Benzene (C₆H₆) – Definition, Structure, Preparation, Properties, Uses, Side Effects
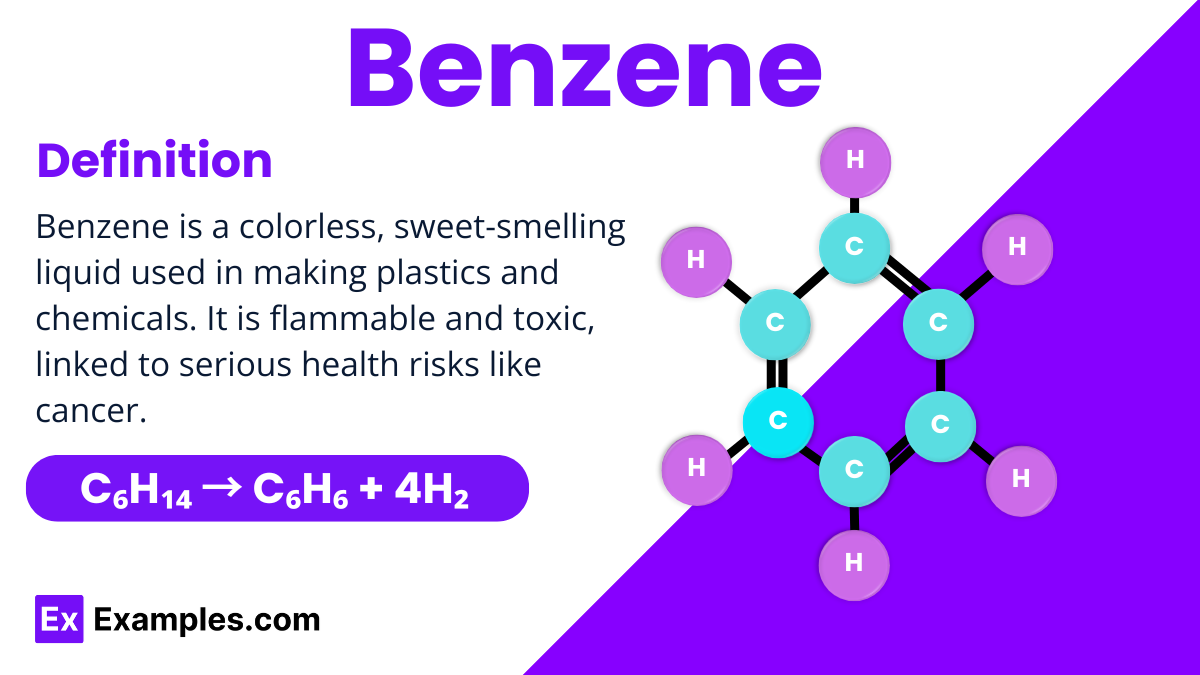
Benzene is a fundamental organic compound in chemistry, known for its simple yet unique structure consisting of six carbon atoms forming a ring, each bonded to a hydrogen atom. This arrangement is often depicted as a hexagon with alternating double bonds, making it a perfect example of aromatic stability. Benzene is colorless, highly flammable, and has a distinct sweet smell, used extensively in manufacturing a wide range of products, from plastics to synthetic fibers.
What is Benzene?
