Yttrium (Y) – Definition, Preparation, Properties, Uses, Compounds, Reactivity
Yttrium, a key player in the world of rare earth metals, stands out for its significant contributions to various high-tech and medical applications. This comprehensive guide delves into the essence of Yttrium, exploring its unique properties, diverse uses, and the role it plays in enhancing modern technology and healthcare solutions. From its application in LED screens to its pivotal role in cancer treatments, Yttrium’s versatility and importance cannot be overstated. Discover how Yttrium powers innovation and improves daily life through its remarkable compounds and applications.
What is Yttrium?
Yttrium is a chemical element with the symbol Y and atomic number 39. It’s a silvery-metallic transition metal that is part of the rare earth group of elements. Despite being less familiar than other metals, yttrium is crucial in modern technology. It’s used in making phosphors for color TV screens, LEDs, and other light-emitting devices. Yttrium is also found in the production of superconductors and in various medical applications, including cancer treatment. This element is not found in its pure form in nature but in mineral compounds such as yttrite and monazite.
Yttrium Formula
- Formula: Y
- Composition: Consists of single yttrium atoms.
- Bond Type: As a pure element, yttrium does not form bonds with itself in the same way as diatomic molecules; in compounds, it typically forms ionic or covalent bonds.
- Molecular Structure: Not applicable as yttrium is not molecular but exists as individual atoms in its elemental form. In its solid form, yttrium adopts a crystalline structure.
- Electron Sharing: In chemical compounds, yttrium can donate or share electrons with other elements, depending on the type of chemical bond formed.
- Significance: Yttrium is important in various high-tech applications, including LEDs, phosphors, and superconductors. It is also used in the production of certain alloys and in medical technologies.
- Role in Chemistry: Yttrium serves as a critical component in many advanced materials and is often used to improve the properties of alloys and ceramics. Its compounds are utilized in a wide range of applications from electronics to cancer treatment.
Structure of Yttrium Gas
