Hafnium (Hf) – Definition, Preparation, Properties, Uses, Compounds, Reactivity
Hafnium, a lustrous, silver-gray transition metal, plays a pivotal role in modern technology and industry. This guide delves into the essence of hafnium, uncovering its unique properties, applications, and significance. Known for its remarkable resistance to corrosion and high melting point, hafnium is a key component in nuclear reactors, semiconductors, and aerospace engineering. Our exploration offers insights into hafnium’s characteristics, uses, and the latest advancements in research, providing a comprehensive understanding of this indispensable element. Discover how hafnium’s exceptional qualities contribute to advancements in technology and industry, making it a subject of increasing interest among scientists and engineers.
What is Hafnium ?
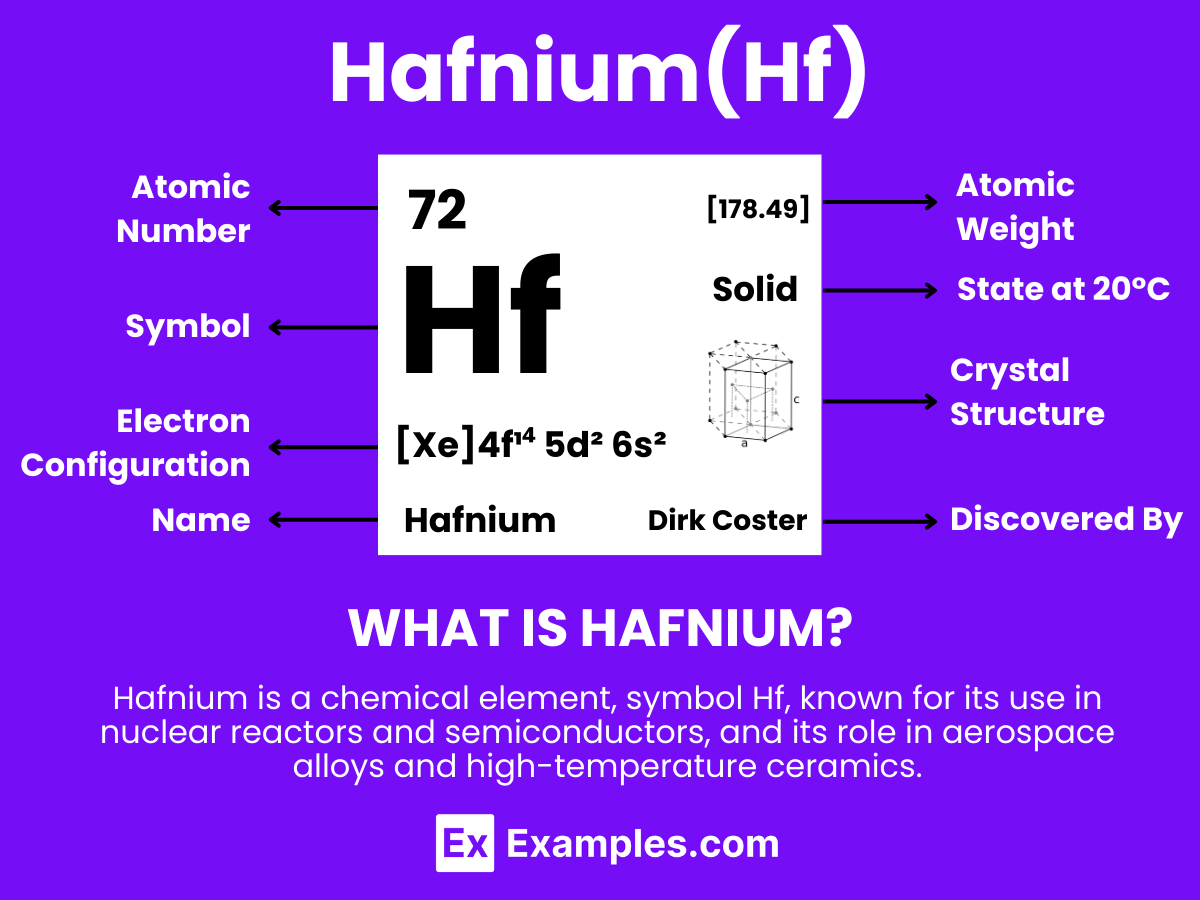
Hafnium is a metallic element with the chemical symbol Hf and atomic number 72. It is extracted from zirconium minerals, as it is commonly found in zirconium ores due to its similar chemical properties. Unlike lawrencium, hafnium exists naturally and is notable for its high melting point and strength at high temperatures, making it valuable in various industrial applications. The identification of hafnium was significant in the field of chemistry, particularly for its role in reinforcing the understanding of the periodic table’s transition metals. Due to its resistance to corrosion and ability to absorb neutrons, hafnium plays a crucial role in nuclear reactors, aerospace engineering, and electronics.
