Wiedemann Franz Law – Examples, Definition, Limitations, FAQ’S
Wiedemann-Franz Law is a principle in physics that establishes a relationship between the thermal conductivity and electrical conductivity of metals. It states that the ratio of thermal conductivity (𝑘) to electrical conductivity (𝜎) is directly proportional to the absolute temperature (𝑇) of the metal.
What is Wiedemann Franz Law?
Wiedemann Franz Law Formula
The formula for the Wiedemann-Franz Law is:
where:
- 𝑘 is the thermal conductivity (W/m·K),
- 𝜎 is the electrical conductivity (S/m),
- 𝐿 is the Lorenz number, approximately 2.44×10⁻⁸ WΩK⁻²,
- 𝑇 is the absolute temperature (in kelvins).
This formula demonstrates that the ratio of thermal conductivity to electrical conductivity is proportional to the temperature of the metal, with the constant 𝐿L remaining consistent for most metals.
Wiedemann Franz Law Derivation
The Wiedemann-Franz Law connects the thermal and electrical conductivity of metals. Here’s an outline of its derivation:
Key Principles
- Free Electron Theory: The free electrons in a metal are responsible for both thermal and electrical conduction.
- Thermal Conductivity (𝑘): Measures the heat transport due to electron motion.
- Electrical Conductivity (𝜎): Measures the charge transport via electron flow.
Step-by-Step Derivation
Thermal Conductivity (𝑘):
- The thermal conductivity depends on the specific heat capacity of electrons, their speed, and the mean free path (l).
- The expression for thermal conductivity is:
- 𝑘=1/3cᵥvբ𝑙
- where:
- cᵥ is the electronic specific heat capacity,
- vբ is the Fermi velocity (electrons near the Fermi energy),
- 𝑙 is the mean free path of electrons.
Electrical Conductivity (𝜎):
- Electrical conductivity can be related to the density and charge of electrons, their velocity, and mean free path:
- where:
- 𝑛 is the density of conduction electron,
- 𝑒 is the charge of an electron,
- 𝜏 is the relaxation time (related to the mean free path),
- 𝑚 is the electron mass.
Linking Thermal and Electrical Conductivity:
- By combining the above equations and applying the free electron theory, one can relate the ratio of thermal conductivity to electrical conductivity.
- The result is the Wiedemann-Franz Law:
- where:
- L is the Lorenz number,
- which is a constant:𝐿≈2.44×10⁻⁸ WΩK⁻²
- 𝑇 is the absolute temperature in kelvins.
Wiedemann Franz Law Limitations

The Wiedemann-Franz Law links thermal and electrical conductivities in metals but has notable limitations.
- High Temperature Effects: At high temperatures, increased electron scattering from phonons leads to deviations in thermal and electrical conductivities, affecting the accuracy of the Lorenz number.
- Electron-Electron Interactions: In dense metals and high temperatures, electron-electron interactions cause deviations from the expected relationship.
- Low Temperature Phenomena: At extremely low temperatures, quantum effects like electron localize alter conductivities, leading to deviations from predictions.
- Non-Metallic Conductors: This law doesn’t apply well to non-metallic materials like semiconductors, which have different charge carriers and conduction mechanisms.
- Impurities and Defects: Impurities and structural defects can disrupt electron flow, causing a difference from the expected ratio.
