Exothermic reaction, Definition, Examples, Differences, FAQ
Exothermic reactions are fascinating chemical processes where energy, in the form of heat, is released into the surrounding environment. This guide delves into the intricacies of exothermic reactions, providing insightful examples to illuminate how they play a crucial role in everyday phenomena and industrial applications. From combustion in engines to the warmth of hand warmers, exothermic reactions are at the heart of countless processes. Understanding their mechanisms offers a window into the dynamic world of chemistry, showcasing the balance between reactants and products in energy exchange. This exploration is essential for students, professionals, and anyone keen on grasping the energetic underpinnings of the material world.
What is an Exothermic Reaction?
An exothermic reaction is a type of chemical reaction that releases energy to its surroundings, primarily in the form of heat, but sometimes as light, electricity, or sound. This release of energy occurs when the total energy of the products is less than the total energy of the reactants. In simpler terms, the chemical bonds formed in the products have more stability (lower energy) than the bonds in the reactants, leading to the excess energy being released.
Understanding the Process
During an exothermic reaction, energy is released as the atoms in the reactants rearrange to form products. This process involves breaking old bonds in the reactants and forming new bonds in the products. Bond breaking requires energy, while bond making releases energy. If the energy released in forming new bonds is greater than the energy consumed in breaking old bonds, the reaction is exothermic.
Examples of Exothermic reaction
Exothermic reactions are widespread in both nature and technology, characterized by their release of energy, typically in the form of heat. Here are some notable examples:
1. Combustion
Combustion is a classic example of an exothermic reaction in which a substance combines with oxygen to produce heat and light. Common instances include the burning of wood in a fireplace, the combustion of gasoline in car engines, and the burning of natural gas in a stove. These reactions release a large amount of energy, which is evident from the flames and heat produced.
2. Respiration
Respiration is a biological process that occurs in living organisms. It involves the breakdown of glucose (or other organic molecules) in the presence of oxygen to produce carbon dioxide, water, and energy. This energy is then utilized by the organism to perform various functions. Respiration is exothermic because it releases energy that the organism can use.
3. Neutralization
Neutralization is a chemical reaction where an acid and a base react to form water and a salt. This reaction is exothermic, releasing heat as the hydrogen ions (H+) from the acid combine with the hydroxide ions (OH−) from the base to form water (H2O). The formation of the ionic salt also releases energy due to the electrostatic attraction between the oppositely charged ions.
4. Formation of Snow in Clouds
The process of snow formation in clouds involves the exothermic reaction of water vapor turning into ice crystals. As water vapor in the clouds cools down, it releases latent heat, which warms the surrounding air slightly, even as the water vapor itself transitions into solid ice forming snowflakes. This process is crucial in the water cycle and weather systems.
5. Rusting of Iron
Rusting is an oxidation reaction where iron reacts with oxygen in the presence of water to form iron oxide, commonly known as rust. This reaction is exothermic, releasing energy as the iron loses electrons to oxygen, a process that gradually weakens the iron or steel objects by causing them to crumble.
6. Synthesis Reactions
Many synthesis reactions, where simpler substances combine to form more complex molecules, release energy. For example, the formation of water from hydrogen and oxygen gas is highly exothermic, releasing energy in the form of heat and sometimes light.
Energy Change in Exothermic Reactions
The energy change in an exothermic reaction can be represented as:
This imbalance results in a net release of energy. In a chemical equation, this is often shown as a negative change in enthalpy ( ), indicating that the system loses energy to its surroundings.