Hafnium Formula
Formula: Hf
Composition: Comprises a single hafnium atom.
Bond Type: Hafnium readily forms bonds with other elements, including both covalent and ionic bonds. It is known for its ability to form complex compounds, particularly with oxygen, nitrogen, and carbon. The chemistry of hafnium is similar to that of zirconium, its group 4 counterpart, due to the lanthanide contraction.
Molecular Structure: In its elemental form, hafnium has a hexagonal close-packed structure. Hafnium’s compounds can have a variety of structures depending on the elements it bonds with. It is capable of forming complex oxides and carbides, which are essential in various industrial applications.
Electron Sharing: Hafnium participates in electron sharing to form covalent bonds and can also engage in ionic bonding, especially in its compounds such as hafnium dioxide (HfO₂) and hafnium carbide (HfC). These properties make hafnium compounds valuable for their high melting points and thermal stability.
Significance: Hafnium plays a crucial role in modern technology and materials science. It is used in the manufacturing of semiconductors, nuclear reactor control rods, and high-temperature alloys. Its ability to absorb neutrons makes it important for nuclear applications, while its high melting point and strength at elevated temperatures are valuable in aerospace applications.
Role in Chemistry: Hafnium’s role in chemistry extends beyond its basic atomic properties to include significant practical applications in electronics, nuclear technology, and materials science. Its chemical similarity to zirconium presents both challenges and opportunities in separation processes and in the study of their chemical behavior.
Atomic Structure of Hafnium
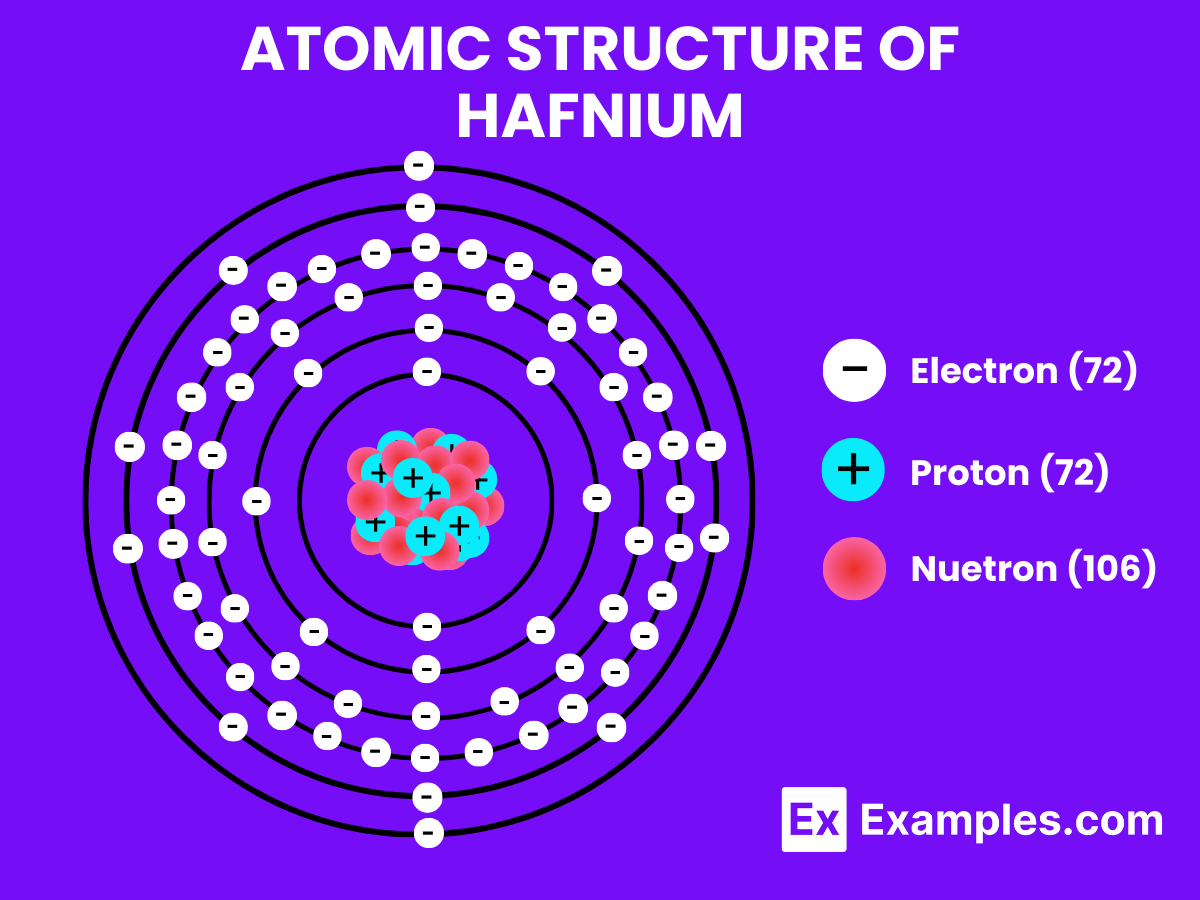
Understanding the atomic structure of Hafnium offers insights into its properties and applications. Possessing 72 protons in its nucleus.