Uses of Wiedemann Franz Law
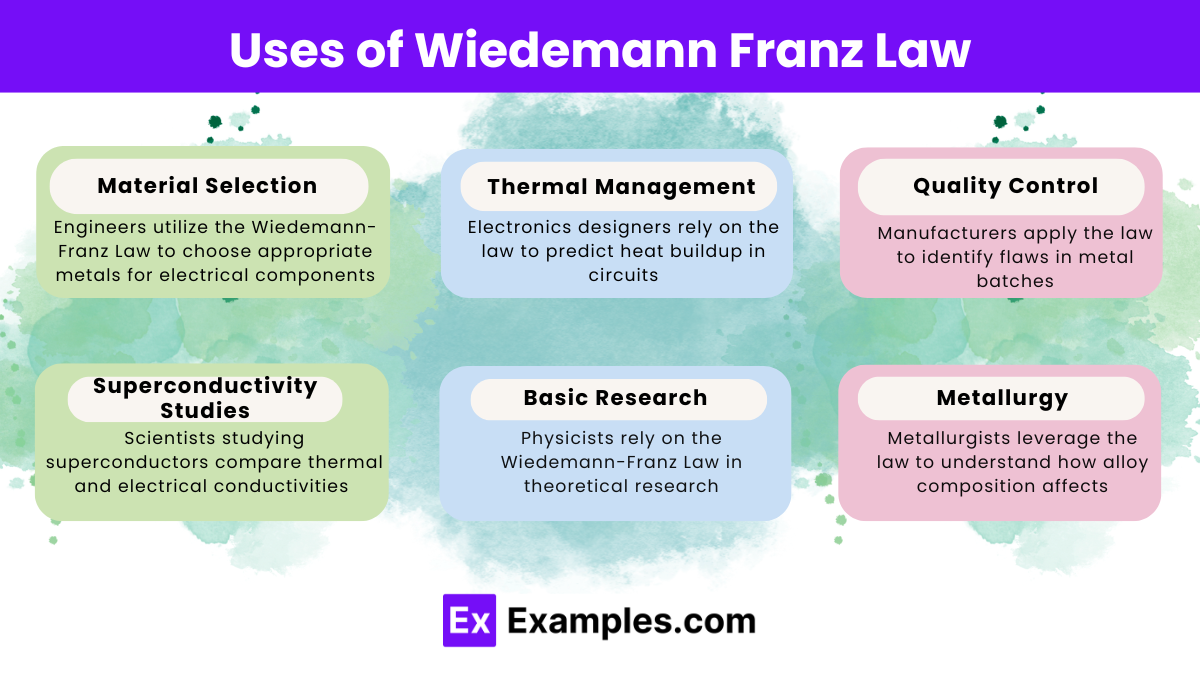
- Material Selection: Engineers utilize the Wiedemann-Franz Law to choose appropriate metals for electrical components. Therefore, they can ensure efficient heat dissipation by picking materials that balance thermal and electrical conductivity.
- Thermal Management: Electronics designers rely on the law to predict heat buildup in circuits. Consequently, they can improve cooling systems by understanding the relationship between electrical conductivity and heat generation.
- Quality Control: Manufacturers apply the law to identify flaws in metal batches. Hence, inconsistencies between thermal and electrical conductivities can reveal impurities or defects.
- Superconductivity Studies: Scientists studying superconductors compare thermal and electrical conductivities at various temperatures. As a result, deviations from the law help identify the onset of superconducting states.
- Basic Research: Physicists rely on the Wiedemann-Franz Law in theoretical research. Consequently, they test free-electron theory models and investigate electron behavior in different materials.
- Metallurgy: Metallurgists leverage the law to understand how alloy composition affects both thermal and electrical properties.
Examples for Wiedemann Franz Law
- Copper: Copper is great at conducto electricity and heat. The Wiedemann-Franz Law shows that both conductivities are linked by temperature.
- Aluminum: Aluminum, a popular choice for electrical wiring, also conducts heat well, following the relationship between heat and electrical flow described by the law.
- Silver: Silver, the best electrical conductor among metals, also moves heat efficiently, align with the principles of the Wiedemann-Franz Law.
- Steel: Steel, although not as conductive as other metals, shows a connection between its heat and electricity conductivity, which varies with temperature.
- Semiconductors: While the law mainly applies to metals, deviate in semiconductors help scientists understand how charge carriers differ from metals.
FAQ’S
What is the theory of Wiedemann-Franz law?
The Wiedemann-Franz Law states that metals have a consistent ratio between thermal and electrical conductivities at a given temperature, reflect the role of free electrons in both processes.
What is the Lorenz number using the Wiedemann-Franz law?
Using the Wiedemann-Franz Law, the Lorenz number is 𝐿≈2.44×10⁻⁸ WΩK⁻², represents the proportionality between heat and electrical conduction in metals.
Which one is correct for Wiedemann-Franz law?
For the Wiedemann-Franz Law to hold, metals should have a consistent relationship between thermal and electrical conductivities, with any deviations linked to impurity or extreme temperatures.