Calcium Sulfate (CaSO₄) – Definition, Structure, Preparation, Uses, Benefits, Side Effects
Calcium sulfate is a versatile chemical compound composed of calcium, sulfur, and oxygen, known for its widespread use in industries such as construction, food, and medicine. Found naturally as gypsum (dihydrate) and anhydrite (anhydrous), it plays a critical role in making plaster and drywall, acting as a food additive for coagulation and stabilization, and aiding in bone regeneration. This white, solid material highlights the interconnectedness of nature and industry, proving essential in building materials, food production, and healthcare applications.
What is Calcium Sulfate?
Chemical Names and Formulas
| Formula | CaSO₄ |
| Hill Formula | CaO₄S |
| Name | Calcium sulfate |
| Alternate Names | Calcium sulfate |
Structure of Calcium Sulfate
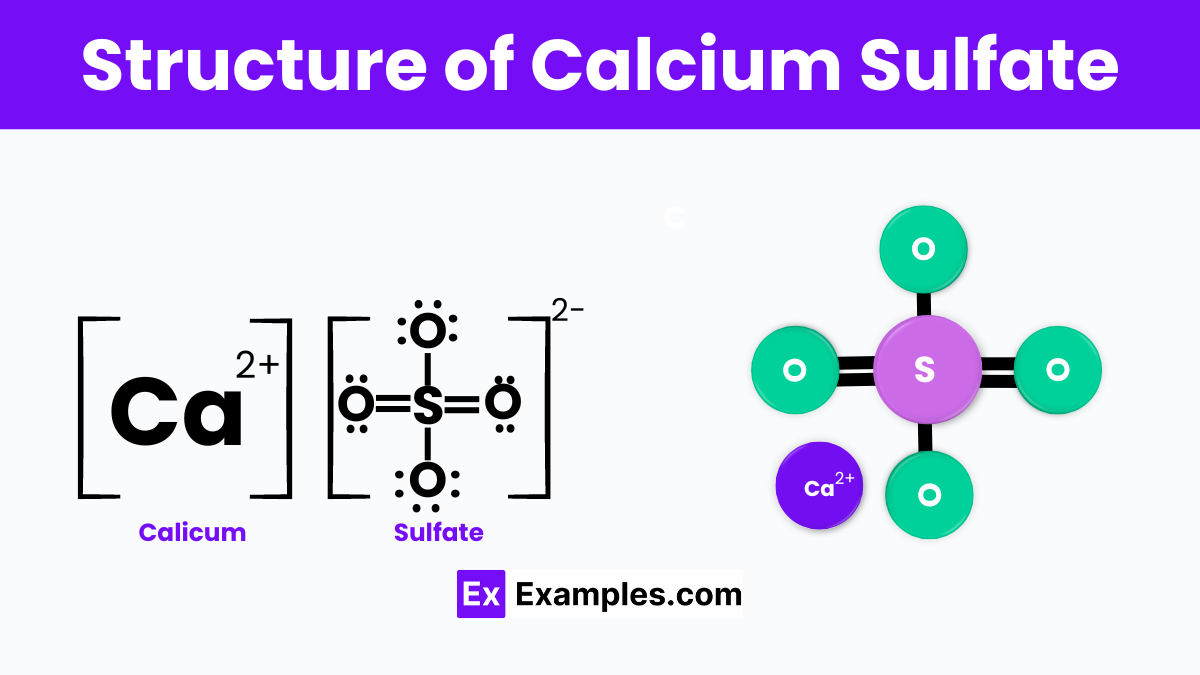
Calcium sulfate, with the chemical formula CaSO₄, is a versatile compound existing in several hydrate forms, including anhydrous calcium sulfate, dihydrate (gypsum), and hemihydrate (plaster of Paris). Each form has distinct properties and applications, making calcium sulfate essential in industries ranging from construction to food additives. Anhydrous calcium sulfate acts as a desiccant, gypsum is used for making plaster and wallboard, and plaster of Paris is utilized for casting molds and decorations due to its quick-setting nature.
The structure of calcium sulfate changes with its hydration level. Anhydrous calcium sulfate has a monoclinic crystal system with calcium ions coordinated by eight oxygen atoms, while gypsum and plaster of Paris have structures that accommodate water molecules, influencing their physical properties. These structural differences make calcium sulfate highly adaptable, serving as a key material in building materials, soil conditioners, food products as an additive (E516), and in pharmaceuticals as an excipient in tablet manufacturing.
Preparation of Calcium Sulfate
Calcium sulfate is synthesized through two primary methods: chemical reaction and dehydration of gypsum. The chemical synthesis process involves reacting calcium carbonate (CaCO₃) with sulfuric acid (H₂SO₄) to produce calcium sulfate (CaSO₄), alongside carbon dioxide (CO₂) and water (H₂O), as shown below:
In the dehydration method, gypsum (CaSO₄·2H₂O) is heated, producing either calcium sulfate hemihydrate (plaster of Paris, CaSO₄·0.5H₂O) or anhydrous calcium sulfate (CaSO₄) depending on the temperature. The equations for these reactions are:
Hemihydrate formation:
Anhydrous calcium sulfate formation:
These methods enable the versatile use of calcium sulfate in industries such as construction, where it’s used in materials like drywall and plaster, and in various industrial processes as a desiccant.
