Niobium (Nb) – Definition, Preparation, Properties, Uses, Compounds, Reactivity
Dive into the fascinating world of Niobium, a remarkable element known for its unique properties and wide-ranging applications in modern technology and industry. This comprehensive guide offers an in-depth look at Niobium, from its fundamental characteristics to its role in enhancing the performance of superalloys and superconductors. With detailed examples, we unveil how Niobium’s versatility and strength contribute to advancements in aerospace, construction, and electronics, making it a cornerstone of innovative engineering solutions.
What is Niobium?
Niobium is a natural element found on the periodic table with the symbol “Nb” and atomic number 41. It’s a metal that is ductile and malleable, exhibiting a lustrous, greyish appearance. Niobium is crucial in various high-tech and engineering applications due to its unique properties, including its ability to enhance the strength of steel and its superconducting capabilities. This metal plays a significant role in the aerospace industry, in the manufacture of jet engines. Beyond its industrial uses, niobium’s resistance to corrosion and high melting point make it valuable in the creation of specialized alloys.
Niobium Formula
- Formula: Nb
- Composition: Consists of a single niobium atom.
- Bond Type: In its elemental form, niobium does not have bonds as it is a pure element.
However, niobium can form covalent or ionic bonds when reacting with other elements. - Molecular Structure: As a pure element, niobium does not form a molecular structure in the same sense as compounds. At room temperature, niobium is in a metallic state with a body-centered cubic crystalline structure.
- Electron Sharing: In compounds, niobium typically forms ionic bonds by transferring electrons to other elements, though it can also participate in covalent bonding, especially in complex niobium compounds.
- Significance: Niobium is crucial for its role in enhancing the properties of steel and superalloys, making them stronger and more resistant to heat and wear. It is also significant for its use in superconductors and electronic components.
- Role in Chemistry: Niobium plays a vital role in advanced materials engineering, including aerospace, structural applications, and electronics. It forms a variety of compounds, such as niobium carbide (NbC) and niobium pentoxide (Nb₂O₅), used in capacitors and optical lenses, making it an important material in a wide range of technological fields.
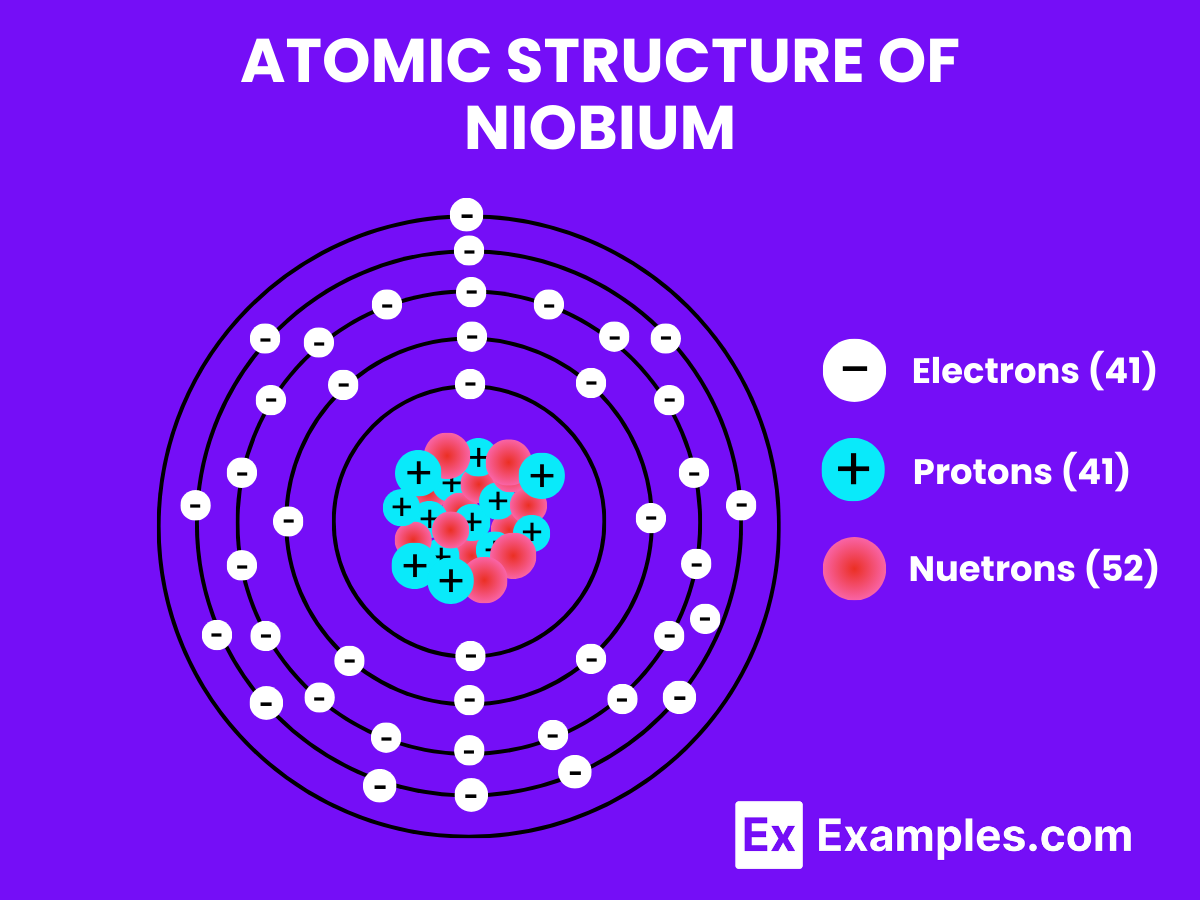
Atomic Structure of Niobium
Niobium, with the chemical symbol Nb and atomic number 41, is a transition metal known for its gray metallic appearance and notable physical and chemical properties. The atomic structure of niobium is characterized by its unique arrangement of electrons, protons, and neutrons:
- Protons and Neutrons: In its nucleus, niobium has 41 protons and typically 52 neutrons in its most abundant isotope, giving it a standard atomic weight of approximately 92.906 u.
- Electron Configuration: The electrons in niobium are arranged in shells around the nucleus. Its electron configuration is [Kr] 4d⁴5s¹. This configuration indicates that niobium has one electron in its outermost 5s orbital and four electrons in the 4d subshell, making it part of the transition metals group.
- Valence Electrons: The valence shell of niobium, which includes the electrons in the 4d and 5s orbitals, contains a total of five electrons. These valence electrons play a critical role in the chemical behavior of niobium, including its ability to form compounds and alloys.
- Crystal Structure: At room temperature, niobium crystallizes in a body-centered cubic (bcc) structure, which contributes to its strength and ductility. In this structure, each niobium atom is surrounded by eight other niobium atoms at the corners of a cube, with one niobium atom in the center.
The atomic structure of niobium underpins its extensive use in various applications, including steel production, superconductors, and electronic components, thanks to its excellent conductivity and resistance to corrosion
Properties of Niobium
Physical Properties of Niobium
Niobium, a chemical element with the symbol Nb and atomic number 41, is known for its remarkable physical and chemical properties. Below is a table detailing the key physical properties of niobium, showcasing why it’s an invaluable resource in various industrial and technological applications:
| Property | Value |
|---|---|
| Appearance | Silvery-gray metallic |
| Atomic Mass | 92.90638 u |
| Melting Point | 2,468°C (4,474°F) |
| Boiling Point | 4,927°C (8,901°F) |
| Density at 20°C | 8.57 g/cm³ |
| Crystal Structure | Body-centered cubic (BCC) |
| Electrical Resistivity | 152 nΩ·m (at 20°C) |
| Thermal Conductivity | 53.7 W/(m·K) |
| Thermal Expansion | 7.3 µm/(m·K) (at 25°C) |
| Young’s Modulus | 105 GPa |
| Shear Modulus | 38 GPa |
| Bulk Modulus | 170 GPa |
| Poisson’s Ratio | 0.40 |
| Vickers Hardness | 1320 MPa |
| Electrical Conductivity | 6.7 × 10⁶ S/m (at 20°C) |
Chemical Properties of Niobium
Niobium is a transition metal with distinct chemical properties that make it valuable in various industrial applications. Its chemical behavior is influenced by its electron configuration and position in the periodic table. Here are some of the key chemical properties of niobium:
- Corrosion Resistance: Niobium exhibits excellent resistance to corrosion due to the formation of a protective oxide layer on its surface.
