Dubnium (Db) – Definition, Preparation, Properties, Uses, Compounds, Reactivity
Dive into the world of Dubnium, a lesser-known marvel of the periodic table, through our detailed guide. Dubnium, with its unique properties and synthetic origins, stands as a testament to human ingenuity in the field of chemistry. This guide unfolds the definition, uses, and intriguing aspects of Dubnium, offering insightful examples to illuminate its role in scientific advancements. Join us on a journey to understand Dubnium’s place in modern science, exploring its characteristics, applications, and the compounds it forms.
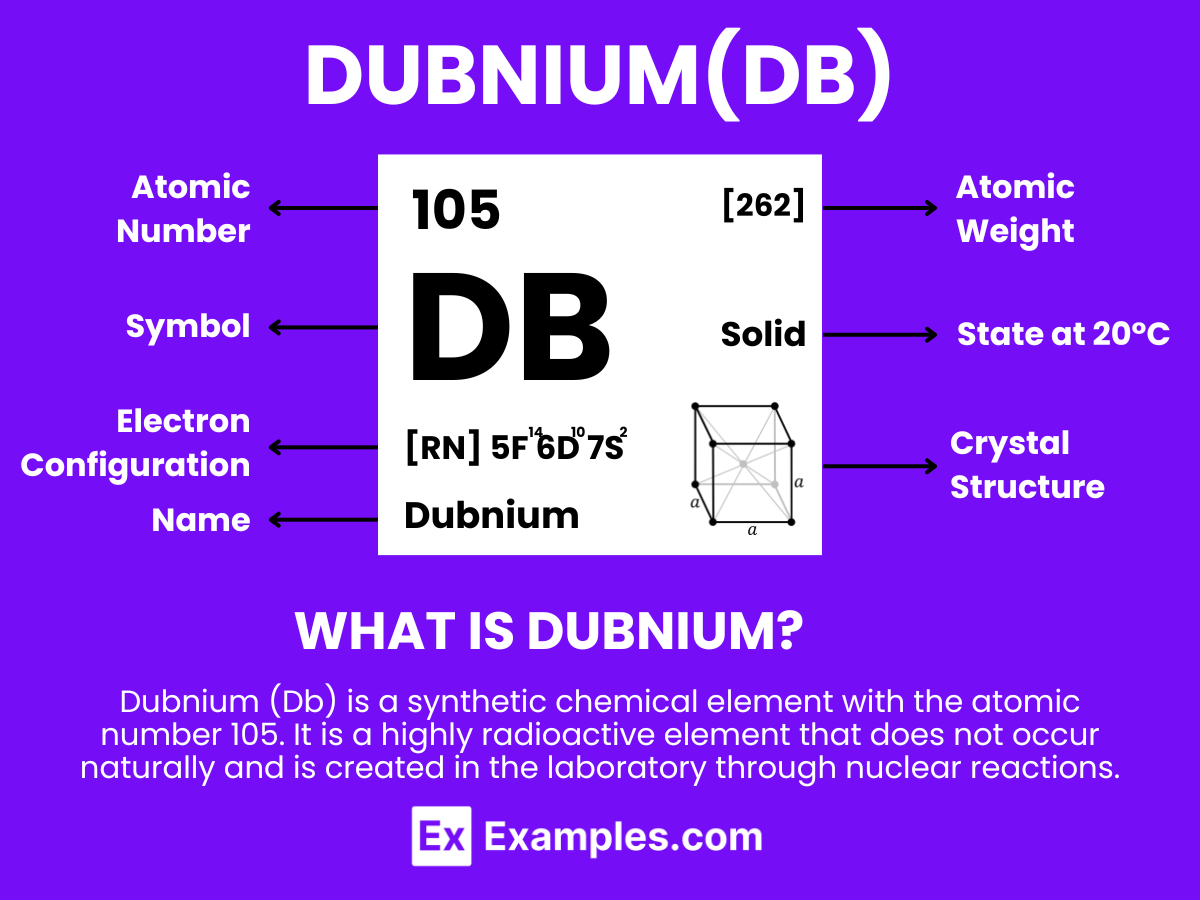
What is Dubnium?
