Titanium (Ti) – Definition, Preparation, Properties, Uses, Compounds, Reactivity
Titanium, a symbol of strength and resilience, stands out in the periodic table for its exceptional properties. This guide embarks on a journey through the world of titanium, showcasing its pivotal role in industries ranging from aerospace to medical implants. With its remarkable strength-to-weight ratio and corrosion resistance, titanium’s applications are as diverse as they are groundbreaking. Through detailed examples, we’ll uncover how this lightweight metal is revolutionizing technology and construction, making the impossible possible. Join us in exploring the multifaceted applications and innovative uses of titanium, a metal that continues to shape the future.
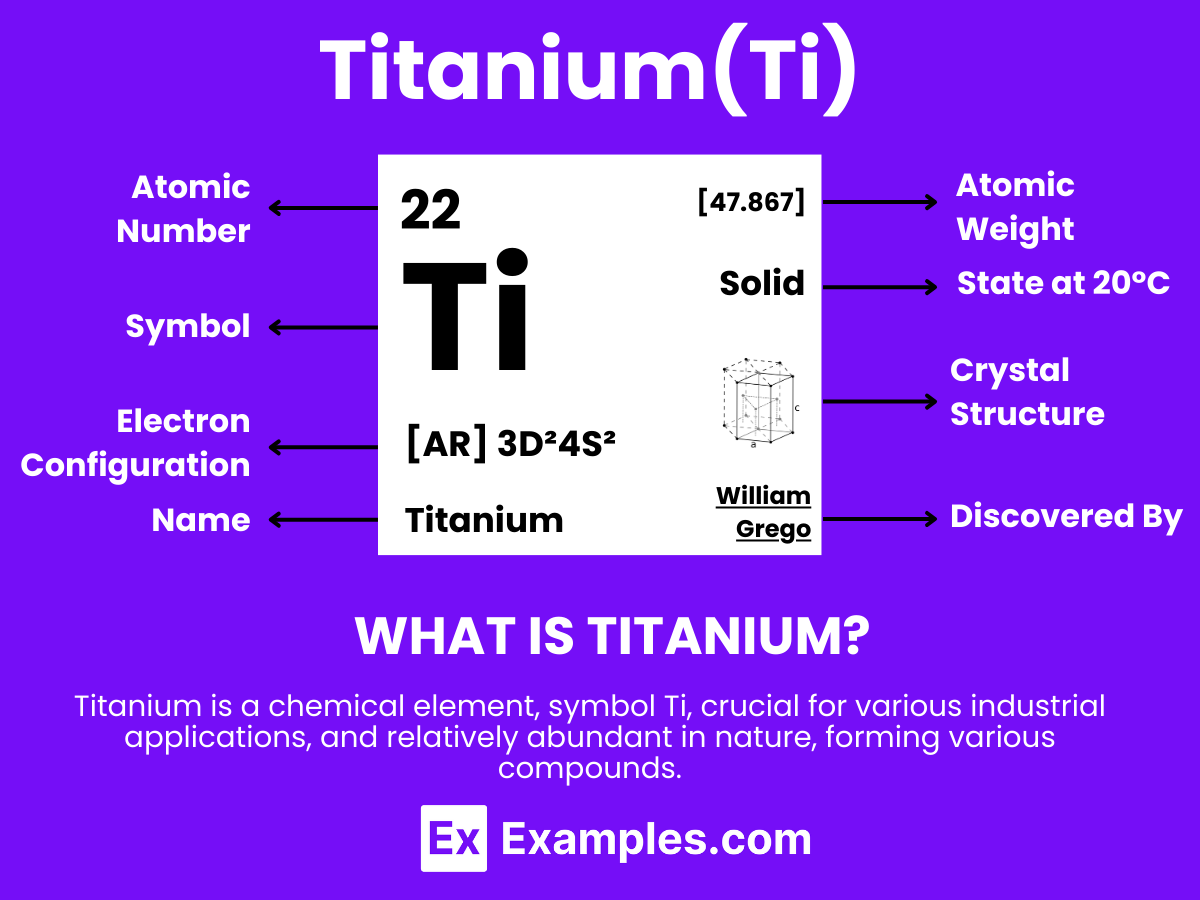
What is titanium?