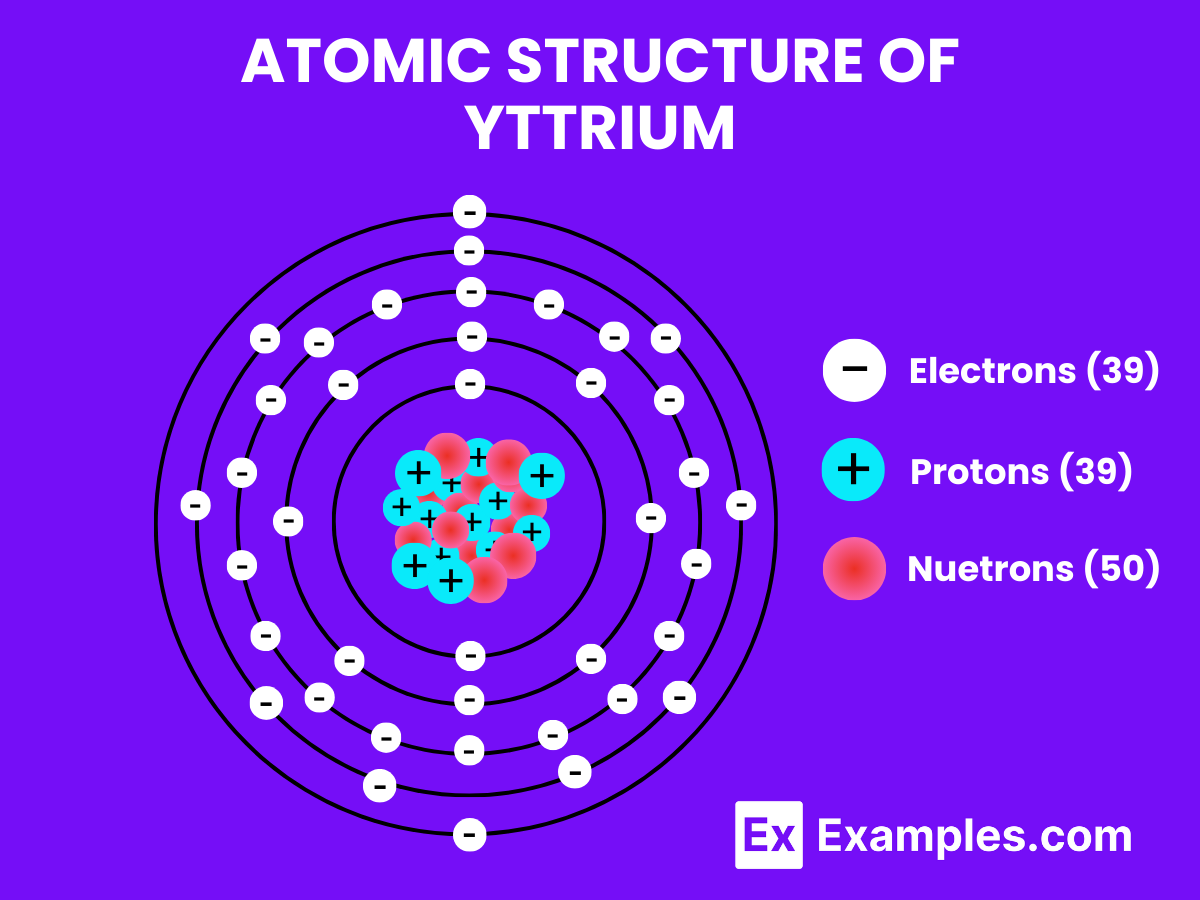
Yttrium, symbolized as Y, is a chemical element with one proton in its nucleus, positioned in the transition metals category of the periodic table. Each yttrium atom has 39 protons in its nucleus and typically 39 electrons, as its atomic number is 39. Yttrium does not form diatomic molecules like hydrogen under normal conditions. Instead, yttrium is more commonly found in various compounds and in solid form due to its metallic nature.
Atomic Level: Each yttrium atom (Y) consists of 39 protons in its nucleus surrounded by 39 electrons.
Molecular Formation: Unlike hydrogen, yttrium does not naturally pair up with another yttrium atom to form a diatomic molecule. In its elemental form, yttrium adopts a crystalline structure, specifically a hexagonal close-packed (hcp) arrangement, which is typical for many transition metals.
Yttrium is known for its use in various applications, including electronics, due to its interesting properties such as enhancing phosphorescence in television screens and LEDs. It is not found in its pure metallic state in nature but usually in the form of minerals like yttrite and xenotime. Yttrium’s compounds, rather than yttrium gas or diatomic molecules, are utilized in industrial and technological applications.
Properties of Yttrium
Physical Properties of Yttrium
| Property | Description |
|---|---|
| Appearance | Silvery-white and metallic |
| Atomic Number | 39 |
| Atomic Weight | 88.90585 |
| Melting Point | Approximately 1,522°C (2,772°F) |
| Boiling Point | Approximately 3,338°C (6,040°F) |
| Density | 4.472 grams per cubic centimeter at 20°C (solid phase) |
| State at Room Temperature | Solid |
| Electron Configuration | [Kr] 4d¹ 5s² |
| Crystal Structure | Hexagonal close-packed (hcp) at room temperature |
| Thermal Conductivity | 17.2 W/(m·K) |
| Electrical Resistivity | About 596 nΩ·m at 20°C |
| Electronegativity | 1.22 (Pauling scale) |
| Standard Atomic Weight | 88.90585 |
| Phase at STP | Solid |
| Solubility | Insoluble in water, reacts with acids |
| Common Oxidation States | +3 |
Chemical Properties of Yttrium
Reactivity with Air: Yttrium is fairly reactive in air; it forms a protective oxide layer that prevents further corrosion. Upon heating, it reacts with oxygen to form yttrium oxide (Y₂O₃).
