in diagnostic imaging and cancer treatment. Understanding their decay helps in determining safe dosages and timing for medical procedures.
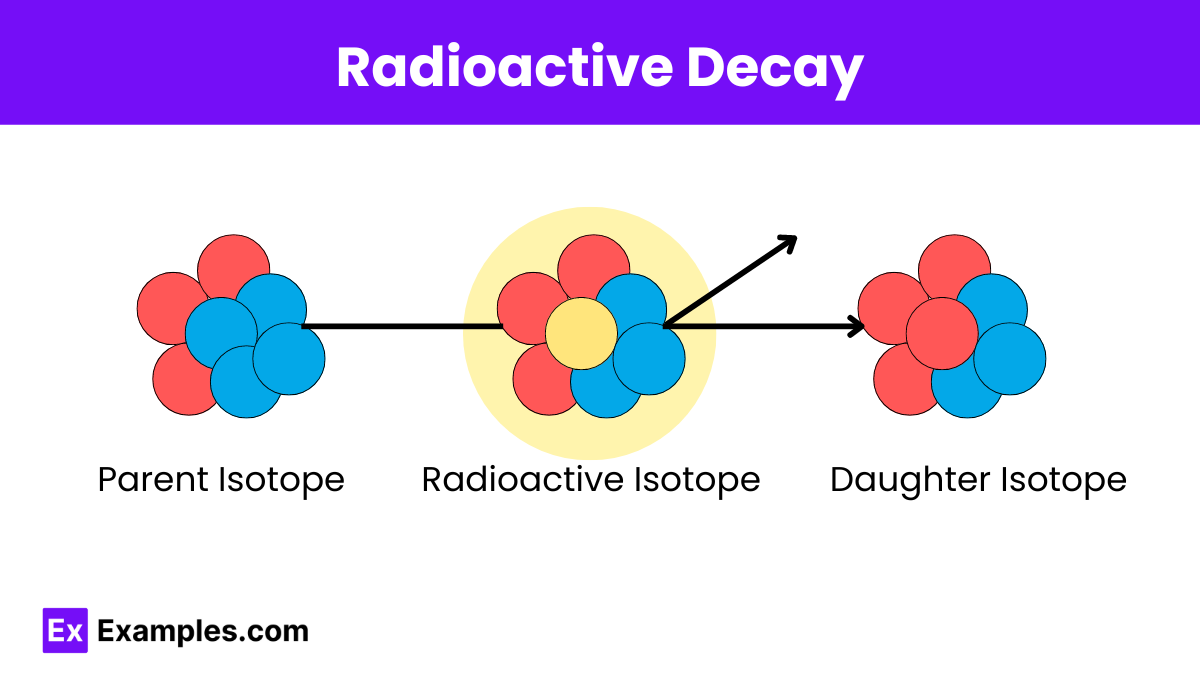
Types of Radioactive Decay
Radioactive decay occurs in various forms, each involving different particles and energy emissions. The primary types of radioactive decay are:
- Alpha Decay
- Beta Decay
- Gamma Decay
1. Alpha Decay
In alpha decay, the nucleus emits an alpha particle. An alpha particle is composed of two protons and two neutrons, resembling a helium-4 nucleus. This process decreases the atomic number by 2 and the mass number by 4.
Formula
The energy released during alpha decay can be expressed as:
where:
- 𝑚ᵢ is the initial mass of the nucleus
- 𝑚բ is the mass of the nucleus after emission
- 𝑚ₚ is the mass of the alpha particle
For example, uranium-238 undergoes alpha decay to form thorium-234:

2. Beta Decay
Beta decay involves the transformation of a neutron into a proton or vice versa, accompanied by the emission of a beta particle, which can be an electron (𝛽−) or a positron (𝛽+).
Types of Beta Decay
Beta Minus Decay (𝛽−): A neutron is converted into a proton, releasing an electron and an antineutrino. Example:

Beta Plus Decay (𝛽+): A proton is converted into a neutron, emitting a positron and a neutrino. Example:
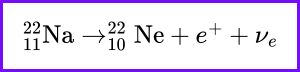
3. Gamma Decay
Gamma decay occurs when an excited nucleus releases excess energy in the form of gamma rays, which are high-energy photons. This decay does not change the number of protons or neutrons in the nucleus but lowers its energy state.
