Livermorium (Lv) – Definition, Preparation, Properties, Uses, Compounds, Reactivity
Livermorium, a synthetic element with the symbol Lv, stands as a pinnacle of human ingenuity in the periodic table. This comprehensive guide delves into the depths of Livermorium, offering insightful examples that illuminate its definition, unique properties, practical applications, and intriguing compounds. By exploring the nuances of Livermorium, readers gain a thorough understanding of this enigmatic element, enhancing their knowledge of modern chemistry and its innovative advancements. Join us as we uncover the fascinating world of Livermorium, showcasing its significance in scientific research and potential uses.
What is Livermorium?
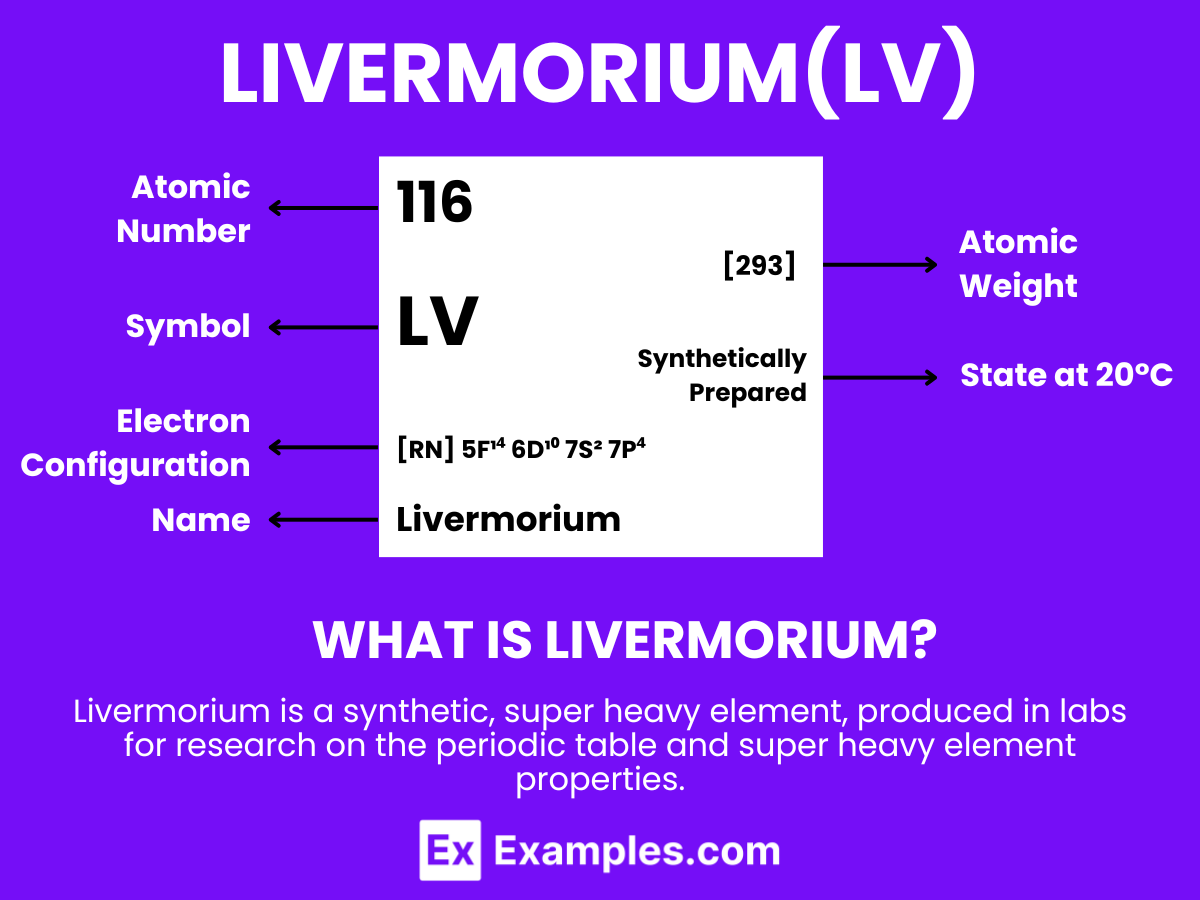
Livermorium is a superheavy, synthetic element with the chemical symbol Lv and atomic number 116. It is notable for being created in particle accelerators by fusing atomic nuclei. Livermorium does not occur naturally and exists only briefly before decaying, making it challenging to study. Its discovery is significant for research in nuclear physics, particularly in understanding the properties and behaviors of superheavy elements in the periodic table. Due to its extreme instability and radioactivity, livermorium has no practical applications outside scientific research, where it contributes to exploring the theoretical “island of stability” and the limits of the periodic table.