Equation: 4Y+ 3O₂→2Y ₂O₃
Reactivity with Water: Yttrium reacts with water, especially at elevated temperatures, to form yttrium hydroxide H₂).
Equation: 2Y+6H ₂O→2Y(OH)₃+3H₂
Reactivity with Acids: Yttrium dissolves readily in dilute acids, forming solutions of yttrium ions (Y₃+) and hydrogen gas.
Equation: 2Y+6HCl→2YCl₃+3H₂
Reactivity with Halogens: Yttrium reacts with all halogens to form yttrium halides. For example, with chlorine, it forms yttrium chloride (3YCl₃).
Equation: 2Y+3Cl₂→2YCl₃
Formation of Alloys: Yttrium forms alloys with a wide range of metals. These alloys often have enhanced physical properties, such as increased strength and reduced grain size in high-temperature applications.
Complex Formation: Yttrium can form complex ions with various ligands. For example, it forms complexes with fluoride ions, such as YF₆₃, in solution.
Catalytic Properties: Yttrium exhibits catalytic properties in various chemical reactions, including polymerization and the dehydrogenation of hydrocarbons.
Thermodynamic Properties of Yttrium
| Property | Value with Unit |
|---|---|
| Boiling Point | 3,345 °C |
| Melting Point | 1,522 °C |
| Critical Temperature | Not Available |
| Critical Pressure | Not Available |
| Heat of Vaporization | 365 kJ/mol |
| Heat of Fusion | 11.4 kJ/mol |
| Specific Heat Capacity (at 25°C) | 0.298 J/g·K |
| Thermal Conductivity | 17 W/m·K |
Material Properties of Yttrium
| Property | Value with Unit |
|---|---|
| Density (at room temperature) | 4,472 kg/m³ |
| Viscosity | Not Applicable (Solid) |
| Solubility | Insoluble in water |
| Color | Silver-metallic |
| Phase at Room Temperature | Solid |
Electromagnetic Properties of Yttrium
| Property | Value with Unit |
|---|---|
| Electrical Resistivity (at 20°C) | 596 nΩ·m |
| Thermal Conductivity | 17 W/m·K |
| Magnetic Susceptibility | +1.2 × 10^-3 cm^3/mol |
| Electronegativity (Pauling scale) | 1.22 |
Nuclear Properties of Yttrium
| Property | Value with Unit |
|---|---|
| Atomic Number | 39 |
| Atomic Mass | 88.90584 amu |
| Isotopes | ^89Y (100%) |
| Nuclear Spin (for ^89Y) | 1/2 ℏ |
| Neutron Cross Section (for ^89Y) | 1.28 barns |
| Nuclear Magnetic Moment (for ^89Y) | -0.1373 µN |
Preparation of Yttrium
The preparation of yttrium metal typically involves several steps due to its occurrence in mixed rare earth minerals rather than in a pure form.
1. Extraction from Minerals
Yttrium is most commonly extracted from minerals such as monazite and xenotime, which contain yttrium as well as other rare earth elements. The extraction process begins with the mining of these minerals.
2. Mineral Processing
The mined minerals are crushed and then subjected to various physical and chemical processes to increase the concentration of yttrium and separate it from other elements. This often involves the use of solvent extraction or ion exchange techniques.
3. Conversion to Yttrium Oxide
The concentrated yttrium is usually converted into yttrium oxide (3Y₂O₃) through a calcination process, where the yttrium-containing compounds are heated to decompose them into their oxide forms.
4. Metallic Yttrium Production
The metallic yttrium is then produced from yttrium oxide using one of several methods:
a. Electrolysis :In this method, yttrium oxide is dissolved in a suitable flux, and then electrolysis is performed to reduce the yttrium ions to metallic yttrium. The electrolysis process typically requires high temperatures.
b. Metallothermic Reduction: Another common method is the metallothermic reduction, where yttrium oxide is reduced to metallic yttrium using a more reactive metal as the reducing agent. Calcium or magnesium is often used for this purpose due to their strong reducing capabilities.
