Calcium (Ca) – Preparation, Properties, Uses, Compounds, Reactivity
Calcium, an essential element in education, plays a pivotal role in both natural processes and classroom discussions. This comprehensive guide aims to enlighten teachers about calcium’s diverse applications, ensuring they can effectively impart knowledge to their students. From understanding its fundamental properties to exploring its real-world significance, this resource provides educators with practical examples and teaching strategies, enriching the learning experience for their students. Delve into the world of calcium, and discover how to make your lessons more engaging and informative.
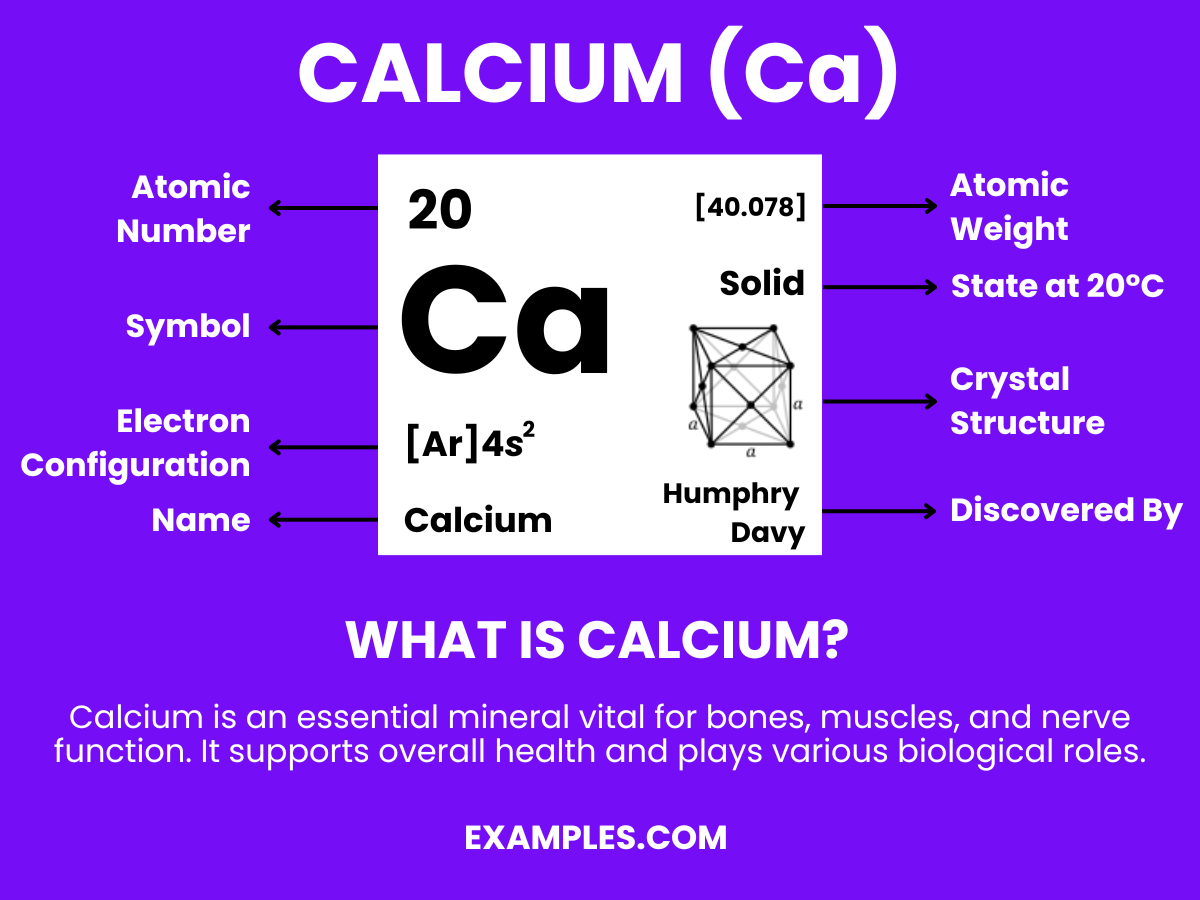
What is Calcium?
Calcium, a chemical element represented by the symbol Ca and atomic number 20, is a vital mineral for life. It’s predominantly known for its crucial role in building and maintaining strong bones and teeth. However, its importance extends beyond this, as calcium is also essential in blood clotting, muscle contraction, and nerve function. In a classroom setting, understanding calcium’s multifaceted role in both the human body and the environment provides a rich context for engaging scientific discussions. This simple yet comprehensive definition allows teachers to effectively introduce calcium in various educational scenarios, fostering a deeper understanding among students.