Livermorium Formula
- Formula: Lv
- Composition: Consists of a single livermorium atom.
- Bond Type: As a synthetic element, livermorium does not naturally form bonds as it exists only for fractions of a second before decaying. In theoretical compounds, it might form covalent or ionic bonds.
- Molecular Structure: Livermorium, in its elemental form, does not have a traditional molecular structure due to its extreme instability and short half-life. It is not observed in large enough quantities to determine its solid-state structure.
- Electron Sharing: Theoretical predictions suggest that livermorium could share electrons covalently or engage in ionic interactions in compounds, though no stable compounds involving livermorium have been synthesized to confirm these properties.
- Significance: Livermorium’s significance lies primarily in the field of scientific research, particularly in the study of superheavy elements and the boundaries of the periodic table. Its synthesis and decay provide valuable insights into nuclear reactions and the stability of super heavy nuclei.
- Role in Chemistry: Livermorium’s role in chemistry is mainly theoretical and research-based. It offers scientists a deeper understanding of the chemical behaviors of super heavy elements, even though it does not have practical applications due to its short half-life and the challenge in obtaining significant quantities.
Atomic Structure of Livermorium
Livermorium, a synthetic element far removed from the lighter and more commonly encountered elements like hydrogen or gallium, is a super heavy element that occupies a unique position in nuclear chemistry due to its place in the periodic table and its classification as a post-actinide.
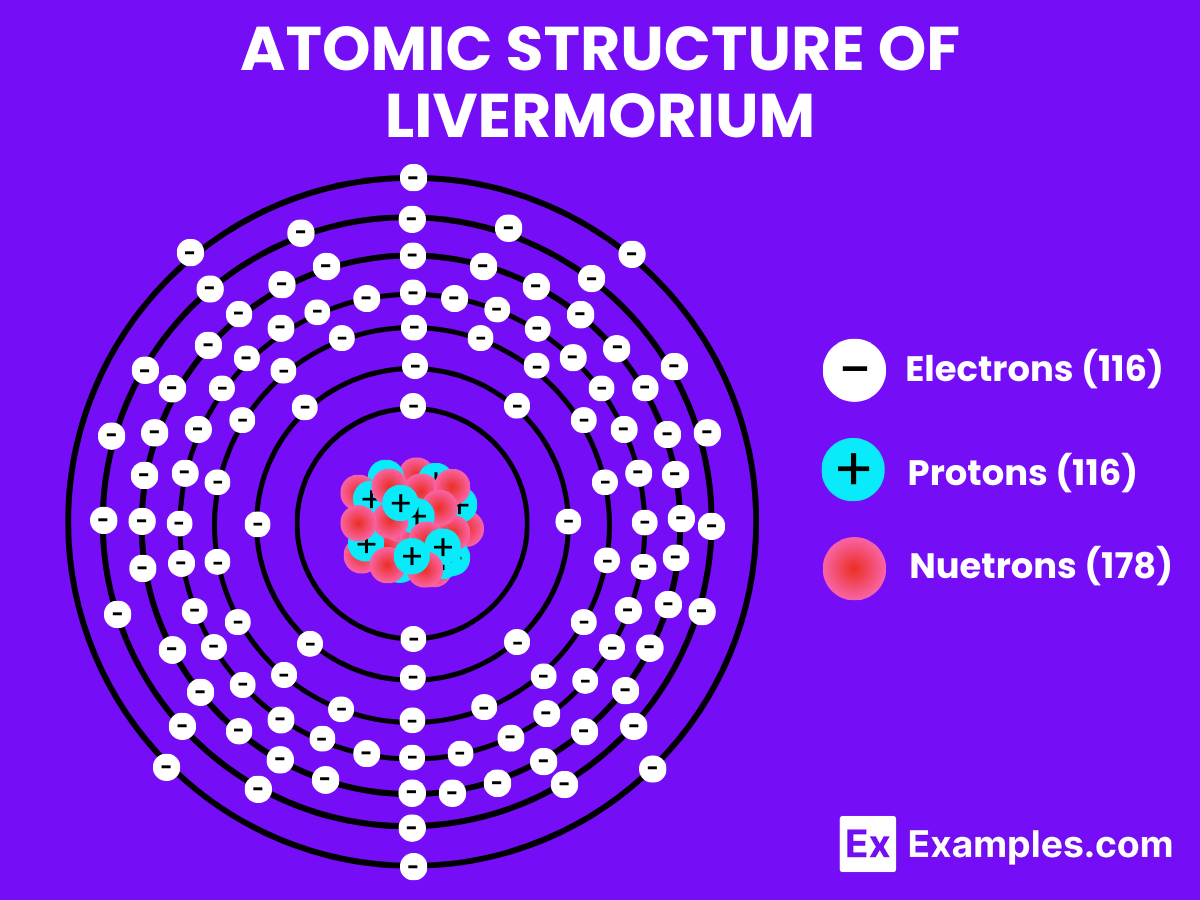
Atomic Level: Each atom of Livermorium (Lv) is characterized by having 116 protons in its nucleus, a feature that defines its atomic number as 116. The theoretical electron configuration of Livermorium is [Rn]5f¹⁴ 6d¹⁰ 7s² 7p⁴ suggesting it has a full 5f and 6d orbital, with four electrons in its 7p orbital, poised for chemical interactions. However, relativistic effects are expected to influence its actual electron configuration, potentially altering its chemical properties.