This oxide layer effectively protects niobium from further reaction with oxygen, making it stable in air at room temperature. - Reactivity with Acids: While niobium is resistant to corrosion in air, it dissolves in hot, concentrated acids.
For example, niobium reacts with hydrofluoric acid (HF) to form niobium(V) fluoride (NbF₅).
Equation: - Formation of Oxides: Upon heating in air, niobium forms niobium pentoxide (Nb₂O₅), indicating its ability to form multiple oxidation states, predominantly +5.
Equation: - Alloying with Other Metals: Niobium readily forms alloys with many other metals, enhancing their strength and resistance to heat. This is particularly valuable in the aerospace and steel industries.
- Formation of Complexes: In various oxidation states, niobium forms complex compounds with other elements, including halogens and oxygen. For example, niobium forms complex halides such as NbCl₅ in its +5 oxidation state.
- Reactivity with Nonmetals: Niobium reacts with nonmetals at high temperatures. For instance, it reacts with nitrogen to form niobium nitride (NbN), which has superconducting properties.
Equation:
Thermodynamic Properties of Niobium
| Property | Value |
|---|---|
| Melting Point | 2,468°C (4,474°F) |
| Boiling Point | 4,927°C (8,901°F) |
| Heat of Fusion | 26.8 kJ/mol |
| Heat of Vaporization | 680 kJ/mol |
| Specific Heat Capacity | 24.60 J/(mol·K) |
Material Properties of Niobium
| Property | Value |
|---|---|
| Density | 8.57 g/cm³ at 20°C |
| Young’s Modulus | 105 GPa |
| Shear Modulus | 38 GPa |
| Bulk Modulus | 170 GPa |
| Mohs Hardness | 6 |
Electromagnetic Properties of Niobium
| Property | Value |
|---|---|
| Electrical Conductivity | 6.58 × 10⁶ S/m |
| Thermal Conductivity | 53.7 W/(m·K) |
| Magnetic Susceptibility | Superconducting below 9.2K |
Nuclear Properties of Niobium
| Property | Value |
|---|---|
| Natural Isotopes | ⁹³Nb (100% abundance) |
| Radioactive Isotopes | ⁹²Nb, ⁹⁴Nb (trace amounts) |
| Neutron Cross Section | 1.15 barns (for ⁹³Nb) |
| Neutron Mass Absorption | 1.1 × 10⁻³ |
Chemical Compounds of Niobium
- Niobium Pentoxide (Nb₂O₅): A white solid used in the production of niobium metal and its alloys, and as a catalyst in the petrochemical industry.
Equation: - Niobium Carbide (NbC): A hard, refractory ceramic material, used in tool bits for cutting and as an additive to improve alloy strength.
Equation: - Niobium Nitride (NbN): Exhibits superconducting properties, used in electronic devices and as a coating to enhance hardness.
Equation: - Niobium Sulfide (NbS₂): A layered compound with lubricating properties, studied for its potential in electronic applications.
Equation: - Niobium Fluoride (NbF₅): Used in the synthesis of organic niobium compounds and as a reagent in chemical research.
Equation: - Niobium Chloride (NbCl₅): A volatile compound used as a starting material for the synthesis of other niobium compounds and in chemical vapor deposition processes.
Equation:
Preparation of Niobium
The preparation of niobium involves several complex processes due to its strong affinity for oxygen and other elements. The primary source of niobium is from the minerals pyrochlore and columbite. The steps to extract and prepare pure niobium metal are outlined below:
- Ore Concentration: The first step involves crushing the niobium-containing minerals and then using flotation processes to concentrate the niobium ore.
- Extraction: The concentrated ore undergoes a series of chemical reactions. It is often processed by first converting it into niobium oxide (Nb₂O₅), which is then reacted with hydrofluoric acid to form niobium fluoride (NbF₅).
- Reduction: The niobium fluoride is then reduced to metallic niobium using either a thermal reduction process with sodium (Na) or by electrolysis in a molten salt solution. The thermal reduction process involves heating niobium fluoride with sodium at high temperatures, which results in the formation of niobium and sodium fluoride (NaF). Alternatively, the electrolytic reduction process in a molten salt bath can produce high-purity niobium.