Dubnium (Db) is a synthetic chemical element with the atomic number 105. It is a highly radioactive element that does not occur naturally and is created in the laboratory through nuclear reactions. Named after the Russian town of Dubna, where it was first discovered, dubnium is part of the group of elements known as the transactinides. Because of its instability and short half-life, detailed studies on dubnium are limited, and it has no known practical applications outside of scientific research. The properties of dubnium, including its chemical behavior, are mostly inferred from its position in the periodic table, as it is expected to resemble the elements in group 5, particularly tantalum and niobium, in its chemical properties.
Dubnium Formula
- Formula: Db
- Composition: Composed of a single dubnium atom.
- Bond Type: In its elemental state, dubnium does not form bonds as it exists as a pure element. Nonetheless, it is capable of forming covalent or ionic bonds when interacting with other elements, although its chemical behavior is not well-documented due to its rare and ephemeral nature.
- Molecular Structure: As a solitary element, dubnium does not create conventional molecular structures. It is presumed to exhibit characteristics of a heavy, volatile metal with a possible close-packed crystalline structure, but these properties are largely hypothetical.
- Electron Sharing: Dubnium is expected to engage in electron sharing through covalent bonding or electron transfer in ionic bonds with other elements. These anticipations are based on its group positioning within the periodic table, although empirical evidence is scarce.
- Significance: Dubnium stands out as a superheavy element synthesized in particle accelerators, offering insights into the behavior of elements at the far reaches of the periodic table and the stability of heavy atomic nuclei.
- Role in Chemistry: The role of dubnium is predominantly in the realm of scientific research, especially in studies aiming to push the limits of the periodic table and the creation of novel elements. Any practical applications of dubnium beyond research purposes remain speculative due to its very limited availability and short half-life.
