Zirconium (Zr) – Definition, Preparation, Properties, Uses, Compounds, Reactivity
Dive into the comprehensive guide on Zirconium, a lustrous, grey-white, strong transition metal that plays a pivotal role across various industries. From its corrosion-resistant properties in harsh environments to its essential uses in medical implants, aerospace engineering, and nuclear reactors, Zirconium’s versatility is unmatched. This guide offers detailed insights into Zirconium’s characteristics, applications, and its significance in advancing technology and improving our daily lives. Explore real-world examples that highlight Zirconium’s integral contributions to modern advancements.
What is Zirconium?
Zirconium is a chemical element with the symbol Zr and atomic number 40. It is a transition metal known for its strong, silver-gray appearance and excellent resistance to corrosion. Zirconium does not easily react with air or water, making it exceptionally stable and useful for harsh environments. Historically, Zirconium has been used in ceramics and refractories, and today, it is vital in nuclear reactors, aerospace materials, and medical devices. For teachers, Zirconium provides a prime opportunity to explore the characteristics of transition metals, their position in the periodic table, and their essential roles in modern technology and industry
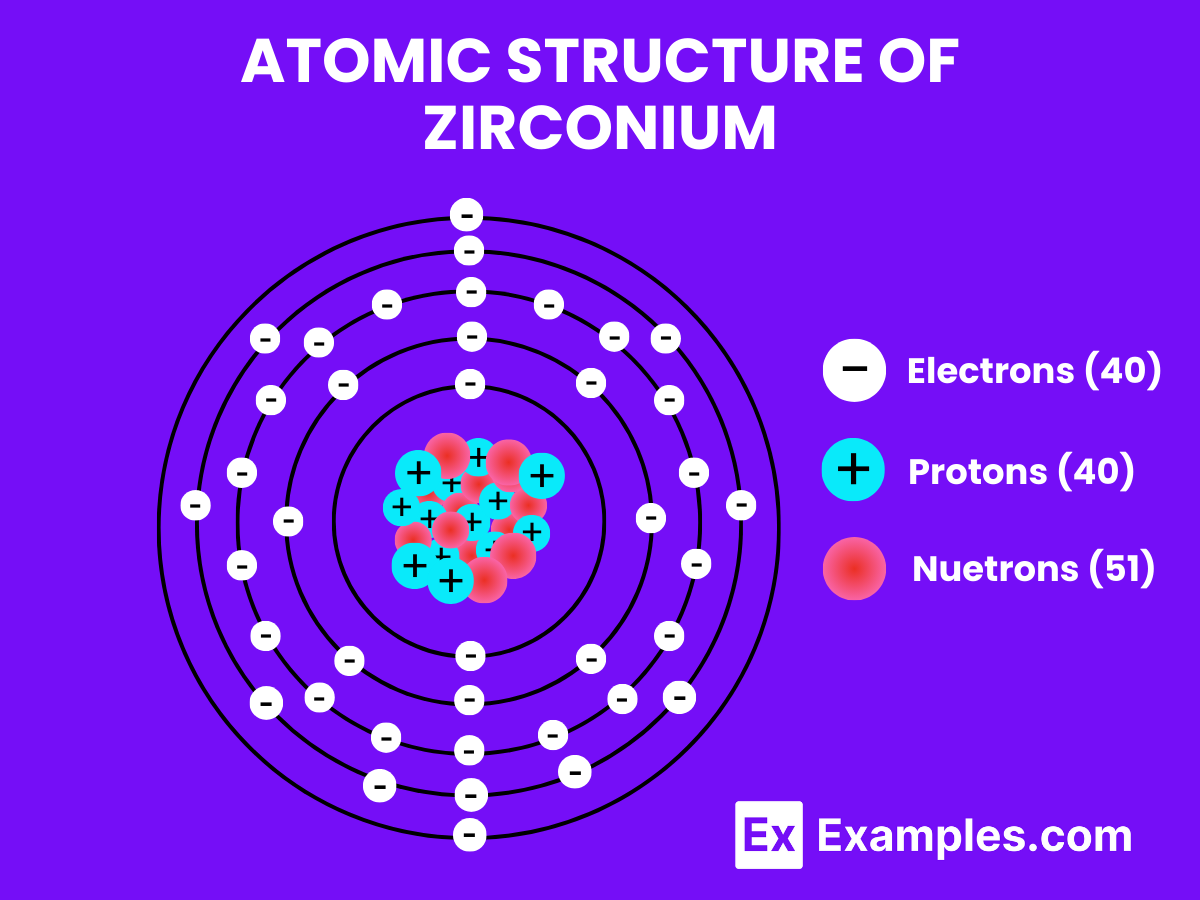
Atomic Structure of Zirconium
Zirconium is not commonly found or used in its gaseous state due to its high melting and boiling points. Zirconium is a solid metal under standard conditions. However, I can explain the atomic structure of Zirconium as an element, which applies to all its physical states (solid, liquid, gas) when referring to its electrons, protons, and neutrons.
