Hassium (Hs) – Definition, Preparation, Properties, Uses, Compounds, Reactivity
The enigmatic world of Hassium, a synthetic marvel in the periodic table, through our comprehensive guide. Delving into the essence of Hassium, this section unfolds its creation, characteristics, and scientific significance. With expert insights and illustrative examples, we illuminate Hassium’s place in nuclear chemistry and its parallelism with other group 8 elements. Our guide is designed to satisfy curiosity, enhance knowledge, and present Hassium in a new light, making it a pivotal resource for enthusiasts and scholars alike. Engage with the fascinating narrative of Hassium, and unlock the secrets of this elusive element.
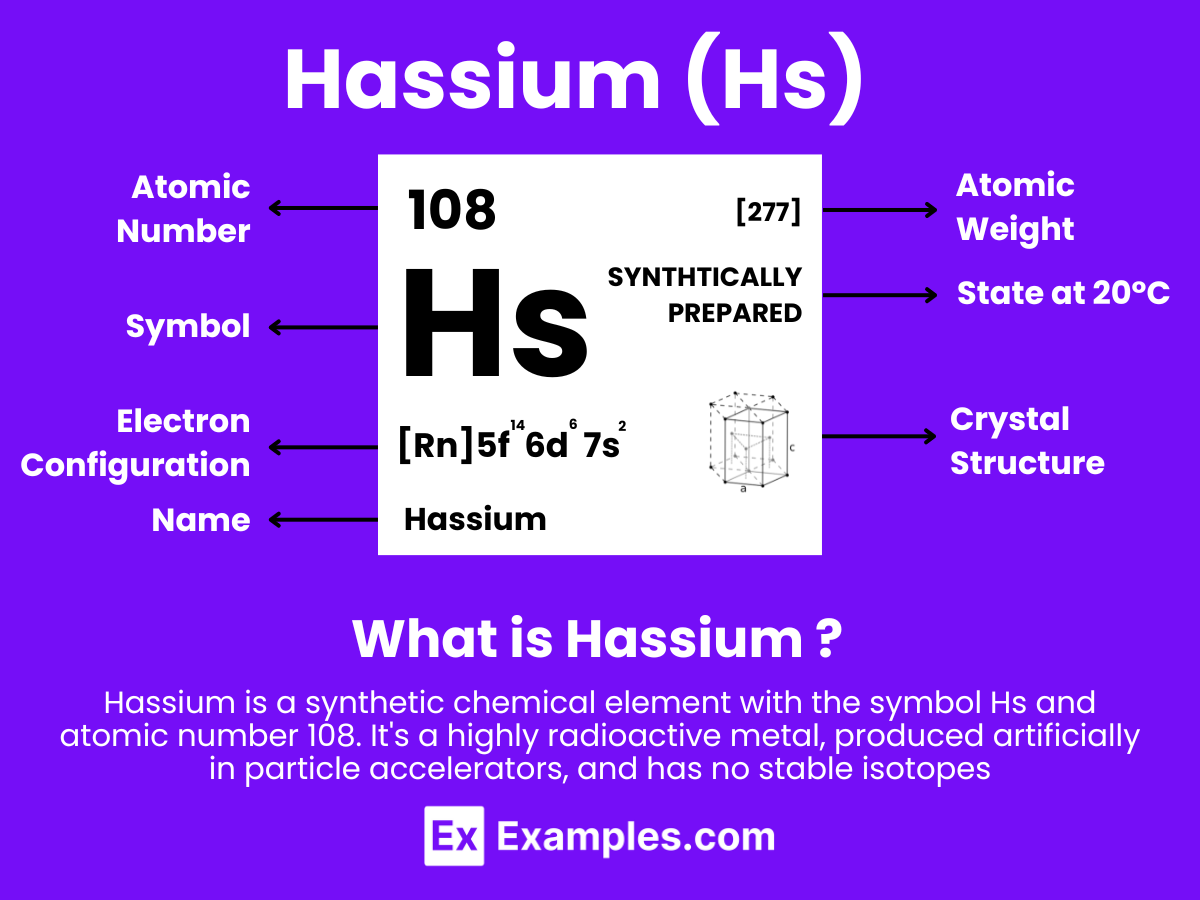
Hassium is a chemical element with the symbol Hs and atomic number 108. It is a synthetic element, and not found in nature. Hassium is produced artificially in a laboratory through the nuclear fusion of lighter atoms. Specifically, it is usually created by bombarding atoms of lead with nuclei of iron in a heavy ion accelerator.