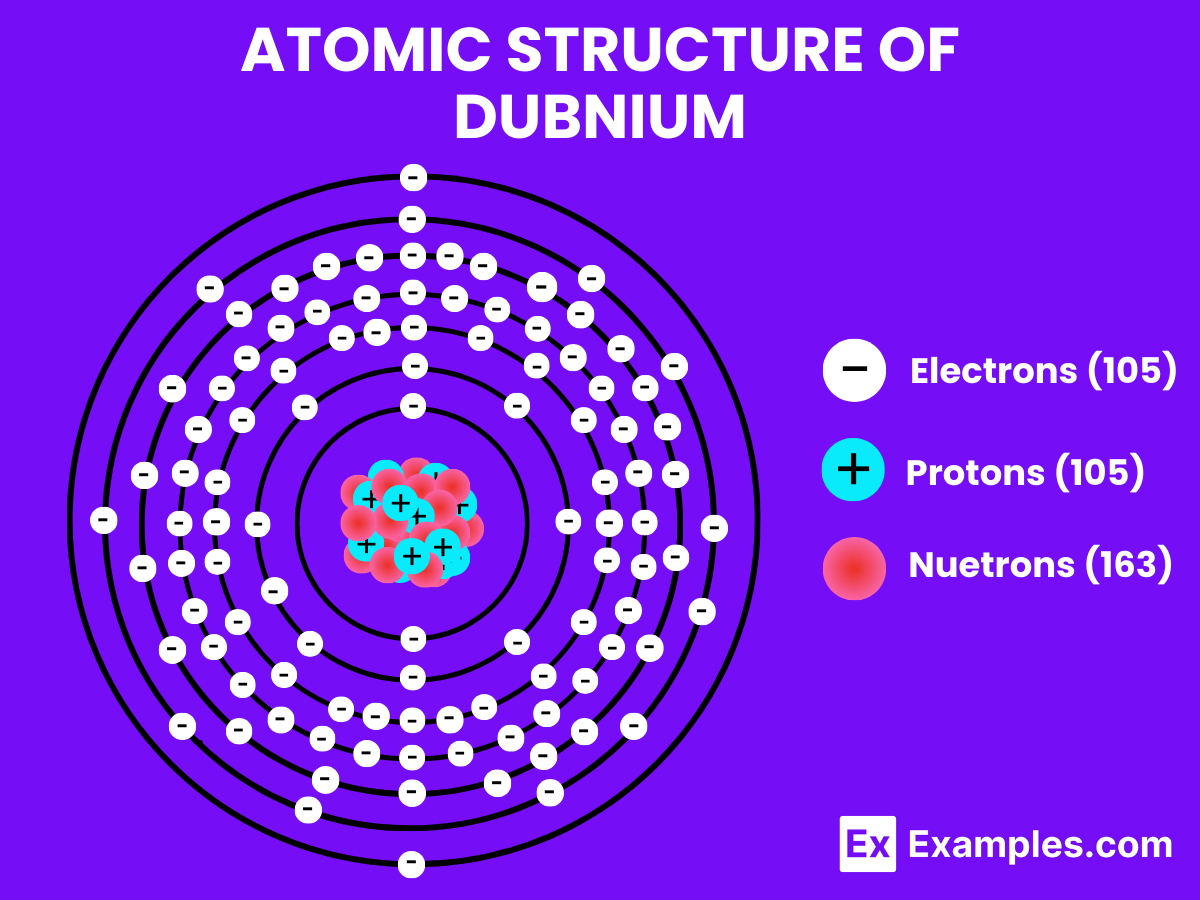
Atomic Structure of Dubnium
Dubnium (Db) is a synthetic, highly radioactive element with the atomic number 105. As an element, its formula is represented simply by its symbol, “Db,” and it consists of a single dubnium atom. In its elemental state, dubnium does not form bonds as it exists as a pure element, but it has the potential to form both covalent and ionic bonds when interacting with other elements. The chemical behavior of dubnium is not thoroughly studied due to its scarcity and short-lived nature.
Regarding its molecular structure, dubnium does not form conventional molecules as an isolated element. It is anticipated to exhibit the traits of a heavy, possibly volatile metal, with a crystalline structure that might resemble those of its group 5 counterparts, such as vanadium (V), niobium (Nb), and tantalum (Ta). However, any definitive description of its properties remains speculative.
Dubnium is expected to participate in electron sharing through covalent bonding or electron transfer in ionic interactions, based on predictions from its placement in the periodic table. Yet, the lack of experimental data limits a complete understanding of these interactions.
The significance of dubnium lies in its role as a superheavy element created in particle accelerators, which aids in the exploration of the periodic table’s boundaries and the stability of heavy nuclei. While dubnium mainly serves as a topic for scientific research, exploring the unknown realms of chemistry, any potential practical applications remain speculative due to its extreme scarcity and transient existence.
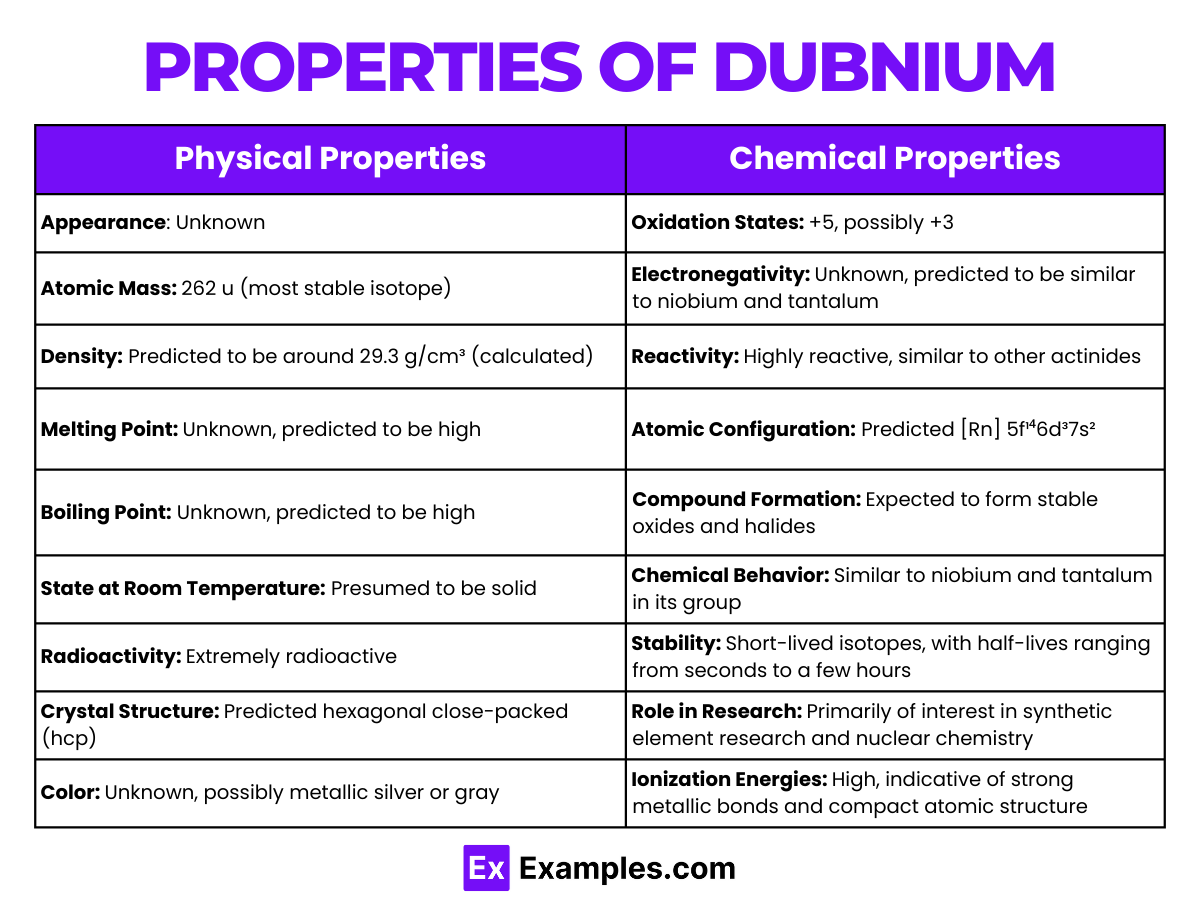
Properties of Dubnium
Physical Properties of Dubnium
| Property | Value |
|---|---|
| Atomic Number | 105 |
| Atomic Mass | [268] |
| State at Room Temperature | Presumed to be solid |
| Melting Point | Unknown, predicted to be similar to that of Ta |
