Ammonia (NH₃) – Definition, Structure, Preparation, Uses, Benefits, Side Effects
Ammonia is a fascinating player in the world of chemistry, known for its sharp smell and clear, colorless gas form. As a covalent compound, it’s formed when nitrogen and hydrogen atoms share electrons tightly, bonding them together in a unique and stable way. This simple yet intriguing substance plays a crucial role in both nature and industry, showcasing the amazing versatility of chemical compounds.
What is Ammonia?
Chemical Names and Formulas
| Property | Value |
|---|---|
| Formula | NH₃ |
| Hill formula | H₃N |
| Name | Ammonia |
| Alternate names | Ammonia anhydrous, Ammonia gas, Azane, Nitro-sil, Spirit of Hartshorn |
Structure of Ammonia
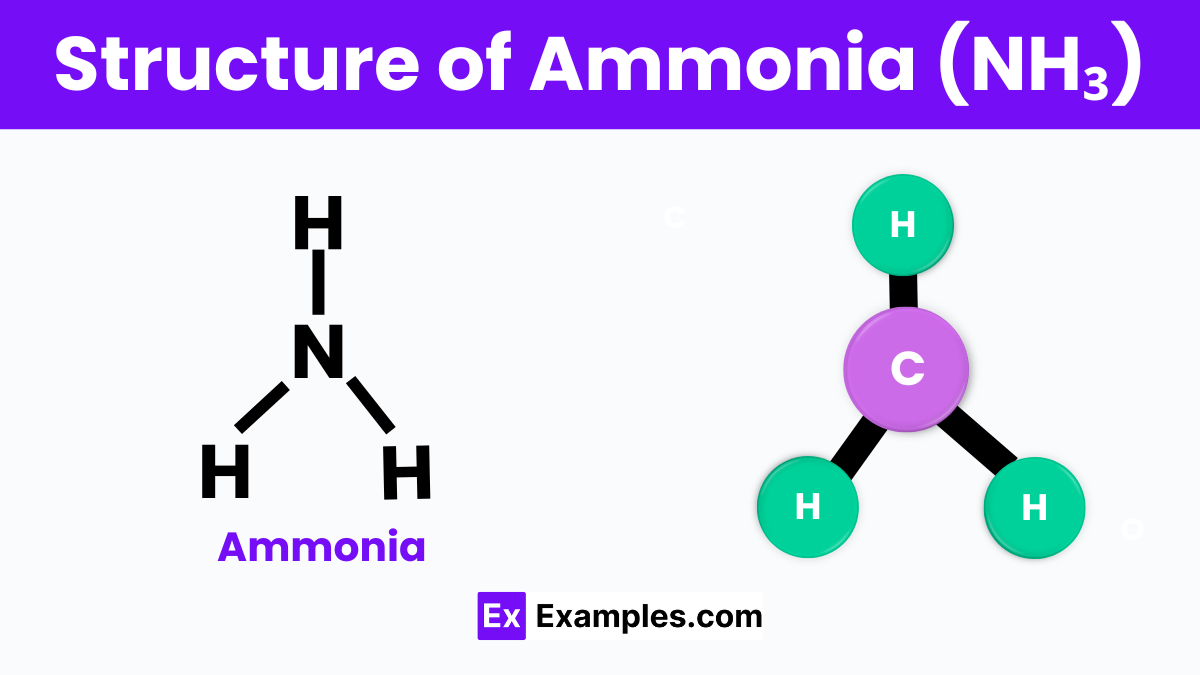
Ammonia is a compound made up of nitrogen and hydrogen with the formula NH3. This means that one molecule of ammonia contains one nitrogen atom and three hydrogen atoms. The structure of ammonia is shaped like a pyramid. At the top of this pyramid is the nitrogen atom, and the three hydrogen atoms form the base. This shape is often referred to as a trigonal pyramidal structure. The nitrogen atom has five electrons in its outer shell, but it shares three of these electrons with three hydrogen atoms to form three covalent bonds. This sharing of electrons allows the ammonia molecule to have a stable structure. Additionally, the nitrogen atom has a lone pair of electrons that are not shared with the hydrogen atoms, which contributes to the molecule’s shape and properties.