Zirconium (Zr) has an atomic number of 40, meaning it possesses 40 protons in its nucleus. The number of neutrons in the most abundant isotope of Zirconium, Zirconium-90, is 50, giving it a mass number of 90 (40 protons + 50 neutrons). The electrons are arranged in orbitals around the nucleus. The electron configuration of Zirconium is [Kr] 4d² 5s², indicating it has two electrons in the 5s orbital and two electrons in the 4d orbital beyond the filled orbitals of Krypton (Kr), a noble gas.
- 40 protons in the nucleus, giving it its unique elemental properties.
- 50 neutrons in its most abundant isotope, contributing to the mass of the atom.
- Electrons arranged in orbitals, with the electron configuration of [Kr] 4d²5s², reflecting its position in the periodic table as a transition metal.
Zirconium Formula
Formula: Zr
Composition: A single zirconium atom.
Bond Type: Zirconium forms metallic bonds in its metallic form and can form covalent bonds in its compounds, utilizing four valence electrons.
Molecular Structure: Zirconium does not exist in different allotropes like tin. It is strong, resistant to corrosion, and has a high melting point, making it valuable in harsh conditions.
Electron Configuration: 40 electrons, with the configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d² 5s².
Significance: Crucial in the nuclear industry for cladding fuel elements due to its low neutron absorption. Also used in dental implants, medical instruments, and aerospace engineering.
Role in Chemistry: Important for understanding transition metals’ properties, applications in advanced materials, and zirconium compound synthesis in organometallic chemistry.
Properties of Zirconium
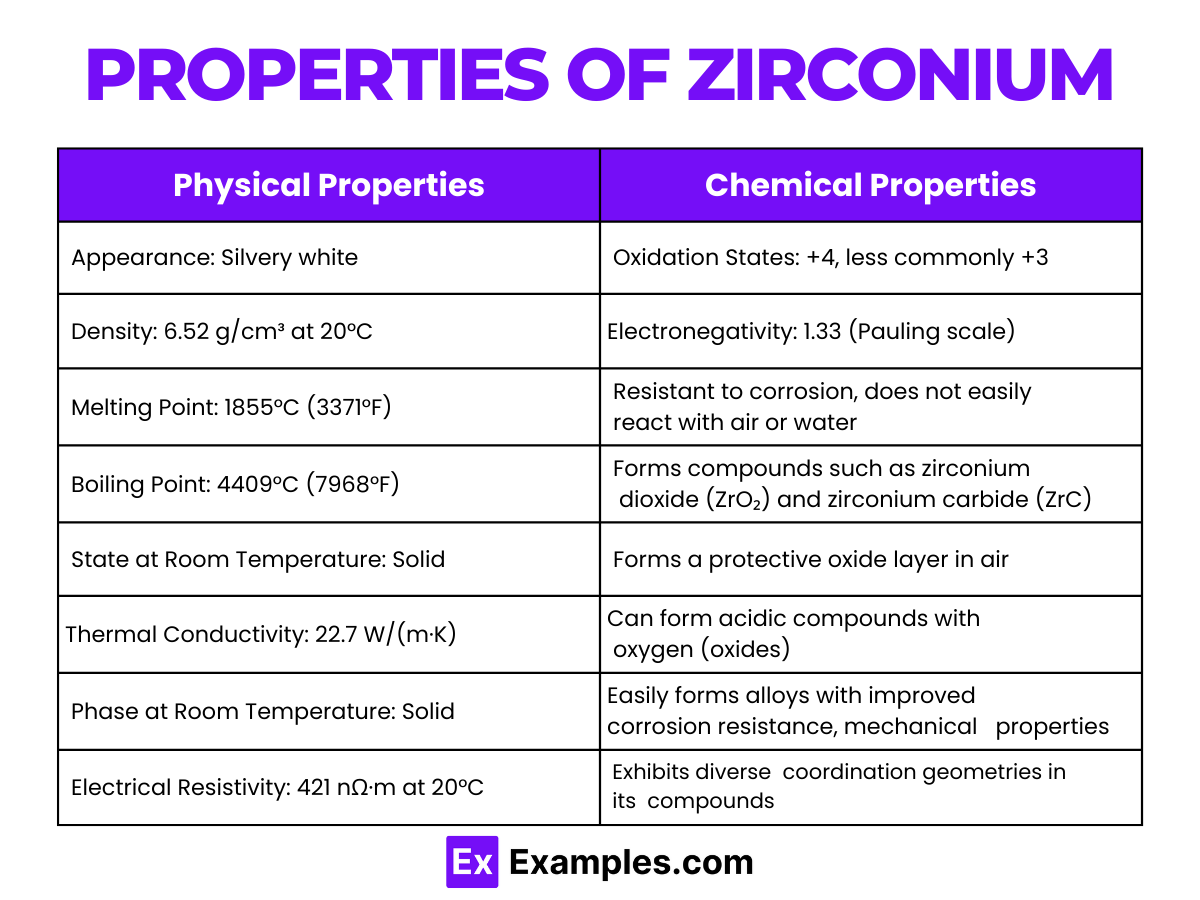
Physical Properties of Zirconium
| Property | Value |
|---|---|
| Appearance | Silvery white and lustrous |