Other Alkaline Earth Metals
| Beryllium(Be) |
| Magnesium(Mg) |
| Strontium(Sr) |
| Barium(Ba) |
| Radium(Ra) |
Calcium Formula
- Formula: Ca
- Composition: A single calcium atom.
- Bond Type: Calcium typically forms ionic bonds due to its two valence electrons.
- Molecular Structure: A soft, silvery-white metal, less dense than magnesium.
- Electron Configuration: Twenty electrons, with the configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s².
- Significance: Crucial in construction (cement, concrete), biology (bones and teeth), and various industries.
- Role in Chemistry: Vital for living organisms, plays a key role in cellular processes, muscle function, and bone health.
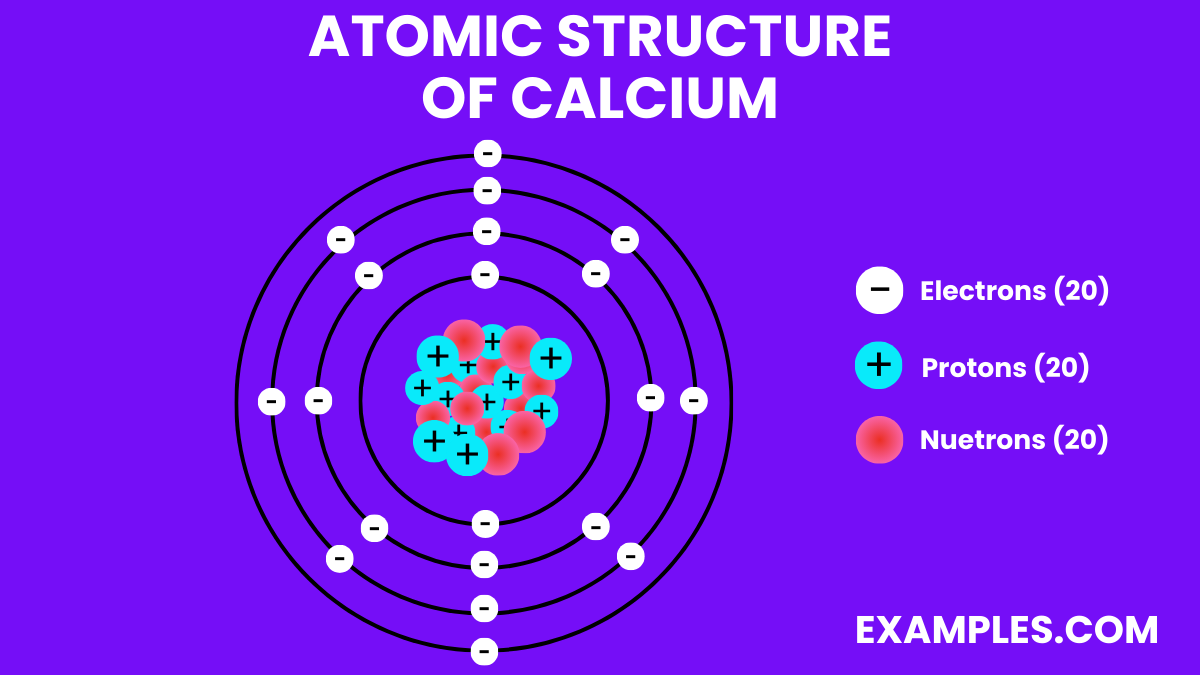
Atomic Structure of Calcium
Properties of Calcium
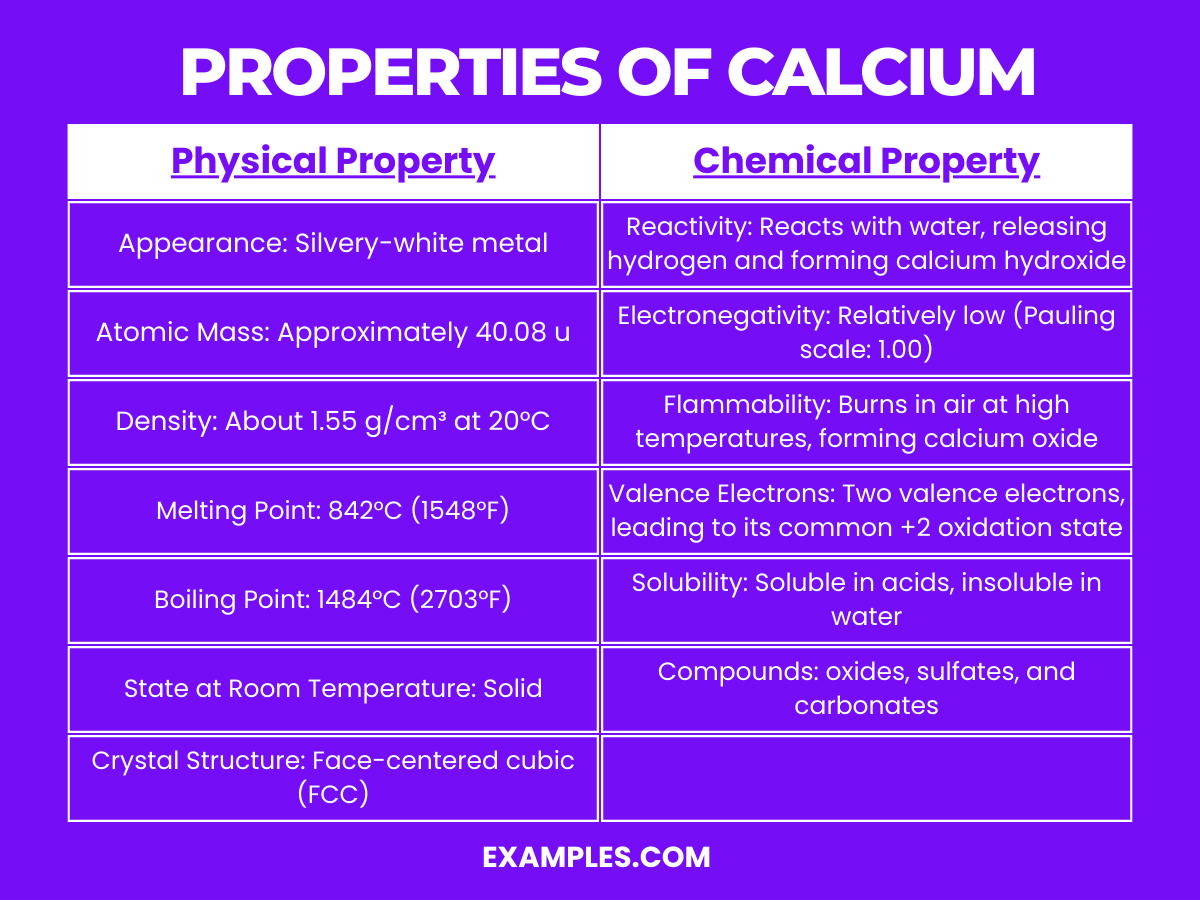
Physical Properties of Calcium
| Property | Description |
|---|---|
| Appearance | Silvery-white, somewhat soft metal |
| Melting Point | Approximately 842°C (1548°F) |
| Boiling Point | About 1484°C (2703°F) |
| Density | 1.55 g/cm³ at 20°C |
| State at Room Temperature | Solid |
| Malleability | Fairly malleable |
| Electrical Conductivity | Good conductor of electricity |
| Solubility | Reacts with water, insoluble in most organic solvents |
Chemical Properties of Calcium