- Purification: The crude niobium obtained from the reduction process contains impurities that must be removed to achieve the desired purity. This is typically done through a vacuum or inert gas purification process, where the metal is heated to high temperatures, causing the impurities to volatilize and separate from the niobium.
- Final Processing: The purified niobium is then melted and cast into ingots or processed further into various forms, such as sheets, rods, or wires, depending on its intended application.
Isotopes of Niobium
| Isotope | Natural Abundance | Half-life | Decay Mode |
|---|---|---|---|
| ⁹³Nb | 100% | Stable | — |
| ⁹⁰Nb | Trace | 14.6 hours | β⁻ decay |
| ⁹¹Nb | Trace | 680 years | ε decay |
| ⁹²Nb | Trace | 34.7 million years | β⁻ decay |
| ⁹⁴Nb | Trace | 20,300 years | β⁻ decay |
| ⁹⁵Nb | Synthetic | 35 days | β⁻ decay |
| ⁹⁶Nb | Synthetic | 23.35 hours | β⁻ decay |
| ⁹⁷Nb | Synthetic | 72 minutes | β⁻ decay |
| ⁹⁸Nb | Synthetic | 2 seconds | β⁻ decay |
| ⁹⁹Nb | Synthetic | 15 seconds | β⁻ decay |
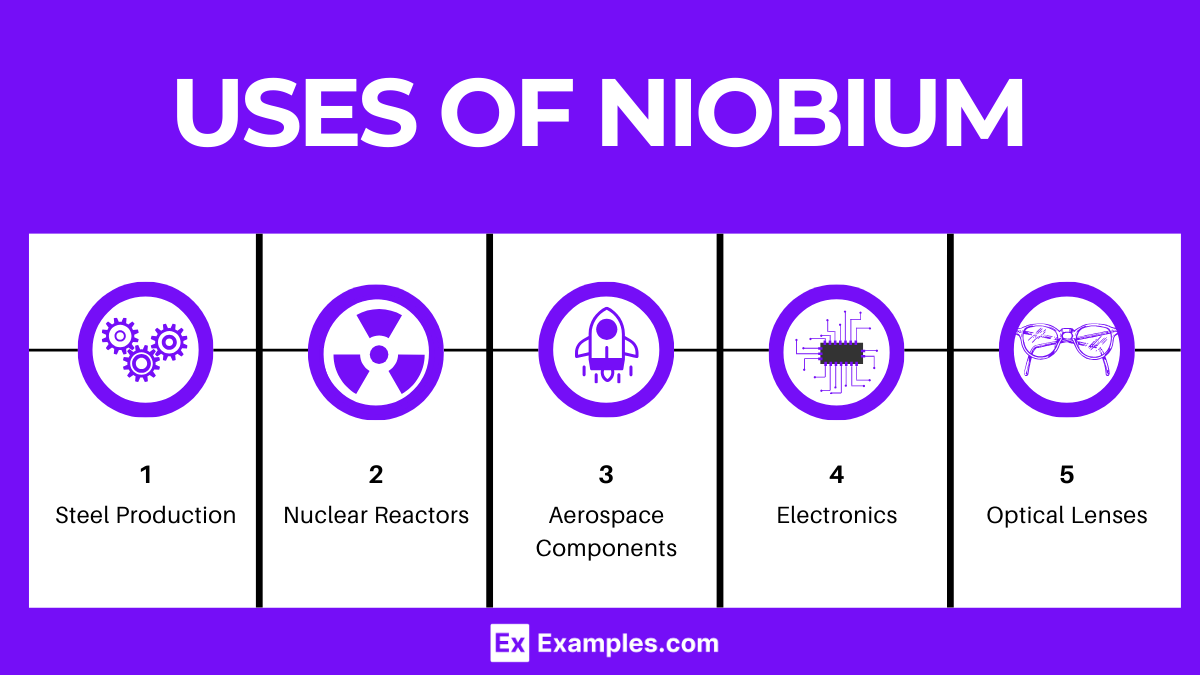
Uses of Niobium
Niobium serves a variety of critical roles across multiple industries due to its unique properties. Some of the primary uses of niobium include:
- Steel Production: Niobium is added to steel to improve its strength and weldability. Even small amounts can significantly enhance the strength of steel, making it ideal for use in structural applications like bridges and buildings.