Hassium is a member of the transactinide elements and is also part of the 7th period in the d-block of the periodic table, which means it is one of the heavy metals. Being a synthetic element, it has no significant applications outside of scientific research due to its extremely limited availability and its radioactive properties. Hassium has several isotopes, all of which are radioactive, and the most stable known isotope, hassium-277, has a half-life of approximately 11 minutes
Hassium Formula
- Formula: Hs
- Composition: Consists of a single hassium atom.
- Bond Type: In its elemental form, hassium does not form bonds as it is a pure element. However, hassium can engage in covalent or ionic bonding when reacting with other elements.
- Molecular Structure: As a pure element, hassium does not exhibit a molecular structure in the traditional sense, like compounds such as H₂O. Being a synthetic and highly radioactive element, its solid-state structure is not well-characterized but is predicted to have a hexagonal close-packed crystalline structure, similar to other group 8 elements.
- Electron Sharing: In compounds, hassium is expected to share electrons covalently or transfer electrons ionically, depending on the nature of the other element(s) it bonds with, though practical examples are limited due to its short half-life and challenging synthesis.
- Significance: Hassium’s importance lies in its contribution to research in nuclear chemistry and physics. It helps scientists understand the properties of superheavy elements and the limitations of the periodic table. Its synthesis and study push the boundaries of experimental techniques and theoretical models.
- Role in Chemistry: Hassium’s role is primarily in scientific research. Due to its radioactivity and the difficulty in producing it, hassium has no commercial applications. It is of interest for studying the chemical properties of transactinides and exploring the effects of relativistic quantum mechanics on heavy elements.
Atomic Structure of Hassium