Calcium, with the atomic number 20 and symbol ‘Ca’, is a highly reactive, alkaline earth metal. It’s the fifth most abundant element in the Earth’s crust and is essential for living organisms, particularly in cell physiology.
- Reactivity with Water: Calcium reacts with water, producing calcium hydroxide and hydrogen gas. This reaction is less violent than the corresponding reactions with sodium and potassium.
- Formation of Compounds: Calcium forms a variety of compounds. It readily reacts with oxygen to form calcium oxide (lime) and will also react with sulfur, nitrogen, and the halogens.
- Role in Biological Processes: In biological terms, calcium ions (Ca²⁺) play a crucial role. They serve as a vital messenger in many cellular processes and are necessary for the function of nerves and muscles, blood clotting, and the structure of bones and teeth.
- Ionic Bonds: In its compounds, calcium most often forms ionic bonds. The calcium ion has a charge of +2, forming stable ionic compounds with various anions.
- Electron Configuration: Calcium’s electron configuration (1s² 2s² 2p⁶ 3s² 3p⁶ 4s²) explains its tendency to donate two electrons, forming Ca²⁺ ions in chemical reactions.
- Presence in Minerals: Calcium is a major component of many minerals, including limestone, gypsum, and fluorite, and it plays a significant role in the formation of cave structures and stalactites/stalagmites.
