Nitrogenase – Definition, Types, Structure, Functions
Nitrogenase is a remarkable enzyme that plays a crucial role in the chemistry of life. This enzyme enables bacteria to convert nitrogen from the atmosphere into complex compounds that plants can use as nutrients. Often found in the root nodules of leguminous plants, nitrogenase catalyzes the conversion process known as nitrogen fixation. This chemical reaction is vital because it helps in the production of amino acids and proteins, essential components for plant growth and survival.
What is Nitrogenase?
Nitrogenase Formula
Nitrogenase, the enzyme responsible for converting atmospheric nitrogen to ammonia, does not have a simple molecular formula like typical chemical compounds due to its complex protein structure. It primarily consists of two proteins: the iron protein (Fe protein) and the molybdenum-iron protein (MoFe protein). The Fe protein, also known as dinitrogenase reductase, has the formula Fe₄S₄, featuring an iron-sulfur cluster. The MoFe protein, also known as dinitrogenase, contains a unique iron-molybdenum cofactor (FeMoco), which can be represented as MoFe₇S₉C-homocitrate. This cofactor is key for the enzyme’s ability to convert nitrogen to ammonia, showcasing nitrogenase’s complexity.
Types of Nitrogenase
Nitrogenase enzymes are classified into three main types, each distinguished by the metal cofactors present in their structure. These types are crucial for the process of nitrogen fixation in various microorganisms.
Molybdenum Nitrogenase
- Description: This is the most common and well-studied type of nitrogenase. It contains molybdenum in addition to iron and sulfur in its cofactor structure.
- Example: Found in many nitrogen-fixing bacteria like Azotobacter and Rhizobium, which are often associated with the root nodules of legumes.
Vanadium Nitrogenase
- Description: Similar to the molybdenum type but contains vanadium instead of molybdenum. It’s used by some bacteria under conditions where molybdenum is scarce.
- Example: Present in organisms such as Azotobacter vinelandii, which can switch between molybdenum and vanadium nitrogenases based on the availability of metals.
Iron-Only Nitrogenase
- Description: This type lacks both molybdenum and vanadium and uses only iron in its active site. It is less efficient than the other types and is usually expressed under extreme conditions of metal limitation.
- Example: Found in some strains of Rhodobacter capsulatus, a photosynthetic bacterium that can fix nitrogen under low molybdenum and vanadium conditions.
Structure of Nitrogenase
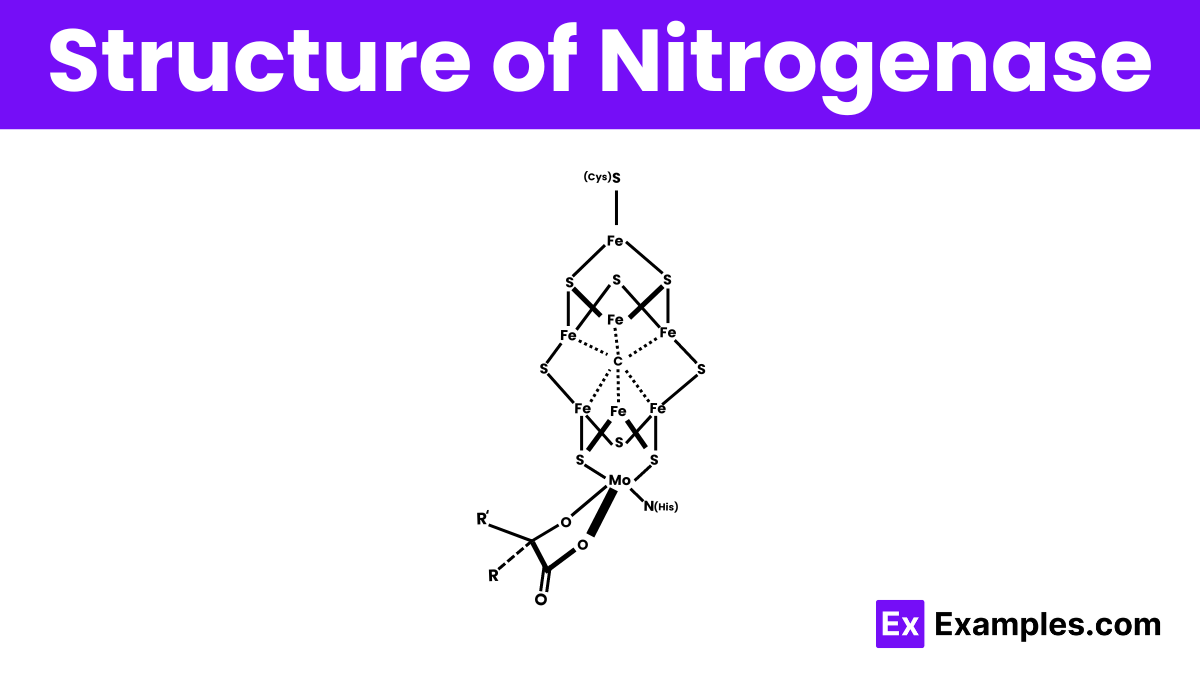
The structure of nitrogenase is complex and consists of two main protein components: the iron (Fe) protein and the molybdenum-iron (MoFe) protein. The Fe protein acts as a catalyst, providing electrons to the MoFe protein through a series of reactions. The MoFe protein, the core of the enzyme, contains a unique metal cluster known as the iron-molybdenum cofactor (FeMoco), which is where the actual nitrogen fixation reaction occurs. This cluster includes atoms of iron, molybdenum, and sulfur, arranged in a way that allows the enzyme to break down molecular nitrogen (N₂) and convert it into ammonia (NH₃), a form usable by living organisms.