| Boiling Point | Unknown, predicted to be similar to that of Ta |
| Density | Unknown, but predicted based on group trends |
| Color | Unknown, likely metallic |
| Electronegativity | Unknown, predictions based on periodic trends |
| Atomic Radius | Estimated based on computational models |
Chemical Properties of Dubnium
Dubnium (Db), with the atomic number 105, is a synthetic, highly radioactive element belonging to group 5 of the periodic table. Its chemical properties have largely been extrapolated from theoretical studies and the behavior of lighter homologs like tantalum (Ta) and niobium (Nb), given the challenges in observing Dubnium directly due to its scarce production and short half-life.
Reactivity and Compounds
- Oxidation States: The most stable and predominant oxidation state for Dubnium is expected to be +5, consistent with other group 5 elements. This suggests possible compounds like DbO₂ or DbCl₅ under specific conditions.
- Oxides: The oxidation state of +5 suggests the formation of dioxides, akin to TaO₂ and NbO₂, implying the potential formation of Dubnium dioxide (DbO₂) in oxidizing conditions.
Behavior in Solutions
- Aqua Ions: In aqueous solutions, Dubnium is speculated to form complex ions, possibly resembling those of its lighter group 5 analogs. The speculative formation of complexes like [Db(H2O)₅]⁵⁺ in acidic solutions highlights its expected behavior.
Chemical Bonding
- Covalent and Ionic Bonding: Dubnium is likely to engage in both covalent and ionic bonding, influenced by its electron configuration, which is theorized to be [Rn] 5f¹⁴6d³7s² for its most stable isotopes, indicating versatile bonding capabilities.
Relativistic Effects
- Influence on Chemical Properties: The heavy atomic mass and high atomic number of Dubnium suggest notable relativistic effects that might modify its chemical properties in comparison to its lighter homologs. These effects could alter atomic and ionic radii, bond lengths, bond strengths, and electronegativity, potentially leading to unique chemical behaviors not entirely predictable by periodic trends.