Molecular Formation: Unlike simpler elements that can form diatomic molecules (such as H₂), Livermorium does not naturally form molecules or exhibit a stable molecular structure due to its extremely short half-life and high instability. The element exists for mere milliseconds before decaying into lighter elements, making the study of its bonding characteristics and molecular formation largely theoretical. In the hypothetical scenario where Livermorium atoms could persist long enough to interact chemically, their behavior would likely be influenced by their electron configuration, but this remains speculative.
The stability and phase of Livermorium under various temperatures and pressures are subjects of theoretical speculation, as its brief existence precludes the observation of solid, liquid, or gaseous states under normal conditions. The term “Livermorium Gas” does not apply in the same way it might for compounds like uranium hexafluoride (UF₆) in uranium’s context; Livermorium’s extreme radioactivity and rapid decay mean it has not been observed in compound form or any state other than as fleeting, individual atoms produced in particle accelerators.
Properties of Livermorium
Physical Properties of Livermorium
| Property | Description |
|---|---|
| Appearance | Not observed directly; assumed to have no stable or long-lasting physical form due to its radioactivity |
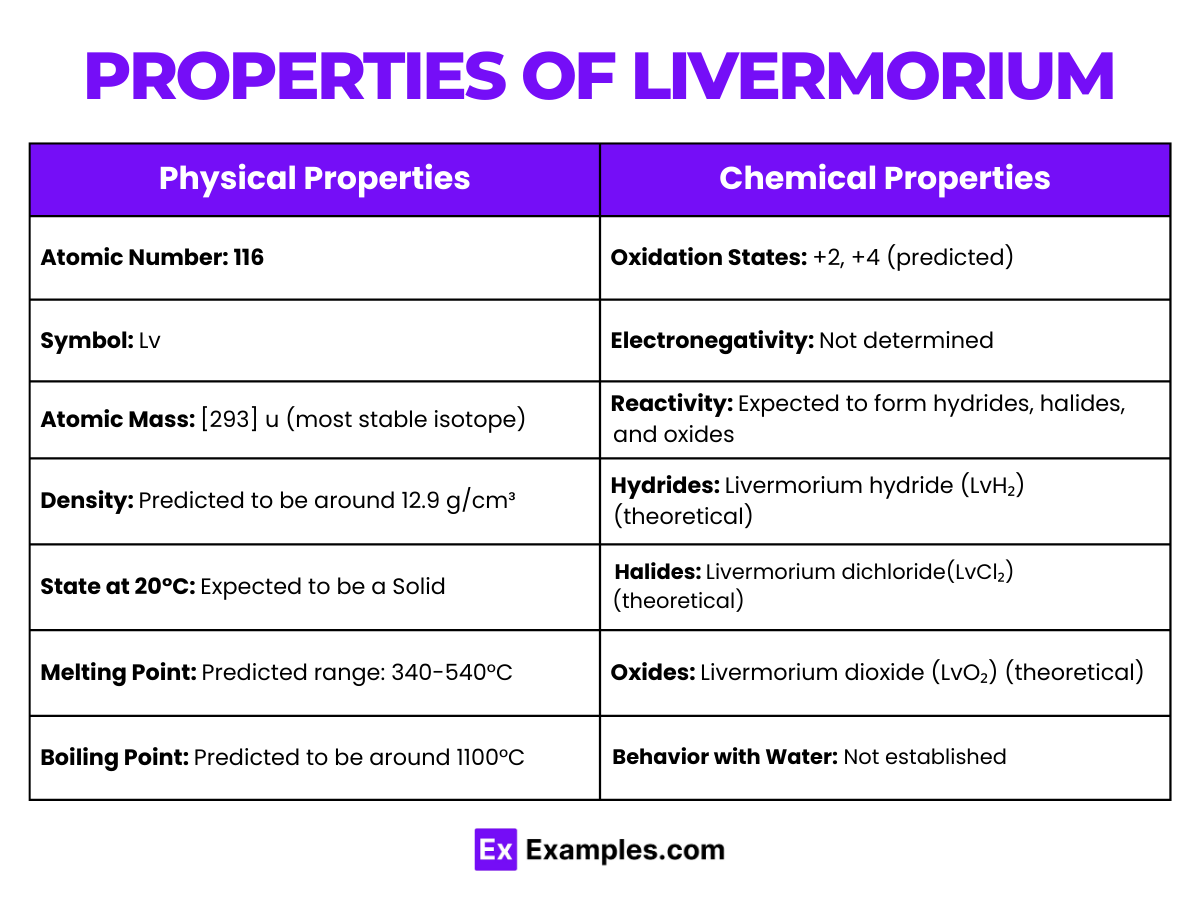
| Atomic Number | 116 |
| Density (at 20°C) | 12.9 g/cm³ |
| Melting Point | 340-540°C |
| Boiling Point | 100°C |
| State at Room Temperature | Expected to be solid |
| Electron Configuration | [Rn] 5f¹⁴ 6d¹⁰ ₇s² ₇p⁴ |
| Common Oxidation States | +2, +4 |
Chemical Properties of Livermorium
Livermorium, with the atomic number 116, is a synthetic element located in group 16 of the periodic table. Its chemical properties are primarily theoretical due to its extremely short half-life and the challenges associated with its production. Here, we explore the anticipated chemical properties of livermorium, based on its position in the periodic table and theoretical predictions.