Understanding the Energy Diagram
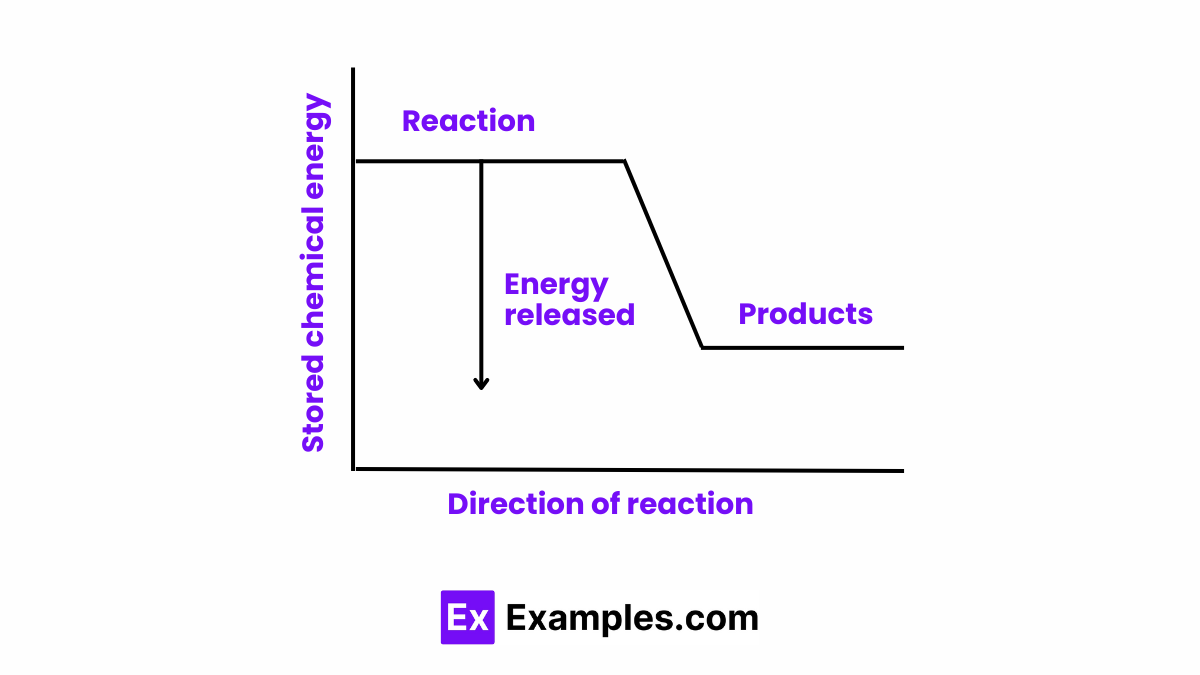
In an energy diagram, an exothermic reaction shows the reactants at a higher energy level than the products. The difference in energy levels represents the energy released to the surroundings. This is visually depicted as a downward slope from reactants to products, highlighting the energy decrease as the reaction proceeds.
Combustion is an Exothermic Reaction
In exothermic reactions, energy changes are characterized by the release of energy from the system to the surroundings. This release occurs because the energy required to break the bonds in the reactants is less than the energy released when new bonds are formed in the products. Essentially, the products of the reaction are at a lower energy level than the reactants, and the difference in energy is released in the form of heat, light, or other forms of energy.
Combustion of Methane
Let’s take the combustion of methane (CH₄) as an example. This reaction involves methane reacting with oxygen (O₂) in the air to produce carbon dioxide (CO₂) and water (H₂O), releasing a significant amount of energy as heat. The reaction can be represented by the following chemical equation:
CH4(g)+2O2(g)→CO2(g)+2H2O(l)+Energy
Energy Changes in Combustion
- Breaking Bonds in Reactants: Initially, energy is absorbed to break the C-H bonds in methane and the O=O bonds in oxygen. This step requires energy input, as breaking bonds always consumes energy.
- Forming Bonds in Products: Subsequently, new bonds are formed when carbon dioxide and water are produced. The formation of C=O bonds in CO₂ and O-H bonds in H₂O releases energy. This step releases more energy than was absorbed in breaking the initial bonds due to the stability of the CO₂ and H₂O molecules compared to the reactants.
- Net Energy Release: The overall reaction is exothermic because the energy released in forming the products’ bonds exceeds the energy required to break the bonds in the reactants. The difference in energy levels between the reactants and products results in a net release of energy to the surroundings.