Preparation of Ammonia
Ammonia is commonly prepared in
the industry through a process called the Haber-Bosch process. This process involves combining nitrogen gas (N2) from the air with hydrogen gas (H2) under high pressure and temperature in the presence of a catalyst. A catalyst is a substance that helps speed up a chemical reaction without being used up in the process. The high pressure and temperature conditions, along with the catalyst, help break the strong bonds of nitrogen gas molecules, allowing them to combine with hydrogen to form ammonia (NH3).The chemical equation for the Haber-Bosch process is:
This equation shows that one molecule of nitrogen gas reacts with three molecules of hydrogen gas to produce two molecules of ammonia gas. The process is very important for producing ammonia on a large scale, which is then used to make fertilizers, explosives, and other chemicals. The Haber-Bosch process is efficient and is the primary method for ammonia production worldwide.
Physical Properties of Ammonia
| Property | Detail |
|---|---|
| Molecular Formula | NH₃ |
| Molecular Weight | 17.031 g/mol |
| State at Room Temperature | Gas |
| Color | Colorless |
| Odor | Sharp, pungent |
| Boiling Point | -33.34°C (-28.01°F) |
| Melting Point | -77.73°C (-107.91°F) |
| Solubility in Water | Highly soluble, forming a basic solution (ammonium hydroxide) |
| Density | 0.73 kg/m³ (gas at 0°C and 1 atm) |
| pH | About 11 when dissolved in water to form ammonium hydroxide |
Chemical Properties of Ammonia
Basic Nature
Ammonia acts as a base when dissolved in water, producing hydroxide ions (OH-). This makes the solution alkaline.
Equation: NH₃ + H₂O → NH₄⁺ + OH⁻
Reacts with Acids
Ammonia reacts with acids to form ammonium salts. For example, when it reacts with hydrochloric acid (HCl), ammonium chloride (NH4Cl) is formed.
Equation: NH₃ + HCl → NH₄Cl
Combustibility
Ammonia can burn in the presence of oxygen, but it requires a high temperature to ignite. The reaction with oxygen produces nitrogen gas (N2) and water (H2O).
Equation: 4NH₃ + 3O₂ → 2N₂ + 6H₂O
Formation of Complexes
Ammonia can form complex compou
nds with metals. For instance, it forms a complex with copper sulfate, resulting in a deep blue solution. This is due to the formation of the [Cu(NH₃)₄]²⁺complex ion.
Decomposition by Heat
When heated, ammonia can decompose into its elements, nitrogen, and hydrogen. However, this requires very high temperatures and is not a common reaction under normal conditions.
Ammonia (NH₃) Chemical Compound Information
Chemical Identifiers
| Property | Value |