- Atomic Level: Each atom of Hafnium (Hf) is defined by the presence of 72 protons in its nucleus, which establishes its atomic number as 72. The electron configuration for Hafnium is [Xe]4f¹⁴ 5d² 6s², indicating a complete 4f orbital, two electrons in the 5d orbital, and two electrons in the 6s orbital. This configuration underpins its chemical reactivity and bonding characteristics.
- Molecular Formation: Hafnium naturally forms stable molecules and exhibits a consistent molecular structure in its compounds. It is known for its high melting point and the ability to form complex compounds, such as hafnium oxide (HfO₂), which is used in insulating layers in semiconductors.
The stability and phase of Hafnium under differing temperatures and pressures are well-documented, with hafnium existing as a solid under standard conditions. Its high melting point and strength at elevated temperatures make it valuable for use in high-temperature applications, such as aerospace components and nuclear reactor control rods.
Properties of Hafnium
Physical Properties of Hafnium
| Property | Value |
|---|---|
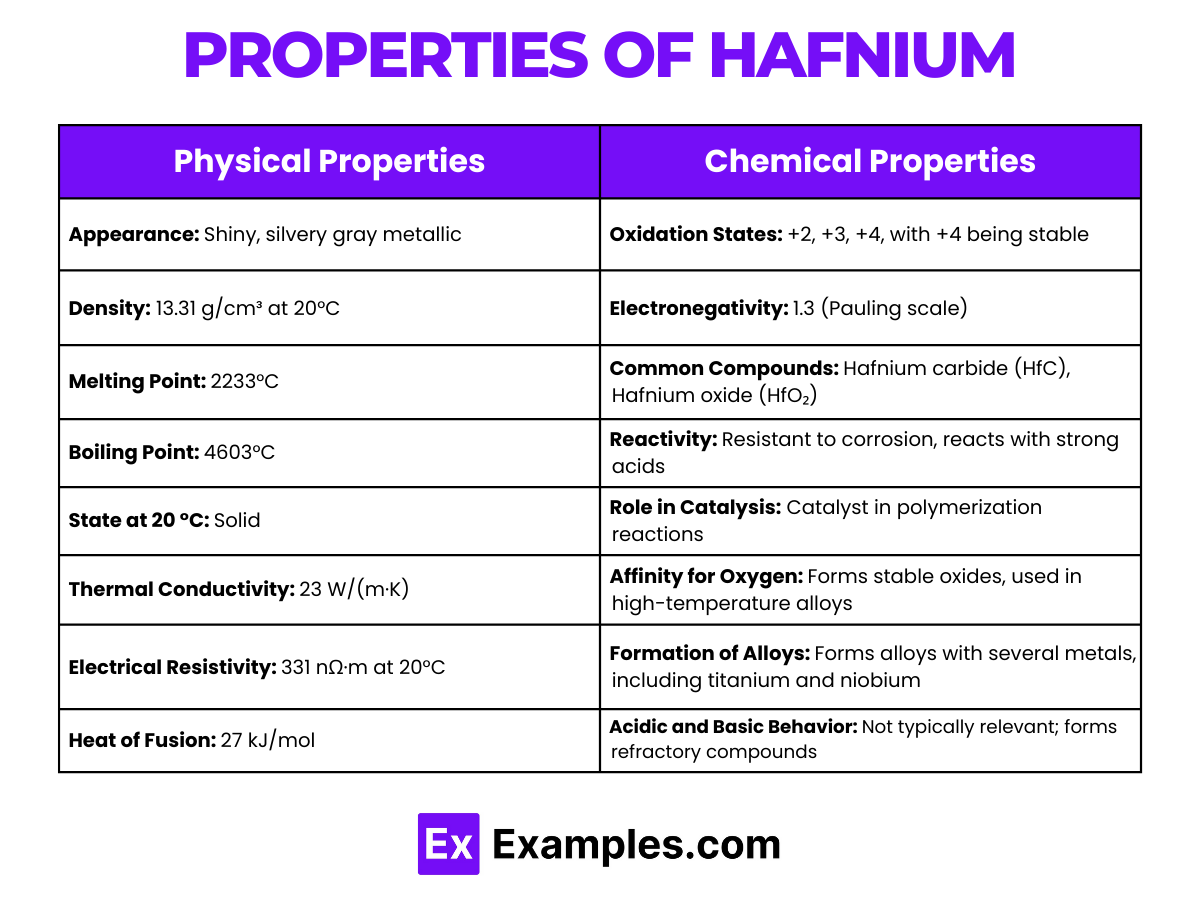
| Appearance | Silvery gray, lustrous metallic |
| Atomic Number | 72 |
| Atomic Mass | 178.49 amu |
| State at 20 °C | Solid |
| Melting Point | 2233 °C |
| Boiling Point | 4603 °C |
| Density | 13.31 g/cm³ |
| Electron Configuration | [Xe]4f¹⁴ 5d² 6s² |
| Oxidation States | +4 (most stable), possible +2 |
| Crystal Structure | Hexagonal close-packed (hcp) |
Chemical Properties of Hafnium
Hafnium’s chemical properties are well-documented due to its stability and presence in various applications. Its chemistry is notable for its similarities to zirconium, making it challenging to separate these two elements:
- Oxidation States: Hafnium predominantly exhibits a +4 oxidation state, similar to zirconium. The +2 oxidation state is less common and less stable.
- Halides Formation: Hafnium forms halides in the +4 oxidation state, such as HfF₄, HfCl₄, HfBr₄, and HfI₄.