| Density | 6.52 g/cm³ at 20°C |
| Melting Point | 1855°C (3371°F) |
| Boiling Point | 4409°C (7968°F) |
| State at Room Temperature | Solid |
| Thermal Conductivity | 22.7 W/(m·K) |
| Phase at Room Temperature | Solid |
| Electrical Resistivity | 421 nΩ·m at 20°C |
| Heat of Fusion | 14 kJ/mol |
| Heat of Vaporization | 580 kJ/mol |
Chemical Properties of Zirconium
Zirconium, a transition metal with the symbol Zr and atomic number 40, exhibits several notable chemical properties due to its position in the periodic table. These properties allow Zirconium to play a crucial role in various industrial and chemical processes:
- Oxidation States: Zirconium primarily exhibits an oxidation state of +4 in its compounds, although the +3 state is also possible in some complex compounds. The +4 state is stable and forms the basis for many of its compounds.
- Example: Zirconium dioxide (ZrO₂), a major compound of Zirconium in the +4 oxidation state.
- Reactivity with Oxygen: Zirconium reacts with oxygen in the air to form a thin, protective layer of zirconium dioxide (ZrO₂). This oxide layer protects the underlying metal from further corrosion.
- Equation: Zr+O₂→ZrO₂
- Reaction with Acids and Alkalis: Zirconium is resistant to most acids but dissolves in hydrofluoric acid (HF), forming zirconium tetrafluoride (ZrF₄). It also reacts with alkalis to form zirconates.
- Equation with HF: Zr+4HF→ZrF4+2H₂
- Equation with Alkalis: Zr+2OH−+2H2O→Zr(OH)₄²−+2H₂
- Affinity for Oxygen: Zirconium’s strong affinity for oxygen is utilized in the removal of oxygen from vacuum tubes and in the deoxidation of steel and other metals.
- Formation of Alloys: Zirconium forms alloys with enhanced corrosion resistance and high-temperature stability. It’s particularly valued in nuclear reactors for its low absorption cross-section for thermal neutrons.