|---|---|
| CAS registry number | 7664-41-7 |
| Beilstein number | 3587154 |
| PubChem compound ID | 222 |
| PubChem substance ID | 24857750 |
| SMILES identifier | N |
| InChI identifier | InChI=1/H3N/h1H3 |
| RTECS number | BO0875000 |
| MDL number | MFCD00011418 |
NFPA label
| Property | Value |
|---|---|
| NFPA health rating | 3 |
| NFPA fire rating | 1 |
| NFPA reactivity rating | 0 |
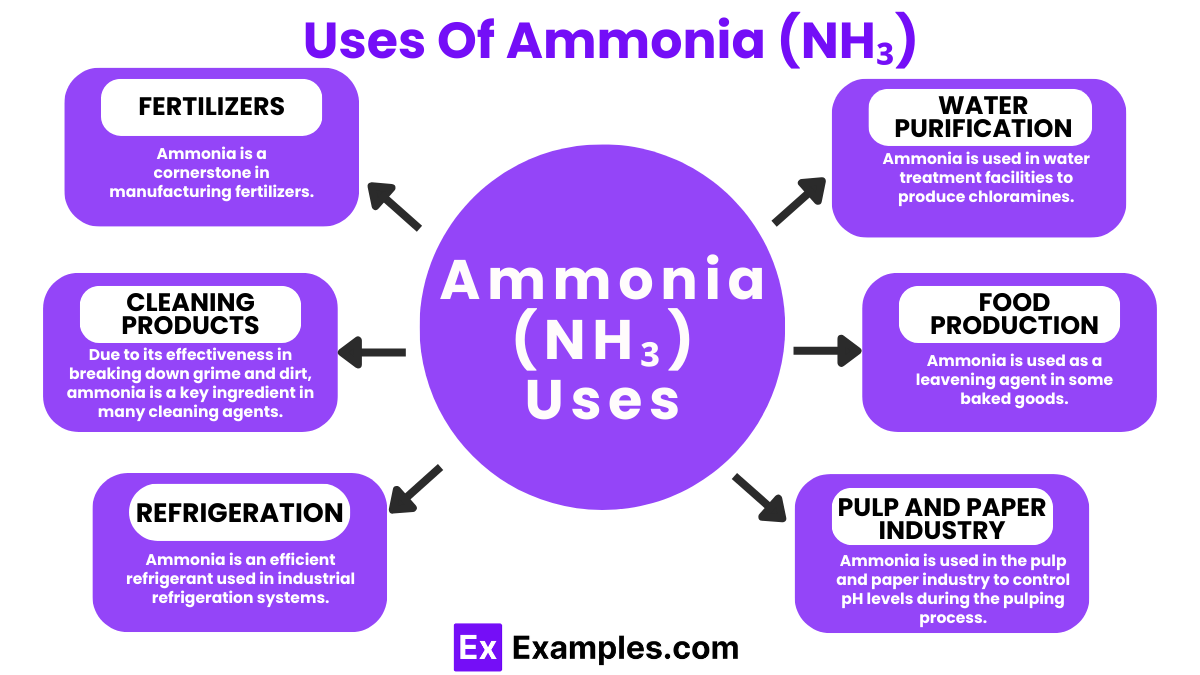
Uses of Ammonia (NH₃)
Fertilizers
Ammonia is a cornerstone in manufacturing fertilizers. It’s directly applied to soil in its anhydrous form or used to produce nitrogen-rich compounds like urea, ammonium nitrate, and ammonium sulfate. These fertilizers are essential for promoting plant growth and increasing crop yields.
Cleaning Products
Due to its effectiveness in breaking down grime and dirt, ammonia is a key ingredient in many cleaning agents. It’s especially popular in solutions for cleaning windows and stainless steel, leaving surfaces sparkling without streaks.
Refrigeration
Ammonia is an efficient refrigerant used in industrial refrigeration systems, including those in food processing plants and cold storage facilities. Its high energy efficiency and low environmental impact make it a preferred choice over some synthetic refrigerants.
Pharmaceuticals
Ammonia plays a role in producing various pharmaceutical products. It’s involved in synthesizing medications, such as antibiotics and vitamins, contributing to health and wellness.
Textile Manufacturing
In the textile industry, ammonia is used in pre-treatment processes of cotton materials. It helps in mercerization, where cotton fibers are treated with ammonia to increase their luster, strength, and affinity for dyes.
Pulp and Paper Industry
Ammonia is used in the pulp and paper industry to control pH levels during the pulping process. Adjusting the pH is crucial for improving the quality and durability of the paper.
Water Purification
Ammonia is used in water treatment facilities to produce chloramines. Chloramines are disinfectants used to purify drinking water, effectively killing harmful microorganisms while minimizing the formation of harmful by-products.
Explosives Manufacturing
Ammonium nitrate, made from ammonia, is a key component in making explosives. These explosives are widely used in mining, construction, and demolition activities.
Plastic and Synthetic Fibers
Ammonia is involved in producing plastics and synthetic fibers. It acts as a feedstock for producing acrylonitrile, which is a precursor for acrylic fibers and various plastics.