Nuclear Properties of Dubnium
| Property | Value |
|---|---|
| Half-lives | Varies by isotope; generally short-lived |
| Isotopes | Known isotopes range from Db-255 to Db-270 |
| Nuclear Spin | Theoretical prediction for specific isotopes |
| Decay Modes | Alpha decay predominantly; some isotopes may undergo spontaneous fission |
Preparation of Dubnium
The preparation of Dubnium (Db), a synthetic element with atomic number 105, involves highly specialized nuclear reactions conducted in particle accelerators. Dubnium does not exist naturally due to its highly unstable and radioactive nature. The creation of Dubnium typically involves bombarding lighter elements with heavy ions to achieve nuclear fusion. Below is a summary of the processes used to synthesize Dubnium:
- Target and Projectile Selection: The preparation begins with the selection of target and projectile materials. The target is usually a heavy element, such as actinides (e.g., Americium, Curium), while the projectile is often a lighter element like nitrogen, oxygen, or calcium ions.
- Acceleration: The projectile ions are accelerated to high energies using a particle accelerator. This acceleration is crucial to overcome the electrostatic repulsion between the positively charged nuclei of the target and projectile.
- Collision and Fusion: The accelerated ions collide with the target material. If the collision has enough energy, it can overcome the repulsive forces, allowing the nuclei to come close enough to fuse, forming a heavier nucleus. This process is known as “hot fusion.”
- Formation of Dubnium: Through hot fusion, new elements are created. For Dubnium, one of the recognized methods involves bombarding a target of Americium-243 (Am) with Neon-22 (Ne) ions.
- Detection and Identification: The products of the nuclear reaction, including Dubnium isotopes, are then separated and identified. This identification is often achieved using techniques such as alpha spectroscopy or liquid chromatography, which can distinguish the decay patterns specific to Dubnium isotopes.
- Stability and Isotopes: Dubnium isotopes are extremely short-lived, with half-lives ranging from a few seconds to a few minutes. The most stable known isotope of Dubnium, Dubnium-268, has a half-life of approximately 28 hours, making the study and characterization of its properties challenging.
Chemical Compounds of Dubnium
1.Dubnium Hexachloride (DbCl₆)
- Equation: Db+3Cl₂→DbCl₆
- Expected to exhibit high stability and be useful in studying Dubnium’s reactivity and coordination chemistry.
2.Dubnium Dioxide (DbO₂)
- Equation:Db+O₂→DbO₂
- Potentially significant for understanding Dubnium’s oxidation states and compound formation.
3.Dubnium Hydroxide (Db(OH)₄)
- Equation:Db⁴⁺+4OH⁻→Db(OH)₄
- Fundamental compound indicating Dubnium’s behavior in aqueous solutions.
4.Dubnium Trifluoride (DbF₃)
- Equation:Db+3F₂→DbF₃
- Exhibiting covalent bonding, akin to other group 5 elements, providing insight into its reactivity.
5.Dubnium Nitrate (Db(NO₃)₄)
- Equation:Db³⁺+4NO₃⁻→Db(NO₃)₄