Formula of Radioactive Decay Law
The law can be expressed using the following formula:
where:
- 𝑁(𝑡) is the number of undecayed nuclei at time t,
- 𝑁₀ is the initial number of undecayed nuclei,
- 𝜆 is the decay constant (a measure of the probability of decay per unit time),
- 𝑡 is the time elapsed.
Derivation of Radioactive Decay Law
According to the radioactive decay law, when a radioactive material undergoes either 𝛼 or 𝛽 or 𝛾 decay, the rate at which the nuclei decay is directly proportional to the number of nuclei present in the sample at that time.
The radioactive decay law states that “The probability per unit time that a nucleus will decay is a constant, independent of time.”
This constant is represented by 𝜆 (lambda) and is known as the decay constant.
The mathematical expression of the law of radioactive decay is:
Δ𝑁/Δ𝑡 ∝ 𝑁
where:
- 𝑁 is the total number of nuclei in the sample,
- Δ𝑁 is the number of nuclei that decays,
- Δ𝑡 is the unit time.
This proportional relationship can be written as:
Δ𝑁/Δ𝑡=−𝜆𝑁 (1)
where:
- 𝜆 is the decay constant, also known as the disintegration constant.
The negative sign indicates that the number of nuclei decreases over time.
Differential Form
The change in the number of nuclei with respect to time can be described using a differential equation:
𝑑𝑁/𝑑𝑡 =−𝜆𝑁
Separating the variables and integrating both sides, we get:
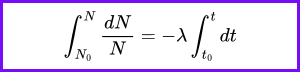
Solving the integrals, we obtain:
ln𝑁 − ln𝑁₀=−𝜆(𝑡−𝑡₀) (3)
Rearranging the equation gives us:
ln(𝑁/𝑁₀)=−𝜆𝑡
Exponentiating both sides, we get the solution to the differential equation:
𝑁(𝑡)=𝑁₀𝑒^−𝜆𝑡 (4)
This final equation (4) represents the law of radioactive decay, showing that the number of un-decayed nuclei decreases exponentially over time.
Uses of Radioactive Decay Law

- Radiometric Dating: You use the Radioactive Decay Law to determine the age of archaeological and geological samples. By measuring the remaining concentration of a radioactive isotope and knowing its half-life, you can calculate the time that has elapsed since the sample formed. This method is crucial in fields like archaeology (carbon dating) and geology (uranium-lead dating).
- Medical Applications: In medicine, you use radioactive isotopes for diagnostic and therapeutic purposes. The decay law helps in planning treatments and diagnostics, such as in cancer radiotherapy, where precise doses of radiation target tumor cells. Radioisotopes like iodine-131 and technetium-99m rely on predictable decay rates for effective use.
- Nuclear Power Generation: The principles of radioactive decay are fundamental in nuclear power plants. By understanding decay chains and the half-lives of various isotopes, engineers can manage fuel usage and waste disposal effectively. This ensures the safe and efficient production of nuclear energy.
- Environmental Monitoring: You use the Radioactive Decay Law to track and measure pollution from radioactive substances. Monitoring the decay of isotopes in the environment helps assess contamination levels and plan remediation efforts, ensuring public health and safety.
- Food Irradiation: The food industry uses radioactive decay to preserve food. By exposing food to radiation, you can eliminate bacteria and pests, prolonging shelf life without significantly affecting nutritional quality. Understanding the decay rates of the isotopes used ensures safe application.
- Scientific Research: In physics and chemistry, researchers use radioactive isotopes to study reaction mechanisms and material properties. The decay law provides a reliable way to measure isotopic concentrations and changes over time, facilitating experiments and discoveries.
- Space Exploration: Radioactive isotopes power some space missions. For example, radioisotope thermoelectric generators (RTGs) use the heat from radioactive decay to generate electricity, powering spacecraft on long-duration missions.