Hassium, unlike hydrogen, is a superheavy synthetic element with distinctive characteristics, including a presumed high melting point and an unknown boiling point, indicating theoretical predictions about its phase under normal conditions are primarily speculative due to its short half-life and instability. Hassium’s behavior at the atomic and molecular levels is quite different from that of hydrogen due to its position in the periodic table as a member of the transition metals and its presumed metallic nature.
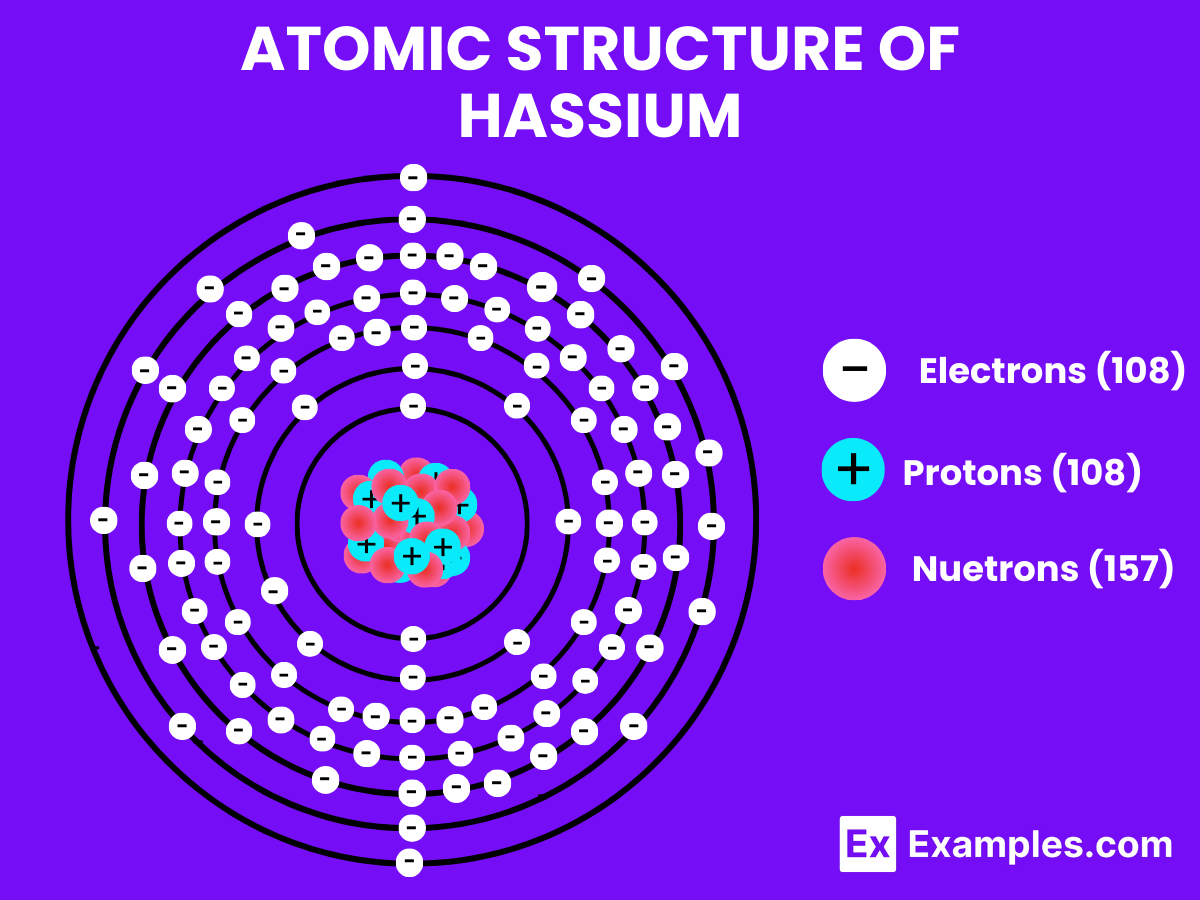
Atomic Level: Each hassium atom (Hs) contains 108 protons in its nucleus and has 108 electrons orbiting around it. The electron configuration of hassium is predicted to be [Rn] 5f¹⁴ 6d⁶ 7s², which means it has six electrons in its outermost shell that could be available for bonding, based on theoretical calculations.
Molecular Formation: In its metallic form, hassium is expected not to form molecules like H₂. Instead, if stable enough to form a solid, hassium atoms would theoretically be arranged in a crystalline lattice structure. This structure would involve the sharing of electrons between many hassium atoms in a metallic bond, which is different from the covalent bonding seen in hydrogen molecules. Due to its very short half-life and the fact that only a few atoms have ever been produced, studies on hassium’s physical and chemical properties, including its phase at various temperatures, are largely theoretical.
Properties of Hassium
Physical Properties of Hassium
| Physical Property | Description of Hassium |
|---|---|
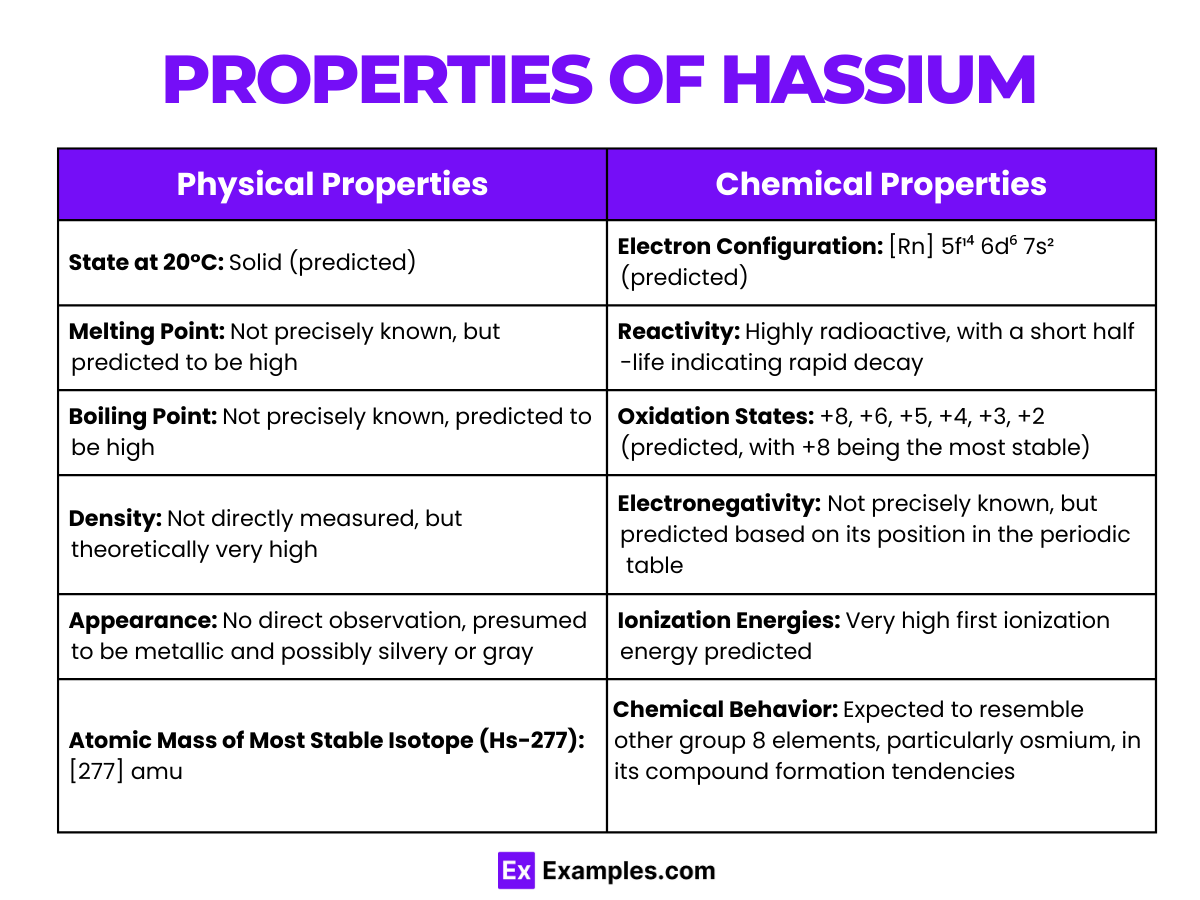
| State at 20°C | Predicted to be solid |
| Melting Point | Unknown, but predicted to be high due to its position in the periodic table |
| Boiling Point | Unknown, but similarly predicted to be high |
| Density | Estimated to be around 41 g/cm³ (predicted) |
| Appearance | Unknown, but possibly metallic and silvery in appearance (predicted) |
| Atomic Mass | [277] (Most stable isotope Hs-277, theoretical) |
Chemical Properties of Hassium
The chemical properties of Hassium (Hs), like those of many superheavy elements, are largely based on theoretical calculations and predictions rather than extensive experimental data, due to its extremely short half-life and the difficulty in producing significant quantities. However, as a member of group 8 of the periodic table, Hassium is expected to exhibit chemical properties similar to its lighter homologs: iron (Fe), ruthenium (Ru), and osmium (Os). Here’s a detailed look at the anticipated chemical properties of Hassium:
Oxidation States
Hassium is predicted to show a variety of oxidation states, with +8 being the most stable and characteristic, similar to osmium. However, oxidation states ranging from +2 to +6 may also be possible in various compounds, reflecting the versatility seen in its lighter counterparts.
Chemical Reactions and Compounds
- Oxides: Hassium is expected to form oxides in its stable oxidation state. A predicted oxide is Hassium tetroxide (HsO₄), analogous to OsO₄(osmium tetroxide), with a tetrahedral molecular geometry. The formation of this compound could potentially be represented by the equation:
- Halides: Similar to other group 8 elements, Hassium is likely to form halides, such as Hassium tetrachloride (HsCl₄), in its +4 oxidation state. The reaction with chlorine gas might be represented as: l₂HsCl₄
- Complexes: Like ruthenium and osmium, Hassium is expected to form a variety of complex compounds with different ligands, showcasing its ability to participate in complex chemistry. For example, Hassium hexafluoride (HsF₆) could form, following the behavior of its lighter homologs in similar conditions.
Atomic and Ionic Radii
The atomic and ionic radii of Hassium are expected to be comparable to those of its group 8 counterparts, adjusted for relativistic effects due to its high atomic number. This means that Hassium atoms and ions would likely be slightly smaller than those of osmium, due to the contraction of electron orbitals caused by the strong electromagnetic field of the nucleus.
Electronegativity and Electron Affinity
While specific values for Hassium’s electronegativity and electron affinity are not precisely known, they are predicted to follow the trend seen in group 8 elements. This suggests a relatively high electronegativity, indicative of its ability to attract electrons in chemical bonds, and a positive electron affinity, suggesting it can gain electrons to form anions, although this is less likely due to its metallic nature.