Titanium is a strong, lightweight, silvery-white metallic element known for its outstanding properties and versatility across numerous applications. With the atomic number 22. Titanium is celebrated for its exceptional strength-to-weight ratio and superior resistance to corrosion, including against seawater and chlorine. This element is not found freely in nature but is primarily extracted from rutile and ilmenite ores along with other metals. Titanium’s widespread use spans various fields, notably in the aerospace industry for aircraft, spacecraft, and missiles due to its durability and lightweight properties. Additionally, it’s used in the medical sector for surgical implants and prosthetics, in the sports industry for high-performance equipment, and in architecture for its aesthetic appeal and longevity
Titanium Formula
- Formula: Ti
- Composition: Consists of a single titanium atom.
- Bond Type: In its elemental form, titanium does not form bonds as it is a pure element. However, titanium can form both covalent and ionic bonds when reacting with other elements.
- Molecular Structure: As a pure element, titanium does not form a molecular structure like compounds (e.g., water). At room temperature, titanium exists in a metallic state with a hexagonal close-packed (HCP) crystalline structure, known as the alpha (α) phase. It transitions to a body-centered cubic (BCC) structure, known as the beta (β) phase, at higher temperatures.
- Electron Sharing: In compounds, titanium typically shares electrons covalently or transfers electrons ionically, depending on the nature of the other element(s) it is bonding with.
- Significance: Titanium is renowned for its strength-to-density ratio, being one of the strongest metals per unit mass. It is also highly resistant to corrosion in sea water, aqua regia, and chlorine.
- Role in Chemistry: Titanium’s resistance to corrosion and high strength make it invaluable in various applications, including aerospace, military, industrial processes, automotive, medical devices, and sports equipment. Titanium dioxide (TiO₂) is extensively used as a white pigment in paints, sunscreens, and cosmetics. Titanium is also used in the production of strong and lightweight alloys for aerospace and other high-performance applications.
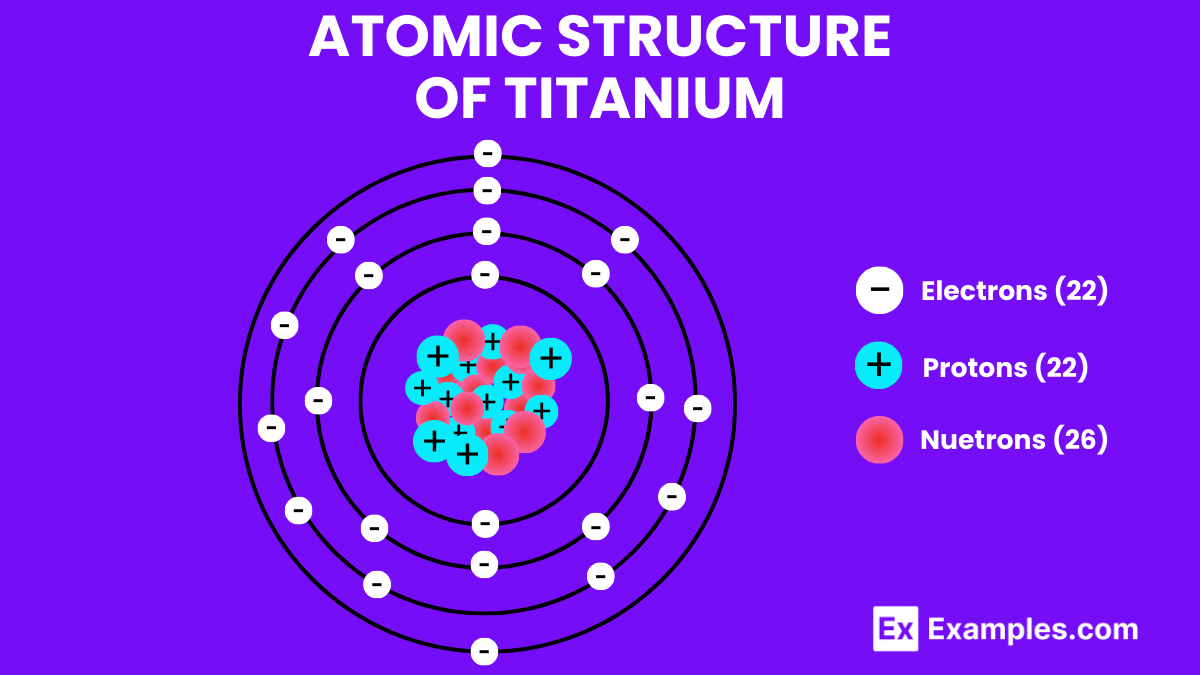
Atomic Structure of Titanium
The atomic structure of Titanium is a cornerstone in understanding this versatile element’s exceptional characteristics, including its strength, corrosion resistance, and lightweight properties. Titanium, symbolized as Ti and with an atomic number of 22, stands out in the periodic table as a transition metal known for its widespread application across various industries. Here’s a detailed exploration of Titanium’s atomic structure:
Protons, Neutrons, and Electrons
- Protons: Titanium has 22 protons in its nucleus, a defining feature that determines its chemical identity and position in the periodic table.