Chemical Names And Formulas
| Property | Value |
|---|---|
| Formula | C₆H₆ |
| Name | Benzene |
| Alternate Names | Benzin, Benzine, Benzol, Benzole, Benzolene, Cyclohexatriene, Phenyl Hydride, Pyrobenzole |
Structure of Benzene
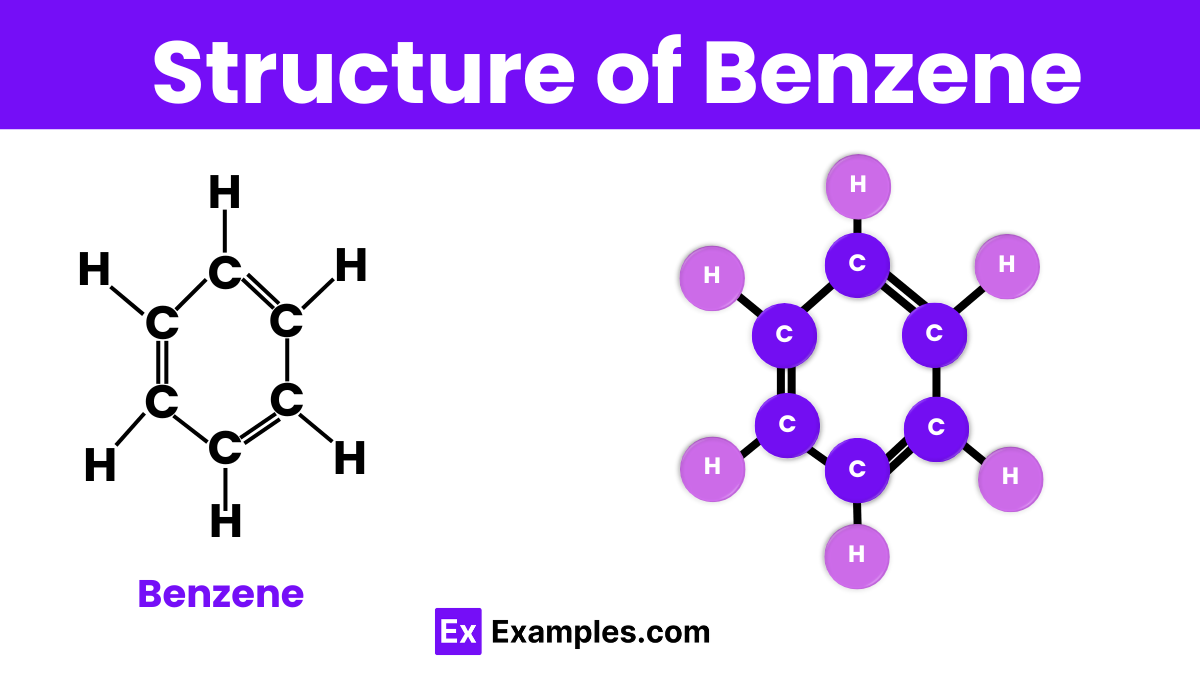
Benzene is a fascinating molecule with a unique structure that makes it stand out in organic chemistry. It consists of six carbon atoms arranged in a hexagonal ring, connected by alternating single and double bonds. This arrangement is known as aromatic due to its stability and special bonding. Each carbon atom in the ring is also bonded to one hydrogen atom. The beauty of benzene’s structure lies in its symmetry and the equal length of all its carbon-carbon bonds, which are shorter than typical single bonds but longer than double bonds. This configuration allows benzene to be exceptionally stable and widely used in various chemical processes.
Preparation of Benzene
Benzene can be prepared through several methods, but one of the most common is the catalytic reforming of alkanes. This process involves converting straight-chain hydrocarbons from petroleum into benzene, among other products. A typical reaction might involve heating an alkane like hexane in the presence of a platinum catalyst, which facilitates the rearrangement and eventual formation of benzene.A basic chemical equation illustrating this transformation is:
This equation shows hexane (C₆H₁₄) being converted into benzene (C₆H₆) and hydrogen gas (H₂). The production of benzene in this manner highlights its importance in industrial applications, where it serves as a building block for many chemicals and synthetic materials.
Physical Properties of Benzene
| Property | Description |
|---|---|
| Appearance | Clear, colorless liquid |
| Smell | Sweet, aromatic |
| Boiling Point | 80.1°C (176°F) |
| Melting Point | 5.5°C (41.9°F) |
| Density | 0.8765 g/cm³ (at 20°C) |
| Solubility in Water | Insoluble |
| Vapor Pressure | 12.7 kPa (at 25°C) |
| Molecular Weight | 78.11 g/mol |
| Flammability | Highly flammable |
Chemical Properties of Benzene
Aromaticity
- Benzene is highly stable despite having alternating double bonds. This stability, known as aromaticity, comes from the electrons in its double bonds being shared around the ring in a phenomenon called resonance.