Oxidation States
- Livermorium is expected to show oxidation states of +2 and +4, with the +2 state being more stable. This pattern is consistent with the heavier elements in group 16, where lower oxidation states are favored due to relativistic effects.
Predicted Halides
- Livermorium(II) Chloride (LvCl₂): Likely to be volatile and more stable in the +2 oxidation state.
- Livermorium(II) Bromide (LvBr₂) and Livermorium(II) Iodide (LvI₂): These halides are also predicted to follow the trend of increased volatility compared to their lighter counterparts.
Oxides
- Livermorium Dioxide (LvO₂): This compound is expected to be structurally similar to polonium dioxide (PoO₂), favoring the +4 oxidation state.
- Livermorium Monoxide (LvO): The formation of this oxide is less certain but could occur, reflecting the element’s ability to exhibit different oxidation states.
Hydrides
- Livermorium Hydride (LvH₂): Anticipated to be analogous to polonium hydride (PoH₂), exhibiting characteristics common to group 16 hydrides.
Chalcogenides
- Although specific formulas and properties are speculative, livermorium could potentially form chalcogenides with other group 16 elements, showcasing its versatility within the group.
Relativistic Effects
- The chemical behavior of livermorium is notably influenced by relativistic effects, altering its electron orbitals and potentially leading to unique chemical properties not observed in lighter group 16 elements.
Experimental Limitations
- The synthesis of livermorium and its compounds faces significant challenges due to the element’s radioactivity and short half-life, making experimental verification of its chemical properties difficult.
The exploration of livermorium’s chemical properties remains largely theoretical, awaiting advancements in experimental techniques and the production of more stable isotopes for in-depth study.