- Use in Reducing Agents: In metallurgy, calcium is used as a reducing agent in the extraction of other metals, such as uranium, zirconium, and thorium.
- Alloy Formation: Calcium is used to make various alloys; for example, with aluminum, it forms a strong, lightweight material.
Thermodynamic Properties of Calcium
| Property | Value with Unit |
|---|---|
| Boiling Point | 1484 °C |
| Melting Point | 842 °C |
| Heat of Vaporization | 154.7 kJ/mol |
| Heat of Fusion | 8.54 kJ/mol |
| Specific Heat Capacity (at 25°C) | 0.647 J/g·K |
| Thermal Conductivity | 201 W/m·K |
| Critical Temperature | Not Available |
Material Properties of Calcium
| Property | Value with Unit |
|---|---|
| Density | 1.55 g/cm³ (at 20°C) |
| Viscosity | Not Applicable (Solid) |
| Solubility in Water | Reacts (Forms Ca(OH)2) |
| Color | Silvery-white |
| Phase at Room Temperature | Solid |
Electromagnetic Properties of Calcium
| Property | Value with Unit |
|---|---|
| Electrical Resistivity (at 20°C) | 3.36 µΩ·m |
| Thermal Conductivity | 201 W/m·K |
| Electronegativity (Pauling scale) | 1.00 |
| First Ionization Energy | 6.1132 eV |
Nuclear Properties of Calcium
| Property | Value with Unit |
|---|---|
| Atomic Number | 20 |
| Atomic Mass | 40.078 amu |
| Isotopes | ^40Ca (96.941%), ^42Ca, ^43Ca, ^44Ca, ^46Ca, ^48Ca |
| Natural Abundance (for ^40Ca) | 96.941% |
| Nuclear Spin (for ^43Ca) | 7/2ℏ |
| Neutron Cross Section (for ^40Ca) | 0.41 barns |
| Nuclear Magnetic Moment (for ^43Ca) | -1.3173 µN |
Chemical Compounds of Calcium
- Calcium Carbonate (CaCO₃)
- Equation: Ca²⁺ + CO₃²⁻ → CaCO₃
- Usage: Found in limestone, marble, chalk; used in cement and antacids.
- Calcium Chloride (CaCl₂)
- Equation: Ca²⁺ + 2Cl⁻ → CaCl₂
- Usage: De-icing roads, desiccant for drying gases and liquids.
- Calcium Hydroxide (Ca(OH)₂)
- Equation: Ca²⁺ + 2OH⁻ → Ca(OH)₂
- Usage: Known as slaked lime; used in mortar, plaster, and pH adjustment.
- Calcium Oxide (CaO)
- Equation: Ca²⁺ + O²⁻ → CaO
- Usage: Also known as quicklime; used in cement, ceramics, and water treatment.
- Calcium Sulfate (CaSO₄)
- Equation: Ca²⁺ + SO₄²⁻ → CaSO₄
- Usage: Found in gypsum; used in plaster, drywall, and fertilizers.
- Calcium Phosphate (Ca₃(PO₄)₂)
- Equation: 3Ca²⁺ + 2PO₄³⁻ → Ca₃(PO₄)₂
- Usage: Major component of bone; used in fertilizers and food industry.
Isotopes of Calcium
| Isotope | Description |
|---|---|
| Calcium-40 | The most abundant isotope, stable, making up about 97% of natural calcium. |
| Calcium-42 | Stable isotope, comprising about 0.6% of natural calcium. |
| Calcium-43 | Stable isotope, used in medical and scientific research. |
| Calcium-44 | Another stable isotope, comprises about 2% of natural calcium. |
| Calcium-48 | The heaviest stable isotope, rare but important in scientific research for its unusual nuclear properties. |
Uses of Calcium

- Bone Health and Medical Uses:
- Calcium is essential for the development and maintenance of strong bones and teeth. It’s used in dietary supplements and is a crucial component in treatments for osteoporosis and other bone-related conditions.
- Agriculture:
- Calcium compounds like calcium nitrate and calcium sulfate are used as fertilizers to provide essential nutrients to plants, promoting growth and fortifying cell walls.
- Construction Materials:
- Calcium carbonate (limestone) and calcium oxide (quicklime) are key ingredients in cement and concrete, providing strength and durability to buildings and infrastructure.
- Food Industry:
- Calcium salts are used as food additives to enhance flavor, act as preservatives, and improve the texture of various foods. Calcium is also a fortifying agent in many food products like dairy alternatives and cereals.