- Neutrons: The most abundant isotope of Titanium, Titanium-48, contains 26 neutrons. However, Titanium has five naturally occurring isotopes with neutron numbers ranging from 24 to 28, influencing the atomic mass but not its chemical behavior.
- Electrons: Titanium has 22 electrons, with its electron configuration being [Ar] 3d²4s². This configuration indicates two electrons in the outermost shell and two electrons in the 3d orbital.
Properties of Titanium
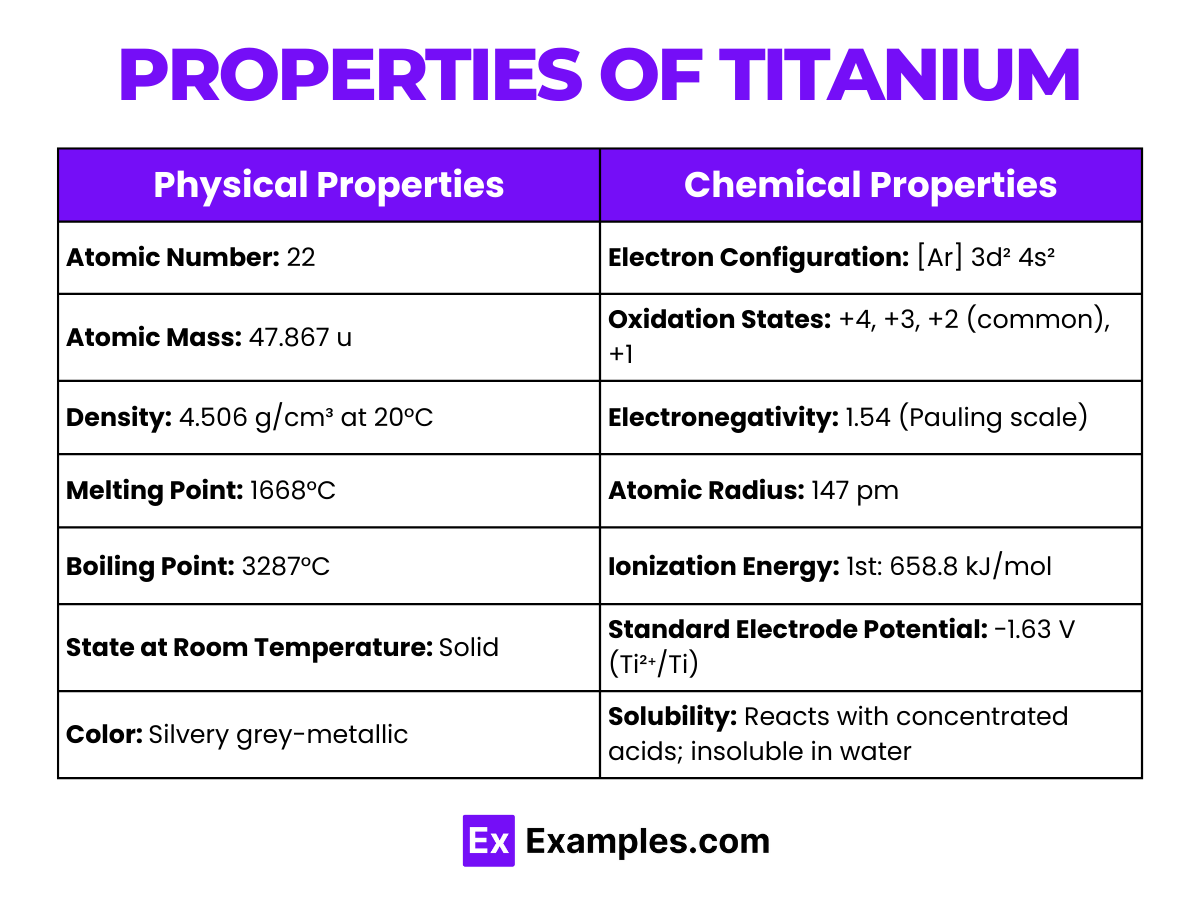
Physical Properties of Titanium
| Property | Value |
|---|---|
| Appearance | Silvery-grey, metallic |
| Atomic Number | 22 |
| Atomic Weight | 47.867 |
| Density | 4.506 g/cm³ at 20 °C |
| Melting Point | 1,668 °C (3,034 °F) |
| Boiling Point | 3,287 °C (5,949 °F) |
| State at 20 °C | Solid |
| Electrical Resistivity | 420 nanoohm-meters at 20 °C |
| Thermal Conductivity | 21.9 W/(m·K) at 300 K |
| Heat of Fusion | 14.15 kJ/mol |
| Heat of Vaporization | 425 kJ/mol |
| Specific Heat Capacity | 0.523 J/(g·K) |
| Electronegativity | 1.54 (Pauling scale) |
| Crystal Structure | Hexagonal close-packed (hcp) |
| Magnetic Ordering | Paramagnetic |
Chemical Properties of Titanium
Titanium, with its symbol Ti and atomic number 22, is a transition metal known for its strength, low density, and high corrosion resistance. This section delves into the chemical properties of titanium, highlighting its reactivity, common reactions, and significant compounds.
Reactivity: Titanium is less reactive than many metals due to the formation of a passive oxide layer on its surface, which protects it from further corrosion. However, it can react with various elements under certain conditions.
Equation with Oxygen:
Titanium burns in air to form titanium dioxide (TiO₂), especially at high temperatures, showcasing its ability to bond with oxygen.
Reaction with Acids: Titanium reacts with concentrated acids, illustrating its reactivity with non-oxidizing acids to form titanium(III) chloride (TiCl₃) and hydrogen gas.
Equation with Hydrochloric Acid:
This reaction demonstrates titanium’s ability to dissolve in strong acids, forming tetravalent titanium compounds.
Reaction with Halogens: Titanium reacts vigorously with halogens to form titanium halides, indicating its high reactivity with these elements.