Y₂O3+3Ca→2Y+3CaO
The reaction is carried out in an inert atmosphere to prevent oxidation and contamination.
5. Refining
The yttrium metal produced through electrolysis or metallothermic reduction often contains impurities. It is further refined using techniques such as vacuum distillation or zone melting to obtain high-purity yttrium metal.
6. Alloy Production (Optional)
Yttrium is often used in alloyed form to improve the properties of other metals. The refined yttrium can be alloyed with other elements depending on the desired application and material properties.
Chemical Compounds of Yttrium

Yttrium, a Group 3 transition metal, forms a variety of chemical compounds that are used across multiple industrial and technological fields. Its compounds are known for their unique chemical and physical properties.
1. Yttrium Oxide (Y₂O₃)
Yttrium oxide is one of the most important yttrium compounds, widely used in ceramics, phosphors, and laser materials due to its high thermal stability and strong luminescence when doped with rare-earth elements.
Formation Equation: 2Y+23O₂→Y₂O₃
2. Yttrium Aluminum Garnet (YAG; Y₃Al₅O₁₂)
YAG is a synthetic crystalline material used as a substrate in laser technology and in the production of various optical components. It serves as a host lattice for doping with different rare-earth ions to produce lasers of various wavelengths.
Synthetic Route: Yttrium oxide reacts with aluminum oxide in a high-temperature furnace to form YAG. 3Y₂O ₃+5Al2O₃→2Y₃Al₅O₁₂
3. Yttrium Iron Garnet (YIG; Y₃Fe₅O₁₂)
YIG is utilized in microwave filters and is essential in radar and communication devices due to its unique magnetic properties.
Synthetic Route: Similar to YAG, YIG is synthesized by reacting yttrium oxide with iron(III) oxide.3Y2O₃+5Fe₂O₃→2Y₃Fe₅O₂
4. Yttrium Barium Copper Oxide (YBa₂Cu₃O₇)
This compound is known for its high-temperature superconductivity properties. It was one of the first materials discovered to exhibit superconductivity above the boiling point of liquid nitrogen, making it practical for various applications.
General Reaction: The synthesis involves solid-state reactions between yttrium oxide, barium carbonate, and copper oxide.Y₂O₃+4BaCO₃+6CuO→2YBa₂Cu₃O₇−δ+4CO₂
5. Yttrium Fluoride (YF₃)
Yttrium fluoride is used in the production of metallic yttrium, fluoride glasses, and in coatings for optical fibers. It is also a component in some dental varnishes for its fluoride-releasing properties.
Formation Equation: Y+2₃F₂→YF₃
6. Yttrium Hydroxide (Y(OH)₃)
This compound is an intermediate in the conversion of yttrium ores into more useful compounds. It can act as a precursor to other yttrium compounds when reacted with appropriate acids or bases.
Formation in Aqueous Solution: Y₃⁺+3OH⁻→Y(OH)₃
These yttrium compounds play crucial roles in modern technology, from improving the efficiency of lasers and LEDs to enabling high-speed communication and groundbreaking medical devices. Their synthesis and applications underscore the importance of yttrium in the advancement of materials science and engineering.
Isotopes of Yttrium
Yttrium naturally occurs as a single isotope, 8989Y is of particular interest for medical applications due to its beta-emitting properties. Here are some details about key yttrium isotopes:
- 8989Y: The only stable and naturally occurring isotope of yttrium.
- 9090Y: A radioactive isotope used in medicine, particularly for cancer treatment in radiotherapy. It decays to zirconium-90, emitting beta radiation.
Uses of Yttrium
Yttrium has a variety of applications, many of which take advantage of its optical and electronic properties. Here are some of the main uses of yttrium:
- Electronics and Lighting: Yttrium is used in the production of red phosphors for LED lights and color television screens. Yttrium oxide is added to the phosphors to enhance brightness and efficiency.