Role of Nitrogenase
Nitrogenase plays a critical role in the nitrogen cycle. An essential ecological process that converts inert atmospheric nitrogen (N₂) into ammonia (NH₃), which is then used to synthesize amino acids, proteins, and other vital organic compounds. This enzyme, primarily found in certain bacteria and archaea, enables these microorganisms to fix atmospheric nitrogen, thus fertilizing the soil naturally. Plants, unable to fix nitrogen themselves, rely on this fixed form of nitrogen for growth. As such, nitrogenase is not only pivotal for agriculture and maintaining fertile soil but also supports the base of the food chain. Through this process, nitrogenase facilitates the biological availability of nitrogen, making it crucial for life on Earth.
Functions Of Nitrogenase

Nitrogen Fixation
The primary function of nitrogenase is to convert atmospheric nitrogen (N₂) into ammonia (NH₃). This conversion is vital as it makes nitrogen available in a form that plants and other organisms can use to synthesize amino acids, nucleic acids, and other nitrogen-containing compounds.
Soil Fertility
By enabling nitrogen fixation in soil, nitrogenase helps increase soil fertility. This is especially significant in agricultural settings where leguminous plants, hosting nitrogen-fixing bacteria in their roots, enrich the soil, reducing the need for synthetic fertilizers.
Supporting Biodiversity
Nitrogenase activity supports biodiversity by facilitating the growth of various plant species, which in turn support diverse animal populations. This enzyme’s role in nitrogen fixation helps maintain ecological balance and supports food webs.
Bioremediation
Some nitrogen-fixing bacteria equipped with nitrogenase can also be used in bioremediation to restore nutrient levels in polluted soils. It helping to degrade organic contaminants and stabilize environments.
FAQs
What is Nitrogenase Sensitive to?
Nitrogenase is highly sensitive to oxygen, which can irreversibly damage its activity and structure.
Why is Nitrogenase Important?
Nitrogenase is important for converting atmospheric nitrogen into ammonia, facilitating plant growth and ecosystem sustainability.
Do Bacteria Have Nitrogenase?
Yes, particularly those involved in symbiotic relationships with plants, possess nitrogenase for nitrogen fixation.
Does Nitrogenase Reduce Nitrogen?
Yes, nitrogenase reduces atmospheric nitrogen (N₂) to ammonia (NH₃), a more chemically reactive form usable by organisms.
What Does Nitrogenase Convert to?
Nitrogenase converts atmospheric nitrogen into ammonia, which is essential for synthesizing vital organic compounds in nature.