- Complex compound essential for studying Dubnium’s coordination chemistry and stability.
6.Dubnium Disulfide (DbS₂)
- Equation:Db⁴⁺+2S²⁻→DbS₂
- Sulfur compound assisting in exploring Dubnium’s sulfide chemistry and compound formation.
Isotopes of Dubnium
| Isotope | Mass Number | Half-life | Mode of Decay |
|---|---|---|---|
| Db-255 | 255 | 1.6 seconds | Alpha decay |
| Db-256 | 256 | 1.5 seconds | Alpha decay, Spontaneous fission |
| Db-257 | 257 | 2.2 seconds | Alpha decay |
| Db-258 | 258 | 4.5 seconds | Alpha decay, Spontaneous fission |
| Db-259 | 259 | 0.5 seconds | Alpha decay |
| Db-260 | 260 | 1.52 seconds | Alpha decay, Spontaneous fission |
| Db-261 | 261 | 1.8 seconds | Alpha decay |
| Db-262 | 262 | 34 seconds | Alpha decay, Spontaneous fission |
| Db-263 | 263 | 27 seconds | Alpha decay |
| Db-264 | 264 | 40 seconds | Spontaneous fission |
| Db-265 | 265 | 16 hours | Alpha decay |
| Db-266 | 266 | 1.2 hours | Alpha decay, Spontaneous fission |
| Db-267 | 267 | 3 hours | Alpha decay |
| Db-268 | 268 | 29 hours | Alpha decay |
| Db-270 | 270 | 23.15 hours | Alpha decay |
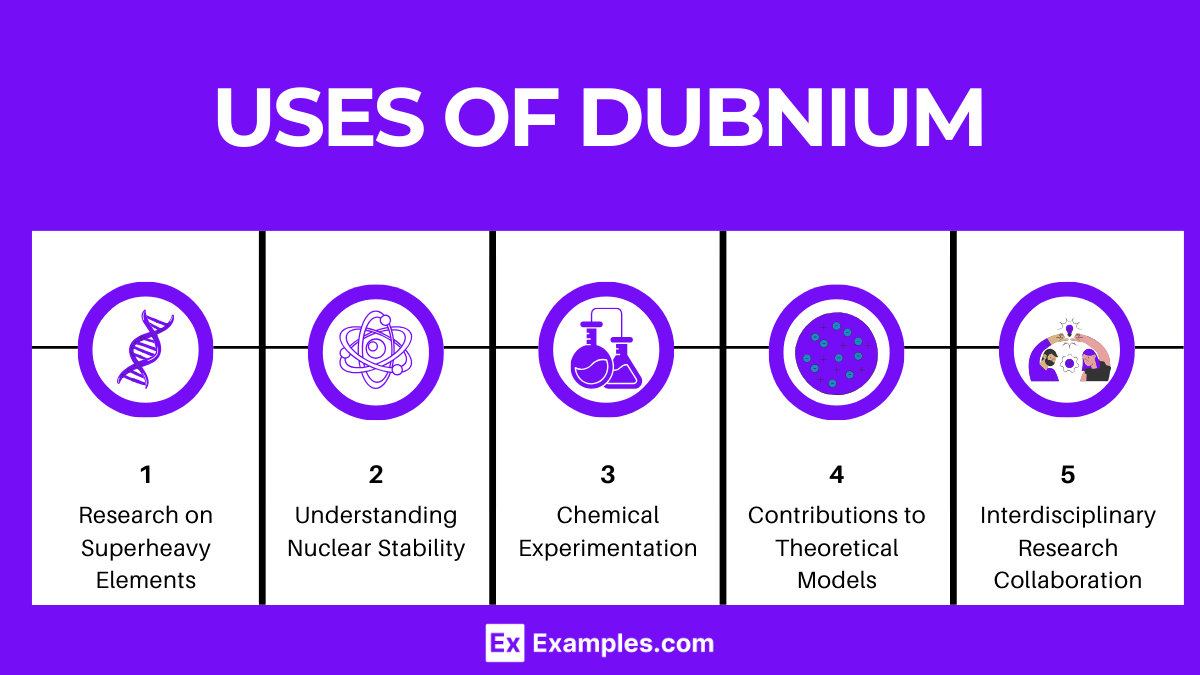
Uses of Dubnium
Dubnium (Db) is a synthetic element with no stable isotopes, making its practical applications outside of scientific research quite limited. The primary use of Dubnium, like many synthetic elements, lies in the field of scientific research, particularly in the areas of nuclear physics and chemistry. Here are some specific contexts in which Dubnium is utilized:
- Research on Superheavy Elements: Dubnium serves as a subject of interest in the study of superheavy elements. Scientists examine its properties and reactions to gain insights into the structure and behavior of the periodic table’s heaviest elements.
- Understanding Nuclear Stability: Research involving Dubnium isotopes contributes to our understanding of nuclear stability and the nuclear shell model. This can help in predicting the stability and properties of other yet-to-be-discovered or synthesized superheavy elements.
- Chemical Experimentation: Although challenging due to its short half-life and the difficulty in producing it, experiments with Dubnium can provide valuable information on the chemical properties of elements in group 5 of the periodic table. This includes potential oxidation states, types of compounds it can form, and its behavior in various chemical environments.
- Contributions to Theoretical Models: The synthesis and study of Dubnium and its compounds contribute to the development and refinement of theoretical models regarding atomic and molecular structures, electron configurations, and the effects of relativistic physics on superheavy elements.
- Pioneering New Synthesis Techniques: The production of Dubnium involves advanced particle accelerator technologies and nuclear reactors. Working with such a challenging element pushes the development of innovative synthesis techniques. These advancements can have broader applications, including the synthesis of other superheavy elements and the enhancement of nuclear research methodologies.