- Superconducting Magnets: Niobium-tin (Nb₃Sn) and niobium-titanium (Nb-Ti) alloys are used to make superconducting wires for magnets. These magnets are crucial in MRI machines and scientific research equipment, such as particle accelerators.
- Aerospace Components: Due to its high melting point and strength, niobium alloys are used in jet engines and rocket subassemblies where high-temperature resistance is required.
- Electronics: Niobium is used in the manufacturing of electronic components, such as capacitors, due to its ability to form a stable oxide layer.
- Optical Lenses: Niobium oxide is used in the production of special glass for lenses, offering improved refractive properties.
- Nuclear Reactors: The high melting point and corrosion resistance of niobium make it suitable for use in the nuclear industry, especially in the construction of reactor components.
Production of Niobium
Niobium is a chemical element with the symbol Nb and atomic number 41. It is a soft, grey, crystalline, ductile transition metal that is often found in the minerals pyrochlore and columbite, with pyrochlore being the primary source of commercial niobium. The production of niobium involves several key steps:
- Mining: Niobium is primarily extracted from the mineral pyrochlore. The largest deposits are located in Brazil and Canada, which dominate the global production of niobium.
- Concentration: The mined ore is initially crushed and then subjected to a process to increase the niobium concentration. This usually involves flotation, magnetic separation, or gravity concentration techniques.
- Pyrometallurgical Process: The concentrated ore is then subjected to a pyrometallurgical process to remove impurities. This often involves smelting in an electric arc furnace, where the ore is mixed with carbon and heated. The carbon reduces the niobium oxide to niobium metal, and slag containing the impurities is removed.
- Hydrometallurgical Process: In some cases, a hydrometallurgical process might follow or replace the pyrometallurgical process. This involves leaching the niobium-containing material with a solution, typically hydrofluoric acid, to dissolve the niobium. The niobium is then precipitated from the solution, often as a niobium oxide.
- Refining: The crude niobium is further refined to achieve the desired purity. This can involve electron-beam melting, vacuum arc remelting, or other processes. The refining process removes impurities and produces high-purity niobium metal or niobium compounds, such as niobium pentoxide (Nb₂O₅).
- Alloy Production: Finally, niobium is often used in alloy form. It is combined with other metals, such as steel, to enhance their properties. The alloying process involves melting the niobium with the other metals and casting the mixture into ingots or other forms for further use.
Applications of Niobium
Niobium has a wide range of applications due to its unique properties, including its high melting point, resistance to corrosion, and ability to form superconducting compounds. Some of the primary applications of niobium include:
- Steel Production: Niobium is added to steel to improve its strength and welding properties. High-strength, low-alloy (HSLA) steels containing niobium are used in automotive, structural, and pipeline applications.
- Aerospace: Due to its high melting point and strength at elevated temperatures, niobium alloys are used in aircraft engines and rocket subassemblies.
- Superconductors: Niobium-tin (Nb₃Sn) and niobium-titanium (NbTi) alloys are used in superconducting magnets, which are essential for magnetic resonance imaging (MRI) machines and particle accelerators.
- Electronics: Niobium oxide is used in capacitors for electronic circuits due to its high dielectric constant, contributing to the miniaturization of electronic devices.
- Optics: Niobium coatings are applied to glass for computer screens and other devices to reduce glare and improve clarity.
- Chemical Industry: Niobium is resistant to acids and is used in the construction of chemical processing equipment.
- Jewelry: Due to its hypoallergenic properties and ability to be anodized to various colors, niobium is sometimes used in jewelry.
Niobium’s unique properties, including its high melting point, corrosion resistance, and ability to enhance the strength of steel alloys, make it indispensable in various high-tech and industrial applications. From enhancing the performance of jet engines and rockets to its critical role in superconducting materials, niobium’s contributions to modern technology and engineering are invaluable. Its ongoing research and development promise to unlock even more potential uses, solidifying its place as a key element in advancing innovation.