Equation with Chlorine: Ti+2Cl₂→TiCl₄
Titanium tetrachloride (TiCl₄) is a significant compound used in the production of titanium metal and titanium dioxide.
Formation of Alloys: Titanium forms alloys with many other metals, such as aluminum, vanadium, and molybdenum. These alloys are stronger and lighter than pure titanium, making them invaluable in aerospace, military, and sporting goods.
Role as a Catalyst: Titanium compounds, especially titanium dioxide (TiO₂), act as photocatalysts under UV light. This property is exploited in self-cleaning surfaces and in the breakdown of pollutants.
Thermodynamic Properties of Titanium
| Property | Value |
|---|---|
| Melting Point | 1668°C (3034°F) |
| Boiling Point | 3287°C (5949°F) |
| Heat of Fusion | 14.15 kJ/mol |
| Heat of Vaporization | 425 kJ/mol |
| Specific Heat Capacity | 25.060 J/(mol·K) |
| Thermal Conductivity | 21.9 W/(m·K) |
| Thermal Expansion | 8.6 µm/(m·K) (at 25°C) |
Material Properties of Titanium
| Property | Value |
|---|---|
| Atomic Mass | 47.867 u |
| Density | 4.506 g/cm³ (at 20°C) |
| Young’s Modulus | 116 GPa |
| Shear Modulus | 44 GPa |
| Bulk Modulus | 110 GPa |
| Poisson’s Ratio | 0.32 |
| Mohs Hardness | 6 |
| Brinell Hardness | 716 MPa |
Electromagnetic Properties of Titanium
| Property | Value |
|---|---|
| Electrical Resistivity | 420 nΩ·m (at 20°C) |
| Magnetic Ordering | Paramagnetic at 300 K |
| Magnetic Susceptibility | +153·10⁻⁶ cm³/mol (at room temperature) |
| Superconducting Point | Below 0.4 K (not naturally occurring) |
Nuclear Properties of Titanium
| Property | Value |
|---|---|
| Atomic Number | 22 |
| Atomic Weight | 47.867 u |
| Isotopes | Ti-46, Ti-47, Ti-48, Ti-49, Ti-50 |
| Radioactive Isotopes | Ti-44, Ti-45, Ti-51, etc. |
| Half-Lives | Varies from fractions of a second to years for radioactive isotopes |
| Neutron Cross Section | 6.1 barns (for Ti-48) |
| Neutron Mass Absorption | 0.003 (for Ti-48) |
Preparation of Titanium
The preparation of titanium is a complex, yet fascinating process that underscores the metal’s value across various high-tech and industrial applications. Titanium is primarily extracted from its most common ores, rutile (TiO₂) and ilmenite (FeTiO₃), through the Kroll process, a method that has stood the test of time for producing high-purity titanium.