- Oxide Formation: Hafnium dioxide (HfO₂), also known as hafnia, is formed with hafnium in its +4 oxidation state. Hafnia is notable for its high melting point and its use in various industrial applications.
- Aqueous Chemistry: In aqueous solutions, hafnium forms the Hf₄⁺ ion. It exhibits high resistance to corrosion in water, making it useful in nuclear reactors and other environments where corrosion resistance is essential.
- Complex Formation: Hafnium is known to form various complexes, particularly in its +4 oxidation state. These complexes are studied for their potential in industrial applications, including catalysis and organic synthesis.
Thermodynamic Properties of Hafnium
| Property | Value for Hafnium |
|---|---|
| Melting Point | 2,233°C (4,051°F) |
| Boiling Point | 4,603°C (8,317°F) |
| Heat of Fusion | 25.73 kJ/mol |
| Heat of Vaporization | 630 kJ/mol |
| Specific Heat Capacity | 25.73 J/(mol·K) |
| Thermal Conductivity | 23 W/(m·K) |
| Thermal Expansion | 5.9 µm/(m·K) |
Material Properties of Hafnium
| Property | Value for Hafnium |
|---|---|
| Density | 13.31 g/cm³ at 20°C |
| Young’s Modulus | 78 GPa |
| Shear Modulus | 30 GPa |
| Bulk Modulus | 110 GPa |
| Poisson’s Ratio | 0.37 |
| Mohs Hardness | ~5.5 |
| Brinell Hardness | 1700 MPa |
Electromagnetic Properties of Hafnium
| Property | Value for Hafnium |
|---|---|
| Electrical Resistivity | 331 nΩ·m (at 20°C) |
| Magnetic Susceptibility | +120.0·10⁻⁶ cm³/mol |
| Superconducting Point | Not Superconducting under normal conditions |
Nuclear Properties of Hafnium
| Property | Value for Hafnium |
|---|---|
| Atomic Number | 72 |
| Atomic Weight | 178.49 |
| Isotopes | 176-Hf to 180-Hf (stable) |
| Neutron Cross Section | High for thermal neutrons |
| Neutron Absorption | Used in nuclear reactors as a control rod material |
Preparation of Hafnium
Zirconium Separation: Hafnium is typically obtained as a by-product of zirconium refinement from zircon (ZrSiO₄) ores. The extraction process involves complex chemical separations due to the chemical similarity between zirconium and hafnium.