Thermodynamic Properties of Hassium
| Thermodynamic Property | Value (Predicted) |
|---|---|
| Melting Point | High (Exact value unknown) |
| Boiling Point | High (Exact value unknown) |
| Heat of Fusion | Unknown |
| Heat of Vaporization | Unknown |
| Specific Heat Capacity | Unknown |
| Thermal Conductivity | Similar to osmium (Predicted) |
Material Properties of Hassium
| Material Property | Value (Predicted) |
|---|---|
| State at 20°C | Solid |
| Density | Approx. 41 g/cm³ (Predicted) |
| Atomic Mass | 277 (Most stable isotope Hs-277) |
| Atomic Volume | Unknown |
| Electrical Conductivity | Similar to other group 8 elements (Predicted) |
| Hardness | Similar to osmium (Predicted) |
Electromagnetic Properties of Hassium
| Electromagnetic Property | Description |
|---|---|
| Electrical Conductivity | Predicted to be high, typical of metals |
| Magnetic Susceptibility | Likely to be paramagnetic or possibly diamagnetic |
| Superconductivity | Unknown, but superheavy elements typically do not exhibit superconductivity at standard conditions |
| Reflectivity | Expected to be similar to that of other heavy metals, indicating good reflectivity |
| Absorption | Predicted to absorb significant amounts of specific electromagnetic wavelengths, typical for metals |
| Permeability | Similar to other group 8 elements, likely very low due to its expected metallic nature |
Nuclear Properties of Hassium
| Nuclear Property | Description |
|---|---|
| Half-life | Most stable isotope (Hs-277) has a half-life of about 11 minutes |
| Decay Modes | Alpha decay primarily, with possible spontaneous fission |
| Neutron Cross Section | Theoretical, expected to be small due to the element’s large nucleus |
| Isotopes | Several isotopes synthesized, with mass numbers from 263 to 277 |
| Nuclear Spin | Theoretical predictions vary, dependent on specific isotopes |
| Nuclear Magnetic Moment | Predicted based on theoretical calculations, not directly measured |
Preparation of Hassium
The preparation of Hassium (Hs), element 108, involves sophisticated nuclear reactions utilizing particle accelerators. Hassium does not occur naturally and can only be created in laboratory settings by bombarding target atoms of lighter elements with accelerated particles. The most common methods for preparing Hassium involve heavy ion fusion reactions, where a lighter, projectile nucleus is accelerated to high energies and then collided with a heavier target nucleus. The general approach aims to fuse these nuclei to form a heavier nucleus, corresponding to Hassium. Here’s how Hassium has been prepared:
Direct Fusion Reactions
- Lead-208 and Iron-58 Reaction: The primary method for producing Hassium involves bombarding lead (Pb) targets with iron (Fe) ions. The reaction between Lead-208 and Iron-58 has been successfully used to synthesize Hassium isotopes, notably Hassium-265. represents a neutron. This reaction has been instrumental in synthesizing Hassium for the first time and studying its properties.
- Other Potential Reactions: While the lead-iron reaction is the most notable, other combinations of target and projectile have been considered theoretically or attempted experimentally to produce different isotopes of Hassium. These include various combinations of heavier and lighter nuclei, but the efficiency and yield of Hassium isotopes in these reactions are generally lower than the lead-iron method.
Target and Projectile Selection
- Target Material: The choice of target material (like lead) is crucial as it must be capable of contributing the necessary neutrons to reach the desired atomic number and mass of Hassium.
- Projectile Ions: Iron ions are typically chosen as projectiles due to their ability to fuse with the target nucleus upon impact at high energies, facilitating the synthesis of Hassium.
Particle Accelerators
- Role of Accelerators: Particle accelerators are essential for the preparation of Hassium, as they accelerate the projectile ions to the required velocities. High kinetic energy is necessary for overcoming the electrostatic repulsion between the positively charged nuclei of the projectile and the target.
- Facilities: Facilities like the GSI Helmholtz Centre for Heavy Ion Research in Germany have played a pivotal role in the discovery and preparation of Hassium, utilizing their advanced particle accelerators.
Detection and Identification
After the bombardment, the resultant nucleus quickly undergoes a series of decay processes. The identification of Hassium and its isotopes is achieved through the detection of these decay chains, typically involving alpha decay and spontaneous fission. Sophisticated detectors and analytical techniques are employed to track these decay events, confirming the production of Hassium.