Electrophilic Substitution
- Benzene often undergoes electrophilic substitution reactions, where an electrophile replaces one of the hydrogen atoms. For example, benzene reacts with nitric acid, yielding nitrobenzene, which is a key step in manufacturing explosives and synthetic dyes.
- Equation: C₆H₆ + HNO₃ → C₆H₅NO₂ + H₂O
Combustion
- Benzene burns in the presence of oxygen, producing carbon dioxide, water, and heat. This reaction releases a significant amount of energy, which is characteristic of hydrocarbon combustion.
- Equation: 2C₆H₆ + 15O₂ → 12CO₂ + 6H₂O
Addition Reactions
- Although less common due to the stability of its aromatic ring. Benzene can undergo addition reactions under specific conditions, like in the hydrogenation process. Benzene is converted to cyclohexane in the presence of a nickel catalyst.
- Equation: C₆H₆ + 3H₂ → C₆H₁₂
Benzene (C₆H₆) Chemical Compound Information
Chemical Identifiers
| Property | Value |
|---|---|
| CAS Registry Number | 71-43-2 |
| Beilstein Number | 969212 |
| PubChem Compound ID | 241 |
| SMILES Identifier | C1=CC=CC=C1 |
| InChI Identifier | InChI=1/C6H6/c1-2-4-6-5-3-1/h1-6H |
| RTECS Number | CY1400000 |
| MDL Number | MFCD00003009 |
NFPA Label
| Property | Value |
|---|---|
| NFPA Health Rating | 2 |
| NFPA Fire Rating | 3 |
| NFPA Reactivity Rating | 0 |
Uses of Benzene
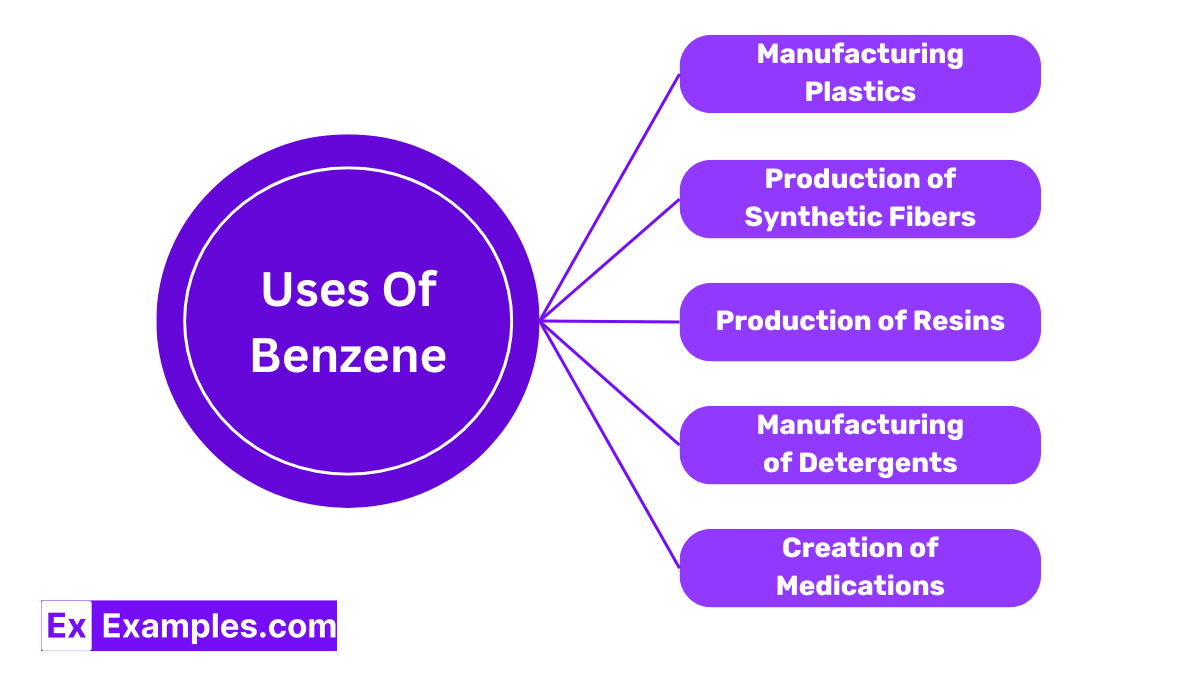
Manufacturing Plastics
Benzene is a crucial ingredient in the production of polystyrene, a type of plastic used widely in everything from packaging to insulation materials. Its ability to polymerize easily makes it invaluable in creating durable and versatile plastic products.