- Example: Zircaloy, an alloy of Zirconium used in nuclear reactor fuel rods.
- Coordination Chemistry: In its compounds, Zirconium exhibits diverse coordination geometries, binding to various ligands. This versatility makes Zirconium complexes useful in organic synthesis and as catalysts.
- Example: Zirconium tetrachloride (ZrCl₄) is used in organic syntheses.
Thermodynamic Properties of Zirconium
| Property | Value |
|---|---|
| Melting Point | 1855°C (3371°F) |
| Boiling Point | 4409°C (7968°F) |
| Heat of Fusion | 14 kJ/mol |
| Heat of Vaporization | 580 kJ/mol |
| Specific Heat Capacity | 0.278 J/(g·K) |
Material Properties of Zirconium
| Property | Value |
|---|---|
| Density | 6.52 g/cm³ |
| Young’s Modulus | 68 GPa |
| Tensile Strength | 230 MPa |
| Hardness | 5 Mohs |
| Malleability | High |
| Ductility | Moderate |
Electromagnetic Properties of Zirconium
| Property | Value |
|---|---|
| Electrical Resistivity | 421 nΩ·m |
Nuclear Properties of Zirconium
| Property | Value |
|---|---|
| Atomic Number | 40 |
| Average Atomic Mass | 91.224 u |
| Neutron Cross Section | Low (varies by isotope) |
| Number of Stable Isotopes | 5 |
Preparation of Zirconium
The preparation of Zirconium typically involves the extraction and purification of zirconium from its primary ore, zircon (ZrSiO₄). The process includes several key steps:
- Mining and Concentration: Zircon ore is mined and concentrated using gravitational and magnetic separation techniques.
- Reduction of Zirconium Compound: The concentrated zircon is then reacted with chlorine in a high-temperature process to produce zirconium tetrachloride (ZrCl₄).
- ZrSiO₄+2Cl₂→ZrCl₄+SiCl₄+2O₂
- Purification: ZrCl₄ is purified by fractional distillation to remove impurities.
- Reduction to Metallic Zirconium: The purified ZrCl₄ is reduced with magnesium in a Kroll process to produce metallic zirconium.
- ZrCl₄+2Mg→Zr+2MgCl₂
- Final Processing: The metallic zirconium is then processed into various forms, such as bars or powders, for industrial use
Chemical Compounds of Zirconium
- Zirconium Dioxide (ZrO₂): Known as zirconia, it’s a white crystalline oxide of zirconium. Its high melting point and strength make it useful in ceramic and refractory applications.
- Zr+O₂→ZrO₂
- Zirconium Tetrachloride (ZrCl₄): This is used as an intermediate in the production of zirconium metal and as a catalyst in organic synthesis.
- Zr+2Cl₂→ZrCl₄
- Zirconium Carbide (ZrC): A very hard material used in cutting tools and as an abrasive.
- Zr+C→ZrC
- Zirconium Nitride (ZrN): Offers a hard, corrosion-resistant surface, used in coatings to reduce wear on metal parts.
- Zr+12N₂→ZrN
- Zirconium Sulfate (Zr(SO₄)₂): Used in leather tanning and as a chemical intermediate.
- Zr+2H₂SO₄→Zr(SO₄)₂+2H₂
- Zirconium Hydride (ZrH₂): Utilized in nuclear reactors as a neutron moderator and in powder metallurgy.
- Zr+H₂→ZrH₂
Isotopes of Zirconium
Zirconium is a chemical element with the symbol Zr and atomic number 40. It is known for its strong resistance to corrosion and high melting point, making it valuable in various industrial applications. Zirconium has several isotopes, which are variants of the element that differ in neutron number. Below is a table outlining the main isotopes of zirconium, their mass numbers, and some of their properties.
| Isotope | Mass Number | Natural Abundance (%) | Half-life | Notes |
|---|---|---|---|---|
| Zr-90 | 90 | 51.45 | Stable | Most abundant isotope, used in nuclear reactors |
| Zr-91 | 91 | 11.22 | Stable | – |
| Zr-92 | 92 | 17.15 | Stable | – |
| Zr-93 | 93 | 0 | 1.53 million years | Produced artificially, potential for nuclear waste disposal |
| Zr-94 | 94 | 17.38 | Stable | – |
| Zr-96 | 96 | 2.80 | Stable | Has potential in isotope geochemistry studies |
Stable isotopes like Zr-90, Zr-91, Zr-92, Zr-94, and Zr-96 have a variety of uses in industry and scientific research, while radioactive isotopes like Zr-93 are studied for their potential applications in nuclear waste management.