- Ore Processing: Initially, the titanium-containing ore is purified to produce titanium dioxide, usually by using the chloride process or the sulfate process.
- Chlorination: The purified titanium dioxide is then reacted with chlorine gas and carbon (coke) at high temperatures in a fluidized bed reactor, producing titanium tetrachloride (TiCl₄ ), a volatile liquid.
- Purification of TiCl₄: The titanium tetrachloride is distilled to remove impurities, ensuring the subsequent reaction produces titanium of high purity.
- Reduction via the Kroll Process: In the core step known as the Kroll process, the purified titanium tetrachloride is mixed with liquid magnesium in a large, sealed reactor. The mixture is heated, and the titanium tetrachloride reacts with the magnesium to produce metallic titanium and magnesium chloride.
- Vacuum Distillation: The resultant mixture is subjected to vacuum distillation to remove the magnesium chloride, leaving behind sponge-like porous titanium.
- Melting and Alloying: Finally, the titanium sponge is melted in a vacuum arc furnace or through electron beam melting, often alloyed with other metals to enhance its properties, and then cast into ingots, bars, or other desired forms
Chemical Compounds of Titanium
- Titanium Dioxide (TiO₂)
- Description: White pigment used in paints, sunscreens.
- Equation: Ti + O₂ → TiO₂
- Titanium Tetrachloride (TiCl₄ )
- Description: Colorless liquid, used in smoke screens, polymer production.
- Equation: Ti + 2Cl₂ → TiCl₄
- Titanium Trichloride (TiCl₃)
- Description: Used in polymer catalysts, organic synthesis.
- Equation: 2Ti + 3Cl₂ → 2TiCl₃
- Titanium Nitride (TiN)
- Description: Hard ceramic material, coating for cutting tools.
- Equation: Ti + 1/2N₂ → TiN
- Titanium Carbide (TiC)
- Description: Very hard material, used in tool bits.
- Equation: Ti + C → TiC
- Titanium Sulfate (Ti(SO4)₂)
- Description: Used in dyeing and as a catalyst.
- Equation: Ti + 2H₄SO₄ → Ti(SO₄ )₂ + 2H₂
Isotopes of Titanium
Titanium is composed of several isotopes, ranging from those that are stable and naturally occurring to those that are radioactive and synthesized in laboratories. Here is a table that highlights the key isotopes of titanium, including their atomic masses, half-lives, and decay modes when applicable.
| Isotope | Atomic Mass | Half-Life | Mode of Decay |
|---|---|---|---|
| Ti-46 | 46 | Stable | N/A |
| Ti-47 | 47 | Stable | N/A |
| Ti-48 | 48 | Stable | N/A |
| Ti-49 | 49 | Stable | N/A |
| Ti-50 | 50 | Stable | N/A |
| Ti-44 | 44 | 60 years | Electron Capture to Sc-44 |
| Ti-45 | 45 | 184.8 minutes | Beta decay to Sc-45 |
| Ti-51 | 51 | 5.76 minutes | Beta decay to V-51 |
The stable isotopes, particularly Ti-48, make up a significant portion of naturally occurring titanium. The radioactive isotopes, such as Ti-44, have applications in medical imaging and scientific research due to their properties.
Uses of Titanium
Titanium is celebrated for its high strength-to-weight ratio, corrosion resistance, and biocompatibility. These properties make it invaluable across various industries and applications.