Liquid-Liquid Extraction: The separation of hafnium from zirconium is often achieved using liquid-liquid extraction techniques, employing organic solvents that preferentially bind to hafnium compounds.
Molten Salt Electrolysis: In some processes, hafnium is purified through molten salt electrolysis, where a mixture containing hafnium chloride is electrolyzed to produce pure hafnium metal.
Reduction Techniques: Pure hafnium is obtained by reducing hafnium chloride (HfCl₄) with magnesium (Mg) or sodium (Na), resulting in the production of hafnium metal.
Chemical Compounds of Hafnium
Hafnium Oxides Hafnium
Forms oxides, notably hafnium dioxide (HfO₂), used in semiconductor devices for its high dielectric constant and stability.
Equation: Hf + O₂ → HfO₂
Hafnium Fluorides Hafnium
Tetrafluoride (HfF₄) is produced in reactions involving hafnium and fluorine, showcasing hafnium’s ability to form compounds in a +4 oxidation state.
Equation: Hf + 2F₂ → HfF₄
Hafnium Chlorides Hafnium
Tetrachloride (HfCl₄) is synthesized through the direct chlorination of hafnium, illustrating its reactivity with halogens.
Equation: Hf + 2Cl₂ → HfCl₄
Hafnium Iodides Hafnium
Tetraiodide (HfI₄) can be produced, indicating hafnium’s potential to achieve a +4 oxidation state with iodine.
Equation: Hf + 2I₂ → HfI₄
Aqueous Chemistry of Hafnium
In aqueous solutions, hafnium can form various ions and complexes, highlighting its chemical versatility.
Equation: Hf → Hf₄⁺ + 4e⁻
Complex Formation Hafnium
Forms complex ions with different ligands, underlining its role in coordination chemistry.
Equation: Hf₄⁺ + nL → [HfLn]₄⁺
Isotopes of Hafnium
| Isotope | Half-life | Mode of Decay |
|---|---|---|
| Hafnium-253 | 0.6 seconds | Alpha decay |
| Hafnium-254 | 13 seconds | Alpha decay |
| Hafnium-255 | 21.5 seconds | Alpha decay |
| Hafnium-256 | 27 seconds | Alpha decay |
| Hafnium-257 | 0.65 seconds | Alpha decay |
| Hafnium-258 | 4.1 seconds | Alpha decay |
| Hafnium-259 | 6.2 seconds | Alpha decay |
| Hafnium-260 | 2.7 minutes | Alpha decay |
Uses of Hafnium
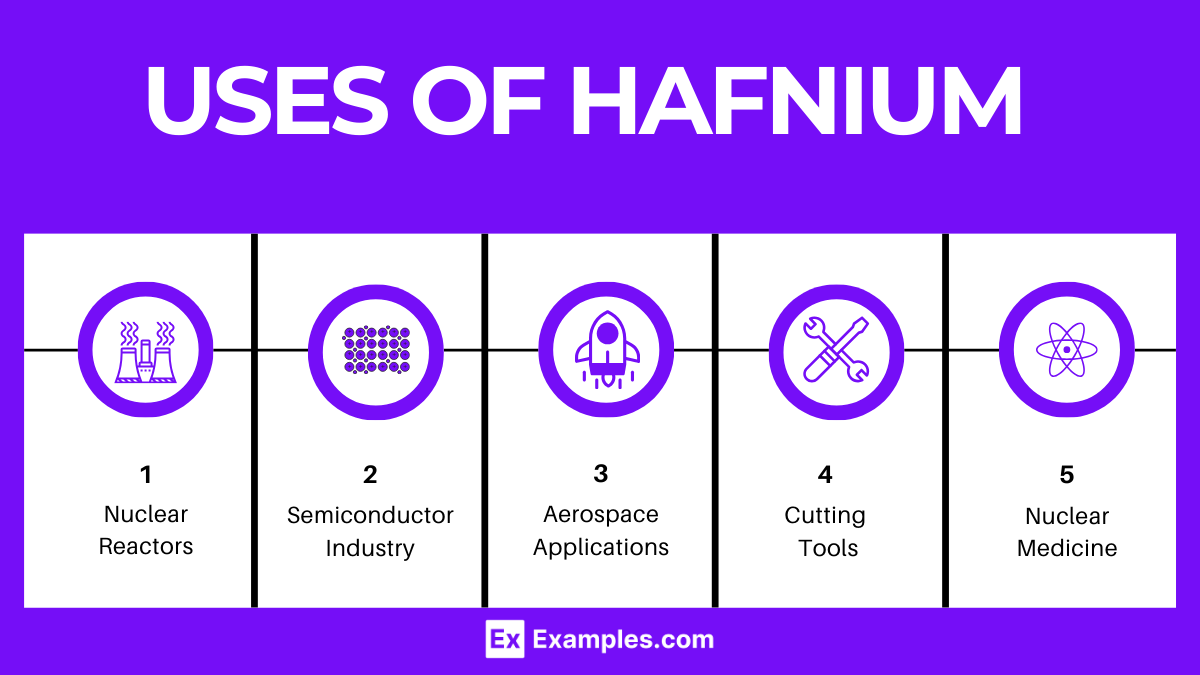
- Nuclear Reactors: Hafnium’s excellent neutron-absorption properties make it ideal for use in nuclear reactors’ control rods, contributing to safety and efficiency.