Challenges
The preparation of Hassium is highly challenging due to the need for precise control over reaction conditions, the extremely low production rates (often just a few atoms at a time), and the rapid decay of Hassium isotopes, which complicates the observation and study of its properties.
Chemical Compounds of Hassium
1. Hassium Oxide (HsO)
- Predicted Properties: Analogous to osmium tetroxide (OsO4), Hassium oxide is expected to exhibit strong oxidizing properties. It might have a tetrahedral molecular geometry.
- Hypothetical Formation Equation: <
- Potential Uses: While theoretical, HsO4 could be a powerful oxidizing agent similar to OsO4, with potential applications in organic chemistry for the oxidation of alkenes, if it were stable and could be handled safely.
2. Hassium Chloride (HsCl)
- Predicted Properties: This compound would likely be similar to osmium tetrachloride (OsCl4), with a possible tetrahedral structure for the HsCl molecule.
- Hypothetical Formation Equation:
- Potential Uses: In theory, HsCl4 could act as a starting material for the synthesis of other Hassium-containing compounds, much like its lighter homologs are used in transition metal chemistry.
3. Hassium Hexafluoride (HsF₆)
- Predicted Properties: Expected to be similar to osmium hexafluoride (OsF6), indicating strong fluorinating properties. Its stability and reactivity would follow trends observed in group 8 hexafluorides.
- Hypothetical Formation Equation: HsF₆
- Potential Uses: If it could be synthesized, HsF6 might be studied for insights into the behavior of superheavy element fluorides and their electronic structures.
4. Hassium Tetrabromide (HsBr)
- Predicted Properties: Analogous to its chlorides and fluorides, Hassium tetrabromide would likely be a volatile, possibly crystalline solid, exhibiting similar chemical behavior to osmium tetrabromide (OsBr4).
- Hypothetical Formation Equation:
- Potential Uses: Theoretical interest in HsBr4 would focus on its potential as a brominating agent and its electronic structure, providing insights into the relativistic effects on heavy element chemistry.
5. Hassium Hexacarbonyl (Hs(CO))
- Predicted Properties: Drawing parallels with other metal hexacarbonyls, Hassium hexacarbonyl could be expected to be a volatile, highly toxic compound with a complex electronic structure, showcasing Hassium’s ability to form stable complexes with carbon monoxide.
- Hypothetical Formation Equation: Hs(CO)₆
- Potential Uses: If it could be synthesized, Hassium hexacarbonyl might provide valuable data on the bonding and electronic properties of superheavy elements, contributing to theoretical models of chemical bonding.
6. Hassium Dihydride (HsH)
- Predicted Properties: This compound would theoretically resemble osmium dihydride (OsH2), suggesting it would be a reactive, possibly unstable dihydride, capable of participating in further chemical reactions.
- Hypothetical Formation Equation:
- Potential Uses: The study of Hassium dihydride could offer insights into the hydride chemistry of superheavy elements and their potential catalytic properties, albeit in a highly theoretical context.
Isotopes of Hassium
| Isotope | Half-Life | Decay Mode | Notes |
|---|---|---|---|
| Hs-263 | ~0.74 milliseconds | Alpha decay | One of the lighter isotopes, very short-lived. |
| Hs-264 | ~540 milliseconds | Alpha decay | Relatively short-lived, showcases alpha decay. |
| Hs-265 | ~2 milliseconds | Alpha decay | Offers insights into the nuclear structure. |
| Hs-266 | Data not available | Predicted alpha decay | Hypothetical isotope, not well-characterized. |
| Hs-267 | ~52 milliseconds | Alpha decay | Provides information on hassium’s alpha decay. |
| Hs-268 | ~1 second | Alpha decay, possibly spontaneous fission | Among the more stable isotopes, still short-lived. |
| Hs-269 | ~9.7 seconds | Alpha decay | Provides valuable data on heavy element decay. |
| Hs-270 | ~3.6 seconds | Alpha decay, possibly electron capture | Interesting for studies on decay processes. |
| Hs-271 | ~4 seconds | Alpha decay | Another isotope contributing to nuclear research. |
| Hs-272 | ~10 seconds | Alpha decay, spontaneous fission observed | Indicates a higher stability range. |
| Hs-277 | ~11 minutes | Alpha decay | The most stable known isotope of Hassium. |
Uses of Hassium
- Research in Nuclear Physics: Hassium’s primary use is in scientific research, particularly in nuclear physics, where it contributes to the understanding of the structure and properties of superheavy elements.