Food Production
Ammonia is used as a leavening agent in some baked goods. It helps to adjust acidity levels in food, ensuring proper growth of yeasts and other microorganisms during fermentation.
Benefits of Ammonia (NH₃)
Agricultural Productivity
- Enhances Crop Growth: Ammonia is a primary source of nitrogen for plants, a critical nutrient that supports healthy growth and development. Its use in fertilizers contributes significantly to increasing crop yields and global food production.
Eco-Friendly Refrigerant
- Low Environmental Impact: Compared to many synthetic refrigerants, ammonia has a zero ozone depletion potential and a negligible global warming potential, making it an environmentally friendly choice for industrial refrigeration systems.
Effective Cleaner
- Powerful Cleaning Agent: Ammonia effectively cuts through dirt, grease, and grime, making it a potent component in many household and industrial cleaners. It leaves surfaces clean and streak-free, especially glass and stainless steel.
Versatile Industrial Chemical
- Broad Industrial Applications: Beyond agriculture and cleaning, ammonia is crucial in producing plastics, fibers, explosives, and pharmaceuticals. Its versatility underpins numerous manufacturing processes.
Water Treatment
- Improves Water Safety: In water treatment, ammonia is used to produce chloramines, which serve as disinfectants. This helps ensure safe drinking water by effectively killing harmful microorganisms.
Economic Efficiency
- Cost-Effective Production and Usage: Ammonia can be synthesized on a large scale using readily available natural resources, such as air and natural gas. Its efficiency and relatively low production cost make it economically favorable for many applications.
Textile Manufacturing
- Enhances Fabric Quality: Ammonia is used in treating cotton and other textiles, improving their strength, luster, and dye affinity. This treatment, known as mercerization, enhances the quality and appearance of textile products.
Waste and Emission Reduction
- Reduces Harmful Emissions: Ammonia’s role in reducing emissions from agricultural activities and its use as a cleaner-burning fuel alternative in some industrial processes contribute to lower environmental pollution.
Scientific Research and Development
- Facilitates Innovation: The properties of ammonia make it an interesting subject for scientific research, leading to new technologies and applications, such as its potential use in hydrogen storage and as an alternative fuel source.
Side Effects Of Ammonia (NH₃)
- Respiratory Irritation: Inhaling ammonia can irritate the respiratory tract, causing coughing and wheezing.
- Skin and Eye Irritation: Direct contact with ammonia can cause burns and severe irritation to the skin and eyes.
- Toxicity: High exposure levels can be toxic, leading to symptoms like headache, nausea, and even respiratory failure.
- Environmental Impact: Ammonia can contribute to water pollution and ecosystem disruption, affecting aquatic life.
- Explosion Risk: Ammonia forms explosive mixtures with air under certain conditions, posing a safety hazard.
- Corrosiveness: It’s corrosive to some metals, which can damage equipment and infrastructure.
- Odor: Ammonia has a strong, unpleasant smell, detectable even at low concentrations.
- Chronic Exposure: Long-term exposure may result in chronic respiratory conditions.
- Occupational Hazards: Workers in industries using ammonia face higher exposure risks.
- Effects on Animals: Ammonia emissions can harm wildlife and domestic animals’ health.
FAQ’S
Is Ammonia Toxic to Us?
Yes, ammonia can be toxic when inhaled, ingested, or if it comes into contact with skin, causing respiratory issues, skin burns, and eye irritation.
Which Is Better: Ammonia or Vinegar?
Both ammonia and vinegar are effective cleaners, but ammonia is stronger for tough grime, while vinegar is safer and eco-friendly for everyday use.
What Should Not Be Cleaned with Ammonia?
Avoid using ammonia on brass, copper, marble, and certain plastics as it can cause damage. Also, never mix ammonia with bleach due to toxic fumes.
Where Does Ammonia Come From?
Ammonia is produced naturally from the decomposition of organic matter and is also synthetically manufactured for industrial and agricultural uses.