- Interdisciplinary Research Collaboration: The quest to create, isolate, and study Dubnium fosters collaboration across multiple scientific disciplines, including nuclear physics, chemistry, and materials science. These interdisciplinary efforts not only enhance our understanding of Dubnium itself but also encourage the exchange of knowledge and techniques among different scientific communities.
Production of Dubnium
The production of Dubnium (Db), a synthetic and highly radioactive element, is achieved through nuclear reactions in particle accelerators. This process involves bombarding target atoms of lighter elements with ions of other elements at high energies. The methods and reactions used to produce Dubnium are complex, requiring sophisticated equipment and precise control. Here’s an overview of the primary methods used in the production of Dubnium:
Fusion Reactions
- Using Actinides as Targets: One common method involves using actinide elements, such as americium, curium, or californium, as target materials. These target atoms are bombarded with ions of lighter elements like nitrogen, oxygen, or neon.
Cold Fusion Reactions
- Using Lead or Bismuth as Targets: Cold fusion reactions involve bombarding targets of lead or bismuth with ions of heavier elements. This method typically produces fewer neutrons and has been used to synthesize elements closer to Dubnium in the periodic table.
- Example Reaction: A specific cold fusion process used for creating Dubnium might involve the bombardment of lead (²⁰⁸Pb) with chromium (⁵⁰Cr) ions, though this exact reaction is more theoretical for Dubnium’s production and illustrates the approach rather than practical application.
Hot Fusion Reactions
- High-energy Collisions: Hot fusion reactions are characterized by the use of more massive ions and higher energies, leading to the formation of compound nuclei at high excitation energies. This method is more commonly used for the synthesis of heavier superheavy elements.
Challenges and Limitations
- Short Half-lives: Dubnium isotopes have very short half-lives, making it challenging to detect and study them before they decay.
- Low Production Rates: The probability of successful fusion events, known as the cross-section, is extremely low, resulting in very few atoms of Dubnium being produced in each experiment.
Applications of Dubnium
Dubnium (Db), a synthetic and highly radioactive element with no stable isotopes, primarily finds its applications within the realm of scientific research rather than practical, everyday uses. Its incredibly short half-life and the complexity involved in its production mean that Dubnium’s applications are confined to theoretical and experimental domains. Here are some of the key applications of Dubnium:
Scientific Research and Study
- Understanding Element Properties: Dubnium’s synthesis and study contribute to the broader understanding of the properties and behaviors of superheavy elements. Researchers analyze its atomic structure, decay patterns, and chemical reactions to gain insights into the complexities of the periodic table’s heavier elements.
- Exploration of Nuclear Stability: The production and analysis of Dubnium and its isotopes help scientists explore the concept of the “island of stability.” This theoretical region in the chart of nuclides suggests a group of superheavy elements that may possess relatively longer half-lives, offering stability beyond the currently observed superheavy nuclei.
- Development of Nuclear Physics Techniques: The challenges in producing and studying Dubnium drive advancements in experimental techniques and technologies in nuclear physics. The processes used to synthesize, isolate, and analyze Dubnium atoms enhance methodologies applicable across the field, including the discovery and study of other synthetic elements.
Interdisciplinary Impact
- Advancement in Particle Acceleration Technology: Efforts to create Dubnium stimulate technological advancements in particle acceleration and detection. These improvements have implications beyond nuclear chemistry, impacting fields such as materials science, medical physics, and even space exploration by enhancing our ability to manipulate and study atomic and subatomic particles.
- Collaborative Scientific Endeavors: The quest to synthesize and understand Dubnium fosters collaboration among international research institutions, bringing together expertise from nuclear physicists, chemists, and materials scientists. These collaborative efforts not only push the boundaries of our understanding of Dubnium but also promote a broader exchange of knowledge and techniques across scientific disciplines.
Educational Value
- Teaching and Learning: The story of Dubnium’s discovery, production, and research serves as a valuable educational tool in chemistry and physics courses. It exemplifies the scientific method, the challenges of experimental science, and the interdisciplinary nature of modern scientific inquiry, inspiring future generations of scientists.
Dubnium stands as a testament to human curiosity and scientific endeavor, offering insights into the complex nature of superheavy elements. Despite its limited practical applications, the study of Dubnium enriches our understanding of the periodic table, challenges existing theories of nuclear stability, and drives advances in nuclear physics and chemistry, underscoring the invaluable role of research in expanding human knowledge.