Aerospace and Aviation
- Airframes and Engine Components: Titanium’s strength, lightweight, and resistance to high temperatures make it ideal for critical aerospace components, including airframes, engines, and landing gear.
Medical Devices
- Implants and Prosthetics: Due to its biocompatibility and ability to withstand bodily fluids without corroding, titanium is widely used in surgical implants, prosthetic devices, and dental implants.
Sports Equipment
- High-performance Gear: Titanium is used in the manufacturing of golf clubs, bicycles, racquets, and other sports equipment, offering durability and lightweight advantage.
Chemical Processing
- Corrosion-resistant Equipment: Titanium’s resistance to corrosion makes it suitable for chemical processing equipment, such as reactors, pipes, and heat exchangers, especially when handling chlorides.
Consumer Products
- Jewelry and Watches: Its hypoallergenic properties and appealing metallic luster make titanium a popular choice for jewelry and high-end watches.
Construction and Architecture
- Cladding and Structural Components: Titanium is used in architecture for its aesthetic appeal and longevity, particularly in cladding, roofing, and structural components exposed to harsh environments.
Marine Applications
- Shipbuilding and Offshore Structures: The metal’s resistance to seawater corrosion makes it ideal for shipbuilding, desalination plants, and offshore oil and gas exploration equipment.
Automotive Industry
- High-performance Vehicle Parts: Titanium is used in the automotive industry for performance parts, including exhaust systems and suspension components, to reduce weight and improve fuel efficiency.
Production of Titanium
The production of Titanium is a sophisticated process that involves several stages, from extraction to refinement, making it a valuable material for various applications. The key steps in the production of Titanium include:
- Extraction of Titanium Dioxide: Titanium is primarily obtained from ilmenite and rutile, minerals rich in titanium dioxide (TiO₂). These minerals are mined from the Earth’s crust.
- Purification of Titanium Dioxide: The extracted titanium dioxide is purified through processes such as the sulfate process or the chloride process, depending on the desired purity level and the specific application of the Titanium.
- Conversion to Titanium Tetrachloride: The purified titanium dioxide is converted into titanium tetrachloride (TiCl₄ ) by reacting with chlorine and carbon at high temperatures.
- Reduction to Titanium Metal: Titanium tetrachloride is then reduced to metallic Titanium using magnesium in the Kroll process, the most common method of Titanium production. In this reaction, Titanium separates from the chloride and forms a sponge that can be further processed.
- Melting and Alloying: The Titanium sponge is melted in a vacuum or argon environment and can be alloyed with other metals to produce various grades of titanium alloys, enhancing its material properties for specific uses.
The meticulous production process contributes to the high value and cost of Titanium, reflecting its significance in high-performance and critical applications.
Applications of Titanium
Titanium’s remarkable properties, such as its high strength-to-weight ratio, corrosion resistance, and biocompatibility, have led to its widespread use across numerous industries:
- Aerospace Industry: Titanium and its alloys are extensively used in aircraft, spacecraft, and missiles due to their strength, low density, and ability to withstand extreme temperatures.
- Medical Devices: Titanium’s biocompatibility makes it ideal for surgical implants, including hip replacements, dental implants, and pacemakers.
- Automotive Industry: High-performance automotive parts, such as connecting rods, valves, and exhaust systems, are made from Titanium to reduce weight and improve efficiency.
- Construction and Architecture: Titanium’s corrosion resistance is beneficial in constructing durable structures, particularly in corrosive environments like coastal areas.
- Sports Equipment: Lightweight and strong, Titanium is used in manufacturing bicycles, golf clubs, and tennis rackets, enhancing performance and durability.
- Jewelry and Watches: Its hypoallergenic properties and appealing metallic luster make Titanium popular in jewelry making and watch casings.
- Chemical Processing: Titanium’s resistance to corrosion is advantageous in chemical processing equipment, such as reactors, heat exchangers, and pipes, especially when handling corrosive substances.
This article has illuminated the exceptional world of titanium, unraveling its vital physical and chemical properties through a concise table. We’ve journeyed from the meticulous preparation processes to its myriad applications, underscoring titanium’s integral role in advancing modern technology and engineering. Titanium’s unparalleled attributes not only define its utility but also forecast a future where its potential is boundlessly explored and utilized.