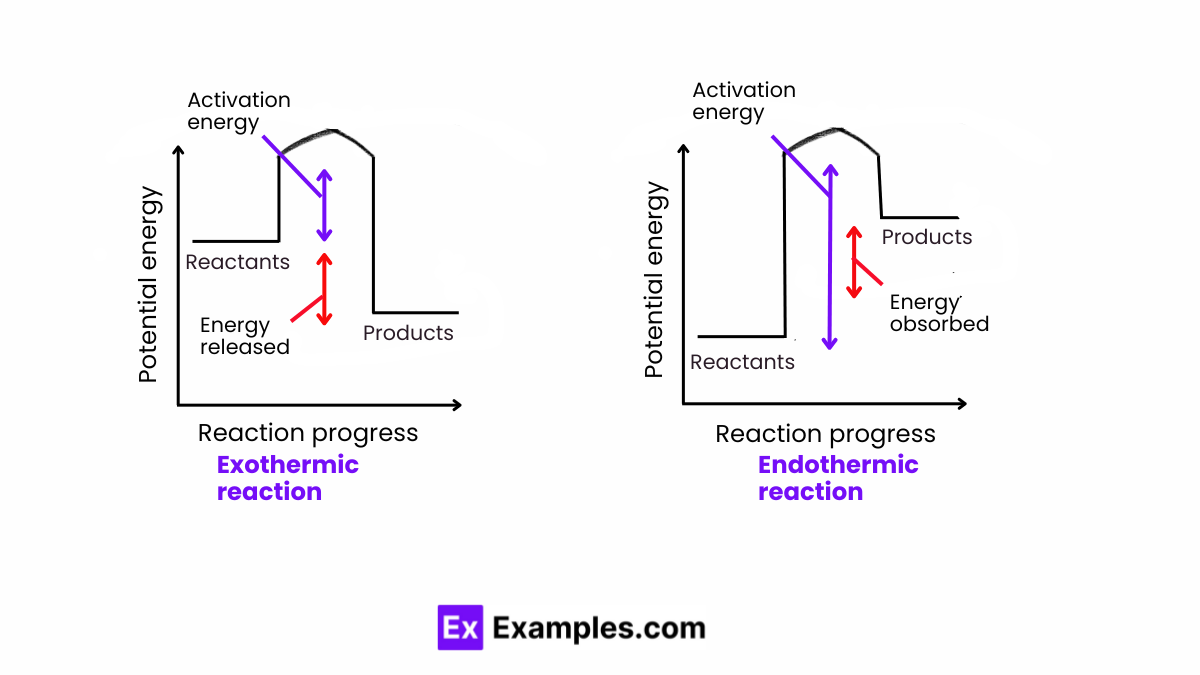
Endothermic vs Exothermic Reaction
Endothermic and exothermic reactions are two fundamental types of chemical reactions that differ primarily in the way energy is transferred between the system and its surroundings during the reaction. Here’s a detailed comparison:
Endothermic Reactions
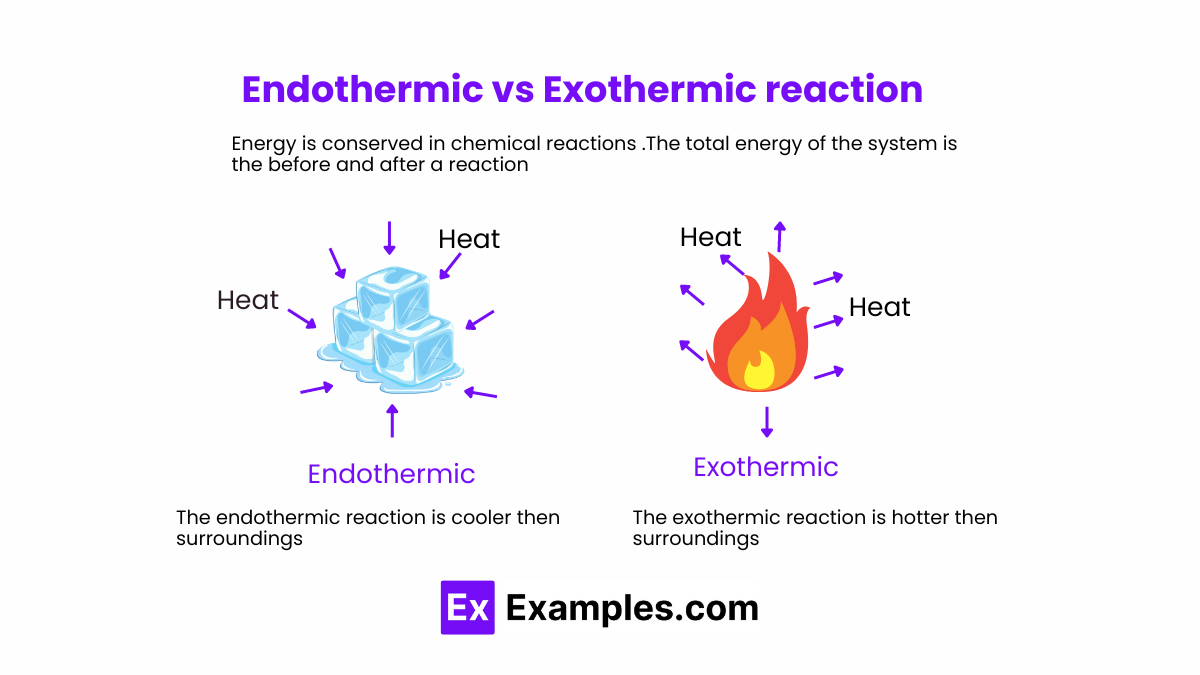
- Energy Absorption: Endothermic reactions absorb energy from their surroundings. This energy is typically in the form of heat, but can also be in other forms like light or electricity.
- Temperature Change: The absorption of energy causes the surrounding temperature to decrease, as energy is taken into the system from the surroundings.