- Superconductors: Yttrium barium copper oxide (7YBa₂Cu₃O₇) is a high-temperature superconductor used in magnetic levitation, magnetic resonance imaging (MRI), and in superconducting wires for power transmission.
- Ceramics: Yttrium stabilizes the zirconia in ceramics, making them stronger and more resistant to thermal shock. This is crucial for cutting tools, dental implants, and protective coatings.
- Metallurgy: Yttrium is added to alloys to improve their workability and to increase the strength of magnesium and aluminum alloys. It is also used in the production of non-ferrous metals and steel as a deoxidizer.
- Lasers: Yttrium aluminum garnet (YAG) lasers are used in various fields, including dentistry, cosmetic skin treatments, and in removing cataracts from the eye.
- Medical: The isotope 9090Y is used in radiation therapy, especially for liver cancer, where it is used in a process known as radioembolization. It targets cancer cells with high doses of radiation while minimizing the impact on surrounding healthy tissues.
- Aerospace: Yttrium oxide is used in the thermal barrier coatings of jet engine components, helping them withstand high temperatures.
- Catalysts: Yttrium is used as a catalyst in the polymerization of ethylene and in various chemical reactions.
Production of Yttrium
The production of yttrium involves several steps, starting from the extraction of yttrium-containing minerals to the purification and production of pure yttrium metal or its compounds. Here’s a general overview:
- Extraction from Minerals: Yttrium is primarily extracted from rare earth minerals such as monazite and xenotime. These minerals are mined, and the ore is processed to extract the rare earth elements.
- Separation and Purification: The rare earth elements extracted from the ore are a mixture of different elements, including yttrium. This mixture is subjected to various separation processes, such as solvent extraction or ion exchange, to isolate yttrium from the other elements.
- Conversion to Oxides: The isolated yttrium is often converted into yttrium oxide (Y₂O₃), a white powder that serves as the starting material for many yttrium-based compounds and alloys.
- Metal Production: Pure yttrium metal is produced from yttrium oxide through electrolysis or by a metallothermic reduction process, using reducing agents like calcium or magnesium in a high-temperature reaction.
Applications of Yttrium
Yttrium has a wide range of applications across various industries due to its unique properties. Some of the key applications include:
- Electronics: Yttrium is used in the manufacturing of phosphors for color television and computer monitor screens, as well as in LED lighting.
- Ceramics: Yttrium-stabilized zirconia (YSZ) is used in the production of strong, durable ceramics that are resistant to wear and thermal shock. These ceramics are used in dental implants, knife blades, and automotive oxygen sensors.
- Metallurgy: Yttrium is added to various metal alloys to improve their properties. For example, adding yttrium to magnesium and aluminum alloys increases their strength and resistance to corrosion.
- Medical Applications: Radioactive isotopes of yttrium, especially Yttrium-90, are used in certain types of radiation therapy for cancer treatment, including radioembolization for liver cancer.
- Lasers: Yttrium aluminum garnet (YAG) is used to make solid-state lasers for a variety of applications, including industrial cutting and welding, military, and medicine.
- Aerospace: Yttrium oxide is applied in thermal barrier coatings on jet engine components, protecting them from high temperatures.
- Superconductors: Compounds such as yttrium barium copper oxide (YBCO) are known for their high-temperature superconducting properties, useful in magnetic resonance imaging (MRI) and potential applications in power transmission.
- Catalysis: Yttrium is used as a catalyst in polymerization and other chemical reactions, improving the efficiency and selectivity of the processes.
Yttrium, a versatile element, plays a pivotal role in modern technology and industry. From enhancing the durability of ceramics to improving the efficiency of LEDs and superconductors, its applications are vast. The production process, though complex, ensures yttrium’s availability for its critical uses in electronics, medicine, and beyond, showcasing its indispensable contribution to advancement and innovation.