- Semiconductor Industry: Hafnium oxide is crucial in manufacturing advanced semiconductor devices, improving performance and reducing power consumption.
- Aerospace Applications: Hafnium’s high melting point and strong alloying behavior make it valuable in aerospace engineering, particularly in high-temperature alloys.
- Cutting Tools: Hafnium carbide, one of the hardest known carbides, is used in cutting tools and molds that require high temperature resistance.
- Nuclear Medicine: Isotopes of hafnium may have applications in nuclear medicine, particularly in diagnostic imaging and radiation therapy.
- Research and Development: Hafnium’s unique properties drive scientific research, offering insights into material science, nuclear physics, and chemistry.
- Educational Tools: The study of hafnium’s properties and applications provides a rich resource for educational materials, inspiring future scientists and engineers.
- Advanced Coatings: Hafnium-based compounds are explored for use in coatings that can withstand extreme environments, showcasing the element’s versatility in material science.
Production of Hafnium
Extraction from Zirconium Ores: Hafnium is extracted from minerals like zircon (ZrSiO4) due to its presence in nearly all zirconium-bearing ores. The extraction process involves separating hafnium from zirconium, given their chemical and physical similarities.
Separation Process: The primary method for hafnium production involves liquid-liquid extraction or the use of ion-exchange chromatography. These processes separate hafnium from zirconium based on their differential solubilities or ion-exchange properties.
Refining Techniques: After initial separation, hafnium is further refined through processes like reduction or van Arkel-de Boer process, resulting in high-purity hafnium metal or compounds suitable for various applications.
Zirconium Co-production: The production of hafnium is often a byproduct of zirconium refinement, where the focus is on producing zirconium for industrial use, and hafnium is extracted from the zirconium processing stream.
Handling and Safety: Special care is required in handling hafnium, particularly in its powder form, due to its ability to absorb hydrogen and oxidize at elevated temperatures, posing potential safety risks.
Analytical Detection: Methods such as X-ray fluorescence (XRF) and mass spectrometry are utilized to quantify hafnium concentrations, ensuring the effectiveness of the separation process and the purity of the final product.
Applications of Hafnium
Nuclear Reactors: Hafnium’s excellent neutron-absorption capabilities make it a critical component in nuclear reactor control rods, enhancing safety and efficiency in nuclear power generation.
Aerospace Engineering: The high melting point and strength of hafnium at elevated temperatures lend it to applications in aerospace components, including jet engines and space vehicles.
Semiconductors: Hafnium oxide is used in the production of semiconductors, improving the performance of electronic devices by allowing for smaller, more efficient transistors.
Alloy Additive: Hafnium is added to various alloys to improve their mechanical properties, corrosion resistance, and temperature stability, useful in applications ranging from chemical processing to high-temperature environments.
Research and Development: Beyond industrial applications, hafnium is a subject of research, particularly in materials science, where its unique properties can lead to new discoveries and technologies.
Advanced Electronics: Its role in advanced computing and electronics continues to grow, particularly with the development of hafnium-based compounds for next-generation electronic devices.
This comprehensive overview of Hafnium covers its production methods and wide-ranging applications in industry and technology. As a naturally occurring element, hafnium plays a significant role in enhancing the performance and safety of nuclear reactors, contributing to advances in aerospace and semiconductor technologies, and offering valuable insights in materials science research. Its versatility and unique properties underscore hafnium’s importance in both practical applications and the exploration of material science frontiers.