- Water Treatment:
- Calcium hydroxide is used in water treatment processes. It aids in purifying water, adjusting its pH level, and removing impurities.
Commercial Production of Calcium
Calcium is primarily produced through the electrolysis of fused calcium chloride (CaCl₂). The process involves several key steps:
- Preparation of Calcium Chloride:
- Calcium chloride is prepared by reacting calcium carbonate (limestone) with hydrochloric acid or as a by-product of the Solvay process in the manufacturing of sodium carbonate.
- Electrolysis Process:
- The prepared calcium chloride is then subjected to electrolysis. This involves melting the calcium chloride and passing a direct current through it.
- Extraction of Calcium:
- During electrolysis, calcium metal is deposited at the cathode (the negatively charged electrode) while chlorine gas is released at the anode (the positively charged electrode).
- Purification:
- The extracted calcium often contains impurities. It undergoes various purification processes, including distillation, to achieve the desired purity level.
- Final Processing:
- The pure calcium is then processed into different forms, such as ingots, depending on its intended use.
This method is widely used because it produces high-purity calcium. However, it requires high energy inputs, making it an energy-intensive process. The demand for calcium and its compounds in various industries justifies the energy and resource investment in its production.
Health Effects of Calcium
Calcium is crucial for maintaining overall health, but it must be consumed in balanced amounts. Here’s an overview of its health effects:
Positive Health Effects:
- Bone and Teeth Health: Calcium is essential for the development and maintenance of strong bones and teeth. It reduces the risk of osteoporosis and dental issues.
- Blood Clotting: It plays a key role in the blood clotting process, which is vital for healing wounds.
- Muscle Function: Calcium aids in the proper functioning of muscles, including heart muscles, enhancing heart health and muscle movements.
- Nerve Signaling: It is involved in nerve signal transmission, which is crucial for various bodily functions including muscle contraction and hormone secretion.
- Regulating Blood Pressure: Adequate calcium intake has been linked to better blood pressure regulation.
Negative Health Effects (due to Overconsumption or Deficiency):
- Hypercalcemia: Excessive calcium can lead to hypercalcemia, causing nausea, vomiting, confusion, and other severe health issues.
- Kidney Stones: High calcium levels, especially when taken with oxalates, can increase the risk of kidney stones.
- Calcium Deficiency: Insufficient calcium can lead to health issues like osteoporosis, rickets in children, and a higher risk of fractures.
- Cardiovascular Disease: Some studies suggest a link between excessive calcium supplement intake and an increased risk of cardiovascular disease.
Overall, a balanced intake of calcium is vital for health, and its consumption should be carefully managed.
Environmental Effects of Calcium
Calcium in the environment is generally considered non-toxic and is a naturally abundant element. However, its compounds and usage can have various environmental impacts:
- Soil Health: Calcium compounds, like lime, are used to neutralize acidic soils, improving soil health and crop yields. However, excessive use can lead to soil alkalinity, which might be detrimental to some plants.
- Water Bodies: Calcium is crucial for aquatic ecosystems, contributing to water hardness which affects aquatic life. However, excess calcium, often due to runoff from agricultural lands or mining activities, can lead to imbalances in water bodies.
- Air Quality: The production and use of calcium compounds can contribute to air pollution. For instance, cement production releases significant amounts of CO2, a greenhouse gas.
- Ecosystem Balance: Calcium plays a role in maintaining the balance of various ecosystems. It is essential for the growth of plants and aquatic life. However, alterations in calcium levels due to human activities can disrupt these ecosystems.
- Biodiversity: In forests and other ecosystems, calcium levels in the soil contribute to biodiversity. Low calcium levels can lead to reduced species diversity.
What is the Element of Calcium?
Calcium is a chemical element, symbol Ca, atomic number 20, essential for living organisms, particularly in cell physiology and bone formation.
What is Calcium Made From?
Calcium is not made from anything; it’s a naturally occurring element, extracted from minerals like limestone, gypsum, and fluorite.
What is Elemental Calcium Used For?
Elemental calcium is used in the production of cement, steel, and alloys, and is crucial in medical supplements and agricultural lime.
Why is Calcium a Good Element?
Calcium is beneficial due to its essential role in biological processes, including bone health, muscle function, and cellular signaling, and its industrial applications.
Calcium is vital for health, particularly in bone and dental strength. Its presence in various foods and supplements makes it easily accessible. Understanding calcium’s role and ensuring adequate intake are crucial for maintaining overall health and preventing bone-related disorders.