Production of Synthetic Fibers
Benzene is used to manufacturing synthetic fibers like nylon and polyester. These fibers are essential for producing a wide range of products, from clothing to home furnishings, due to their strength and resistance to shrinking and stretching.
Production of Resins
Benzene is also used in the synthesis of various resins, which are substances used as the basis for coatings and adhesives. These resins enhance the durability and effectiveness of paints and varnishes, ensuring they adhere better and last longer.
Manufacturing of Detergents
Benzene derivatives are employed to enhance the cleaning power of laundry and dish washing products. These derivatives help break down stains and grease, making them effective cleaning agents.
Creation of Medications
Benzene is a building block for some pharmaceuticals, contributing to the synthesis of drugs used to treat various medical conditions. Its chemical properties allow for the creation of complex drug formulations, improving health care and treatment options.
Benefits of Benzene
Versatility in Industrial Applications
Benzene is highly used in the manufacturing of a wide range of products including plastics, synthetic rubber, and dyes. This makes it a cornerstone chemical in the industrial sector, driving innovation and production across various fields.
Economic Benefits
Due to its critical role in many manufacturing processes, benzene helps stimulate economic growth within the chemical industry and downstream sectors such as automotive, construction, and electronics. Its widespread use contributes significantly to economic development and job creation.
Advancements in Healthcare
Benzene derivatives play a vital role in pharmaceutical development, leading to the creation of new medications and treatments. These advancements improve healthcare outcomes and contribute to the well-being of populations worldwide.
Enhancement of Everyday Products
Benzene is used in the production of everyday items like lubricants, improving their effectiveness and efficiency. This enhances consumer products, making daily chores easier and supporting modern lifestyles.
Side Effects of Benzene
- Health Risks: Exposure to benzene can cause significant health issues, such as blood disorders like anemia. It negatively impacts bone marrow, impairing its ability to produce blood cells, which can weaken the immune system.
- Cancer Risk: Benzene is well-documented as a carcinogen, meaning it can lead to various forms of cancer, with leukemia being the most common. This association underscores the importance of minimizing exposure in industrial and laboratory settings.
- Environmental Impact: Benzene can contribute to environmental pollution when not handled properly. Its release into the air, water, or soil poses risks to wildlife and ecosystems, highlighting the need for effective waste management and pollution controls.
- Respiratory Issues: Inhaling benzene can cause immediate respiratory problems, ranging from dizziness and headaches to more severe conditions like respiratory tract irritation.
FAQs
What is Benzene Known For?
Benzene is famous for its use in making plastics, resins, and synthetic fibers, and for being a significant health hazard.
What Kind of Cancer Does Benzene Cause?
Benzene exposure is primarily linked to an increased risk of leukemia and other blood-related cancers.
Why is Benzene Banned?
Benzene is banned in many places due to its severe toxicity and strong links to cancer, making it a significant public health risk.