- Study of Atomic Nuclei: Hassium provides valuable insights into the behavior of atomic nuclei at the extreme end of the periodic table, helping scientists explore the limits of atomic stability.
- Investigation of the Island of Stability: The production and study of hassium isotopes are crucial in the search for the theorized “island of stability,” a region in the nuclear chart where superheavy elements might exhibit longer half-lives.
- Chemical Properties Research: Hassium’s chemical properties, though largely theoretical, are of interest to chemists studying the periodic trends and electron configurations in superheavy elements.
- Astrophysical Research: Although not directly observable in space due to its instability, studying elements like hassium can enhance our understanding of nucleosynthesis in extreme astrophysical environments.
- Development of Particle Accelerators: The synthesis of hassium requires highly sophisticated particle accelerators, and its study drives the development and refinement of these technologies.
- Educational Purposes: Hassium and its properties serve as educational tools in advanced chemistry and physics courses, highlighting the complexities of element synthesis and the periodic table’s boundaries.
- Quantum Chemistry Modeling: Theoretical studies on hassium can improve quantum chemical models, particularly those predicting the behavior of electrons in superheavy elements.
Production of Hassium
Hassium (Hs) is a synthetic element that is produced in particle accelerators through the fusion of smaller atomic nuclei. The production process involves highly sophisticated techniques and equipment designed to overcome the repulsive forces between atomic nuclei. Here are the key steps and methods used in the production of hassium:
- Selection of Target and Projectile: The production of hassium typically involves bombarding a target material made of a heavy element, such as lead (Pb) or bismuth (Bi), with a projectile composed of lighter atoms, like iron (Fe) or chromium (Cr). The choice of target and projectile is crucial for optimizing the likelihood of fusion reactions.
- Acceleration: The projectile ions are accelerated to extremely high velocities using a particle accelerator. This high kinetic energy is necessary to overcome the electrostatic repulsion between the positively charged nuclei of the target and the projectile.
- Collision and Fusion: When the accelerated ions collide with the target material, they can fuse to form a heavier nucleus if the kinetic energy is sufficient to bring them close enough for the nuclear strong force to take effect. This results in the formation of a compound nucleus that is highly excited and unstable.
- Evaporation of Neutrons: The compound nucleus loses energy by emitting several neutrons, a process known as neutron evaporation. This step is crucial for the nucleus to reach a lower energy state that is more stable.
- Formation of Hassium: After the emission of neutrons, the nucleus may settle into a state that corresponds to hassium, with its characteristic atomic number of 108. The specific isotope of hassium produced depends on the number of neutrons retained in the nucleus.
- Detection and Identification: The newly formed hassium atoms are then identified based on their decay patterns, which are unique to each element and its isotopes. The identification often involves sophisticated detectors that can observe the alpha particles and other radiation emitted as the hassium atoms decay.
Applications of Hassium
The applications of hassium are confined almost exclusively to the field of scientific research due to its extremely short half-life and the challenges associated with producing it in measurable quantities. Here are some of the research areas that benefit from the study of hassium:
- Nuclear Physics: Hassium plays a role in advancing the understanding of nuclear physics, particularly the properties and behaviors of superheavy elements.
- Elementary Particle Research: The production and study of hassium contribute to knowledge about elementary particles and the fundamental forces that govern atomic interactions.
- Chemical Research: Although practical chemical experiments with hassium are nearly impossible, theoretical studies based on its presumed properties can provide insights into the behavior of elements at the end of the periodic table.
- Search for the Island of Stability: Hassium’s synthesis is part of the broader search for the island of stability, an area in the chart of nuclides where superheavy elements might have significantly longer half-lives.
- Advancements in Particle Accelerator Technology: The need to create hassium and other superheavy elements drives the development of more powerful and precise particle accelerators.
- Astrophysics and Cosmology: Theoretical aspects of hassium’s production and decay can offer insights into processes occurring in supernovae and the formation of heavy elements in the universe.
Hassium, element 108, remains one of the most intriguing yet elusive members of the periodic table. With its theoretical properties and speculated compounds, Hassium pushes the boundaries of chemistry and physics, offering a glimpse into the behaviors of superheavy elements. Although its isotopes are short-lived and challenging to study, Hassium continues to captivate scientists, driving advancements in nuclear research and theoretical chemistry.