Examples for Radioactive Decay Law
- Carbon-14 Dating: Archaeologists use the radioactive decay of carbon-14 to date ancient organic materials. By measuring the remaining carbon-14 in a sample and knowing its half-life (about 5730 years), they can determine the age of artifacts, fossils, and other historical items. For instance, if an artifact has half the carbon-14 compared to a living sample, it is approximately 5730 years old.
- Cancer Treatment with Radioisotopes: In radiation therapy, doctors use the decay of isotopes like iodine-131 to treat cancer. The predictable decay helps in planning doses to maximize the destruction of cancer cells while minimizing harm to healthy tissue. For example, iodine-131 has a half-life of about 8 days, so doctors can calculate the appropriate dosage and timing for treatments.
- Smoke Detectors: Many smoke detectors use americium-241, a radioactive isotope. The decay of americium-241 emits alpha particles, which ionize the air in the detector. When smoke disrupts this ionization, the alarm sounds. Understanding the decay rate ensures the detector functions correctly over its lifespan.
- Uranium-Lead Dating: Geologists use uranium-238 decay to lead-206 to date rocks and minerals. The half-life of uranium-238 is about 4.5 billion years, making it suitable for dating the age of the Earth. By measuring the ratio of uranium to lead in a sample, geologists can estimate its age. For example, if a rock has an equal amount of uranium-238 and lead-206, it is approximately 4.5 billion years old.
- Medical Imaging: Technetium-99m is used in various diagnostic imaging procedures. It has a half-life of about 6 hours, which is long enough to conduct medical tests but short enough to minimize radiation exposure. For example, in a cardiac stress test, technetium-99m helps create images of blood flow in the heart.
- Environmental Monitoring: Scientists monitor the decay of cesium-137 to track nuclear contamination. Cesium-137 has a half-life of about 30 years, making it useful for studying long-term environmental impacts of nuclear accidents, such as the Chernobyl disaster. For instance, measuring cesium-137 levels in soil samples helps assess contamination spread over time.
- Powering Spacecraft: Radioisotope thermoelectric generators (RTGs) use the decay of plutonium-238 to generate electricity for spacecraft. Plutonium-238 has a half-life of about 88 years, providing a steady power source for long-duration missions. For example, the Voyager probes, launched in 1977, still communicate with Earth thanks to RTGs.
- Agricultural Pest Control: Radioactive isotopes like cobalt-60 are used to sterilize pests. The decay of cobalt-60 emits gamma rays, which are effective in sterilization. This method helps control pest populations without the need for chemical pesticides. For instance, sterilizing male insects with gamma rays and releasing them into the wild reduces pest numbers over time.
- Tracing Biological Processes: Scientists use carbon-14 to trace biochemical processes in plants and animals. By incorporating carbon-14 into molecules and tracking its decay, researchers can study metabolic pathways and rates of synthesis and degradation of biomolecules.
FAQ’s
What is radioactive decay?
Radioactive decay is the process by which an unstable atomic nucleus loses energy by emitting radiation, transforming into a different element or isotope.
Can radioactive decay be predicted accurately?
Individual decay events are random, but the decay rate of a large sample follows predictable statistical patterns.
What units are used for measuring radioactivity?
Radioactivity is measured in becquerels (Bq) or curies (Ci), indicating the number of decays per second.
How is the decay constant related to half-life?
The decay constant and half-life are inversely related; a higher decay constant means a shorter half-life.
What is a decay constant?
The decay constant is a probability factor that represents the likelihood of a single nucleus decaying per unit time.
How does radioactive decay affect isotopes?
Radioactive decay transforms an unstable isotope into a more stable one, often of a different element.
What types of radioactive decay exist?
The main types of radioactive decay are alpha decay, beta decay, and gamma decay.
How is radioactive decay measured?
Radioactive decay is measured using a Geiger counter, scintillation counter, or other radiation detectors that count emitted particles.
What is a half-life?
A half-life is the time required for half of the radioactive nuclei in a sample to decay.
Who discovered the radioactive decay law?
Ernest Rutherford and Frederick Soddy discovered the radioactive decay law in the early 20th century.