Uses of Zirconium
Zirconium is a versatile metal that finds applications in numerous fields due to its excellent corrosion resistance, high melting point, and unique properties. Here are some of the primary uses of zirconium:
- Nuclear Energy: Zirconium’s low neutron absorption makes it an ideal material for use in nuclear reactors, particularly in the cladding of fuel rods.
- Aerospace Industry: Due to its high resistance to heat, zirconium alloys are used in the construction of jet engines and space vehicles.
- Medical Applications: Zirconium’s biocompatibility allows its use in dental implants and other prosthetics.
- Chemical Processing: Its corrosion resistance makes zirconium suitable for pipes, fittings, and heat exchangers in corrosive environments.
- Consumer Products: Zirconium compounds are used in deodorants, antiperspirants, and as an opacifier in ceramics and porcelain products.
- Jewelry and Watches: Zirconium can be transformed into a black, ceramic-like material used in jewelry and watch cases for its aesthetic appeal and durability
Production of Zirconium
The production of zirconium involves several steps to extract the metal from its ore, primarily zircon (ZrSiO4), and refine it to its pure form or into useful alloys. Here’s an overview of the zirconium production process:
- Mining: The first step involves mining zirconium-containing minerals, with zircon being the most commercially significant. These minerals are usually extracted from heavy mineral sands through open-pit or dredging mining techniques.
- Extracting Zirconium from Zircon: The extracted zircon is purified through various processes, including crushing, milling, and sometimes magnetic separation. The purified zircon (ZrSiO₄) is then converted into zirconium chloride (ZrCl₄) or other zirconium compounds through chlorination.
- Conversion to Zirconium Metal: Zirconium metal is produced from zirconium chloride using the Kroll process. In this process, zirconium chloride is reduced by magnesium in a high-temperature reaction to produce metallic zirconium and magnesium chloride. The metallic zirconium is then purified to remove any remaining impurities.
- Forming Zirconium Products: The purified zirconium metal is melted and cast into ingots, which can be further processed into various forms, such as sheets, bars, or tubes, depending on the intended application. Zirconium alloys, particularly with niobium, are also produced for specific applications, such as in the nuclear industry.
Applications of Zirconium
Zirconium is utilized in various applications due to its excellent properties, such as high resistance to corrosion, good heat resistance, and low absorption of neutrons. Below are some key applications of zirconium and its alloys:
- Nuclear Industry: Zirconium alloys, especially zircaloy, are widely used as cladding for nuclear reactor fuels due to their low neutron absorption rate and good corrosion resistance in high-temperature water.
- Chemical Industry: Zirconium’s excellent corrosion resistance to acids and alkalis makes it suitable for pipes, valves, and reactors in chemical plants.
- Aerospace and Defense: Zirconium is used in the aerospace industry for its heat resistance, in applications such as jet engine components and heat shields.
- Biomedical Devices: Due to its biocompatibility, zirconium is used in dental implants, hip replacements, and other medical devices.
- Consumer Electronics: Zirconium compounds are used in the production of scratch-resistant glass for smartphones, tablets, and other electronic devices.
- Jewelry: Zirconium can be processed into a black, corrosion-resistant metal that is popular in jewelry for its aesthetic appeal and durability.
- Catalysts: Zirconium compounds serve as catalysts in certain chemical reactions, including polymerization processes.
Zirconium, a versatile element, plays a pivotal role across various sectors due to its remarkable properties. From nuclear reactors and aerospace components to medical implants and consumer electronics, zirconium’s applications are extensive. Understanding its isotopes, production process, and multifaceted uses not only highlights its significance in modern technology but also its potential for future innovations.