- Energy of Products vs. Reactants: In endothermic reactions, the energy of the products is higher than the energy of the reactants. This is because the energy absorbed from the surroundings is stored in the chemical bonds of the products.
- Examples: Photosynthesis in plants (where sunlight is absorbed to convert carbon dioxide and water into glucose and oxygen), the melting of ice into water, and the evaporation of water into vapor.
Exothermic Reactions
- Energy Release: Exothermic reactions release energy to their surroundings, usually in the form of heat, but sometimes as light or sound.
- Temperature Change: The release of energy causes the surrounding temperature to increase, as energy is emitted from the system to the surroundings.
- Energy of Products vs. Reactants: In exothermic reactions, the energy of the products is lower than the energy of the reactants. The difference in energy, which is released during the reaction, reflects the greater stability (lower energy) of the products’ chemical bonds compared to those of the reactants.
- Examples: Combustion of fuels (like burning wood or gasoline), respiration in cells (where glucose is broken down to release energy), and the synthesis of water from hydrogen and oxygen gas.
Key Differences in a Nutshell
- Direction of Energy Flow: The fundamental difference lies in the direction of energy flow. Endothermic reactions absorb energy, leading to a cooling effect, whereas exothermic reactions release energy, causing a warming effect.
- Applications: The differences between these reactions have practical applications. Endothermic processes are used in cooling technologies, such as refrigeration and air conditioning, where energy absorption is desirable. Exothermic reactions are harnessed for heating and energy production, evident in combustion engines and power plants.
Why is Respiration an Exothermic Reaction?
Respiration is an exothermic reaction because it releases energy to the surroundings, primarily in the form of heat. This biological process involves the breakdown of glucose (or other nutrients) with oxygen to produce carbon dioxide, water, and energy. The energy released during this process is a result of the chemical reactions that occur in the cells of living organisms.
The Process of Respiration
Respiration can be summarized by the following chemical equation:
C6H12O6+6O2→6CO2+6H2O+Energy
In this equation, glucose () reacts with oxygen () to produce carbon dioxide (), water (), and energy.
Energy Release
- Breaking Bonds: Initially, energy is used to break the chemical bonds in glucose and oxygen molecules. Although this step requires energy input, it is less than the total energy released in the later steps of the process.
- Forming New Bonds: The formation of water and carbon dioxide involves creating new chemical bonds. This step releases a significant amount of energy because the new bonds in the products are more stable (lower energy) than the bonds in the reactants.
- Net Energy Release: The overall process releases energy because the energy required to break the bonds in the reactants is less than the energy released by forming the bonds in the products. This net release of energy is captured in the form of ATP (adenosine triphosphate) molecules, which cells use as a direct source of energy for various biological functions.
Why It’s Exothermic
The exothermic nature of respiration is crucial for living organisms because it provides the energy needed for vital processes, including growth, repair, and maintenance of cellular functions. The heat produced as a byproduct also helps maintain body temperature in warm-blooded animals, contributing to homeostasis. Thus, respiration is an exothermic reaction because it efficiently converts the chemical energy stored in food into usable energy for the cell, releasing excess energy as heat.
Exothermic Reaction FAQ?
What Is an Exothermic Reaction?
An exothermic reaction releases energy, usually as heat, to its surroundings, making the environment warmer. Examples include combustion, rusting of iron, and cellular respiration.
How Do Exothermic Reactions Occur?
Exothermic reactions occur when the energy needed to break bonds in reactants is less than the energy released by forming bonds in products, resulting in a net energy release.
Why Are Exothermic Reactions Important?
Exothermic reactions are crucial for various applications, including energy production in power plants, heating systems, and biological processes like respiration that sustain life.
Can Exothermic Reactions Be Reversed?
While the specific conditions of exothermic reactions can be reversed, leading to endothermic reactions, the original exothermic process itself doesn’t reverse but can be part of a reversible cycle.
What Are Examples of Exothermic Reactions?
Common examples of exothermic reactions include the combustion of fuels (like wood or gasoline), the synthesis of water from hydrogen and oxygen, and the process of rusting.
How Do Exothermic Reactions Affect Temperature?
Exothermic reactions increase the temperature of their surroundings by releasing energy, making them integral to processes requiring heat, such as cooking and industrial manufacturing.
Exothermic reactions play a pivotal role in both nature and technology by releasing energy, predominantly as heat. These reactions are fundamental to various processes, from powering engines and heating homes to sustaining life through respiration. Understanding exothermic reactions enhances our ability to harness energy efficiently and innovate in fields like energy production, environmental science, and chemical engineering.