Physical Properties of Calcium Sulfate
| Property | Value |
|---|---|
| Molecular Weight | 136.14 g/mol (Anhydrous) |
| Appearance | White powder or colorless, crystalline solid |
| Density | 2.96 g/cm³ (Anhydrous), 2.32 g/cm³ (Dihydrate) |
| Melting Point | 1,460°C (Anhydrous), 145°C (Dihydrate loses water) |
| Solubility in Water | 0.21 g/100 mL (20°C, Anhydrous), 2.4 g/L (20°C, Dihydrate) |
| Crystal Structure | Monoclinic (Anhydrous and Dihydrate) |
| Thermal Conductivity | 0.17 W/(m·K) (Dihydrate) |
| Hardness (Mohs Scale) | 2 (Dihydrate) |
Chemical Properties of Calcium Sulfate
Reactivity and Stability
Calcium sulfate is characterized by low reactivity, maintaining stability across various conditions. Being non-flammable and non-toxic, it is utilized in a broad spectrum of applications, including construction materials and food additives.
Solubility
A significant property of calcium sulfate is its solubility. Dihydrate gypsum has limited water solubility, which decreases with rising temperature. Anhydrite, its anhydrous form, is less soluble, impacting its use in drywall and plaster.
Thermal Behavior
It shows unique thermal properties; gypsum loses water to become plaster of Paris (CaSO4·0.5H2O) upon heating. This dehydration is reversible, enabling plaster to harden when mixed with water. This is utilized in molds and bandages.
pH Neutral
Calcium sulfate is pH neutral in aqueous solutions, suitable for applications needing pH stability, like in agriculture for soil conditioning.
Compatibility with Other Substances
It reacts with strong acids and bases, producing calcium salts and sulfuric acid or sulfate salts. This makes it a flexible additive in chemical processes, pharmaceuticals, and food production.
Calcium Sulfate (CaSO₄) Chemical Compound Information
Chemical Identifiers
| Property | Value |
|---|---|
| CAS Registry Number | 7778-18-9 |
| PubChem Compound ID | 24497 |
| PubChem Substance ID | 24854355 |
| SMILES Identifier | [O-]S(=O)(=O)[O-].[Ca+2] |
| InChI Identifier | InChI=1/Ca.H2O4S/c;1-5(2,3)4/h;(H2,1,2,3,4)/q+2;/p-2/fCa.O4S/qm;-2 |
| RTECS Number | WS6920000 |
| MDL Number | MFCD00010912 |
Uses of Calcium Sulfate (CaSO₄)
Food Industry
- Food Additive: Enhances texture as E516 in tofu, bread, and canned veggies.
- Preservative: Extends food shelf life.
- Nutritional Supplement: Adds dietary calcium in fortified foods and drinks.
Construction and Building Materials
- Plaster of Paris: Transforms into Plaster of Paris when heated, used in construction, art, and casts.
- Cement Production: Controls cement setting time, enhancing strength.
- Drywall Manufacturing: Key ingredient in drywall for fire resistance and durability.
Agriculture
- Soil Conditioner: Improves soil structure, reducing compaction and salinity.
- Fertilizer: Supplies calcium and sulfur, aiding plant growth.
Health and Medicine
- Dental Plaster: Essential for dental impressions.
- Bone Repair: Serves as a scaffold for bone regeneration.
Industrial Applications
- Water Treatment: Removes impurities from water.
- Paper Manufacturing: Contributes to paper’s whiteness and smoothness.
Environmental Management
- Desulfurization: Used in power plants to remove sulfur dioxide from exhaust gases, cutting air pollution.
Benefits of Calcium Sulfate
Environmental Friendliness
Calcium sulfate is non-toxic and biodegradable, making it environmentally friendly. Its natural occurrence and compatibility with the environment add to its appeal in sustainable practices.
Versatility
The wide range of applications, from construction materials to food additives, highlights its versatility. This adaptability makes calcium sulfate a valuable resource across different sectors.
Nutritional Value
In the food industry, calcium sulfate provides a source of dietary calcium, an essential nutrient for bone health and metabolic functions.
Side Effects of Calcium Sulfate (CaSO₄)
- Gastrointestinal Discomfort: High intake may lead to nausea, bloating, or diarrhea.
- Allergic Reactions: Rare but can include skin irritation or respiratory difficulties.
- Calcium Imbalance: Excessive consumption might cause hypercalcemia, affecting bone health.
- Respiratory Issues: Inhalation of dust in industrial settings can lead to breathing problems.
FAQ’S
Is Calcium Sulfate Healthy to Eat?
Yes, calcium sulfate is safe and commonly used as a food additive, enhancing texture and stability in various products.
How Does Calcium Sulfate Affect the Body?
Calcium sulfate provides essential nutrients, supporting bone health and cellular function without adverse effects when consumed appropriately.
What is Another Name for Calcium Sulfate?
Calcium sulfate is also known as gypsum when in its mineral form, widely used in industries and agriculture.
What is the Household Name for Calcium Sulfate?
In households, calcium sulfate may be known as Plaster of Paris, especially in crafting and repair works.
What is the Natural Source of Calcium Sulfate?
Calcium sulfate naturally occurs in the mineral gypsum, extensively mined from the earth’s crust for various applications.