Thermodynamic Properties of Livermorium
| Property | Description |
|---|---|
| Standard State | Solid (expected) |
| Melting Point | 340-540°C (estimated range based on theoretical calculations) |
| Boiling Point | Approximately 1100°C (predicted) |
Material Properties of Livermorium
| Property | Value with Unit |
|---|---|
| Density (at 20°C) | Estimated to be around 12.9 g/cm³(theoretical) |
| Viscosity | Not Applicable (Solid, theoretical) |
| Solubility in Water | Insoluble (theoretical) |
| Color | Not observed directly; no stable or visible appearance due to rapid decay |
| Phase at Room Temperature | Expected to be solid (theoretical) |
Nuclear Properties of Livermorium
| Property | Value with Unit |
|---|---|
| Atomic Number | 116 |
| Atomic Mass | Most stable isotope: Livermorium-293 (293 u) |
| Isotopes | ^293Lv (most stable), ^292Lv, ^291Lv, among others |
| Half-Life (for ^293Lv) | ~60 milliseconds |
| Half-Life (for ^292Lv) | ~20 milliseconds |
| Half-Life (for ^291Lv) | ~18 milliseconds |
| Nuclear Spin | Not precisely determined due to short half-lives |
| Neutron Cross Section | Not determined (extremely short-lived isotopes make measurement challenging) |
Preparation of Livermorium
Livermorium is a super heavy, synthetic element that is not found naturally and can only be created in a laboratory setting. The preparation of livermorium involves highly specialized equipment, advanced nuclear reactors, and ion accelerators. Below is an outline of the general process used to create livermorium:
- Selection of Target and Projectile:
- The preparation of livermorium typically involves a nuclear reaction between a lighter element (the projectile) and a heavier element (the target).
- A common combination used is curium (Cm) as the target and calcium (Ca) as the projectile.
- Acceleration:
- The calcium ions are accelerated to high speeds using a particle accelerator. This process imparts the necessary kinetic energy to the calcium ions to overcome the electrostatic repulsion between the target and the projectile nuclei.
- Collision and Fusion:
- The accelerated calcium ions are directed towards a thin layer of curium. When these ions collide with the curium atoms, they can fuse together, resulting in the formation of a heavier, compound nucleus. This process is known as hot fusion.
- Nucleus Cooling and Decay:
- The compound nucleus thus formed is usually in an excited state and tends to lose its excess energy by emitting one or more neutrons, cooling down to a more stable state. This process may lead to the formation of livermorium.
- Detection and Identification:
- The newly formed livermorium atoms are highly unstable and undergo radioactive decay in a very short time. Researchers detect and identify these atoms by observing their decay products and patterns, often using detectors that are sensitive to alpha particles, gamma rays, and spontaneous fission events.
- Isolation of Isotopes:
- Various isotopes of livermorium can be produced depending on the specific nuclear reaction and the energy involved in the process. The identification of isotopes is crucial for studying the chemical and physical properties of livermorium.
Chemical Compounds of Livermorium
-
- Livermorium Oxide Formation
- Description: A theoretical compound suggesting the interaction between livermorium and oxygen to form an oxide.
- Equation: 2Lv+O₂→LvO₂
- Livermorium Fluoride Reactivity
- Description: Predicts the formation of a fluoride compound when livermorium reacts with fluorine.
- Equation: 2Lv+F₂→LvF₂
- Livermorium Chloride Interaction
- Description: Suggests the possibility of livermorium combining with chlorine to form a chloride compound.
- Equation:2Lv+Cl₂→LvCl₂
- Livermorium Bromide Chemistry
- Description: Indicates the theoretical reaction between livermorium and bromine to produce a bromide.
- Equation:2Lv+Br₂→LvBr₂
- Livermorium Iodide Inference
- Description: Inference about livermorium’s ability to react with iodine to form an iodide compound.
- Equation:2Lv+I₂→LvI₂
- Livermorium Hydride Speculation
-
- Description: Speculates on the reaction between livermorium and hydrogen to create a hydride.
- Equation:2Lv+H₂→LvH₂
-
- Livermorium Oxide Formation
Isotopes of Livermorium
Livermorium is a synthetic element with no stable isotopes. It has several isotopes that have been identified through nuclear reactions, each exhibiting unique decay properties.
| Isotope | Half-Life | Decay Mode |
|---|---|---|
| Lv-290 | 8 milliseconds | Alpha decay to Fl-286 |
| Lv-291 | ~18 milliseconds | Alpha decay to Fl-287 |
| Lv-292 | ~20 milliseconds | Alpha decay to Fl-288 |
| Lv-293 | ~60 milliseconds | Alpha decay to Fl-289 |
| Lv-294 | ~40 milliseconds | Alpha decay to Fl-290 |
| Lv-295 | Predicted, not observed | Predicted alpha decay |
| Lv-296 | Predicted, not observed | Predicted alpha decay |
Uses of Livermorium
Livermorium is a synthetic, superheavy element with the atomic number 116. Due to its extremely short half-life and the fact that it can only be produced in minute quantities. Below are the potential uses and areas of interest related to livermorium:
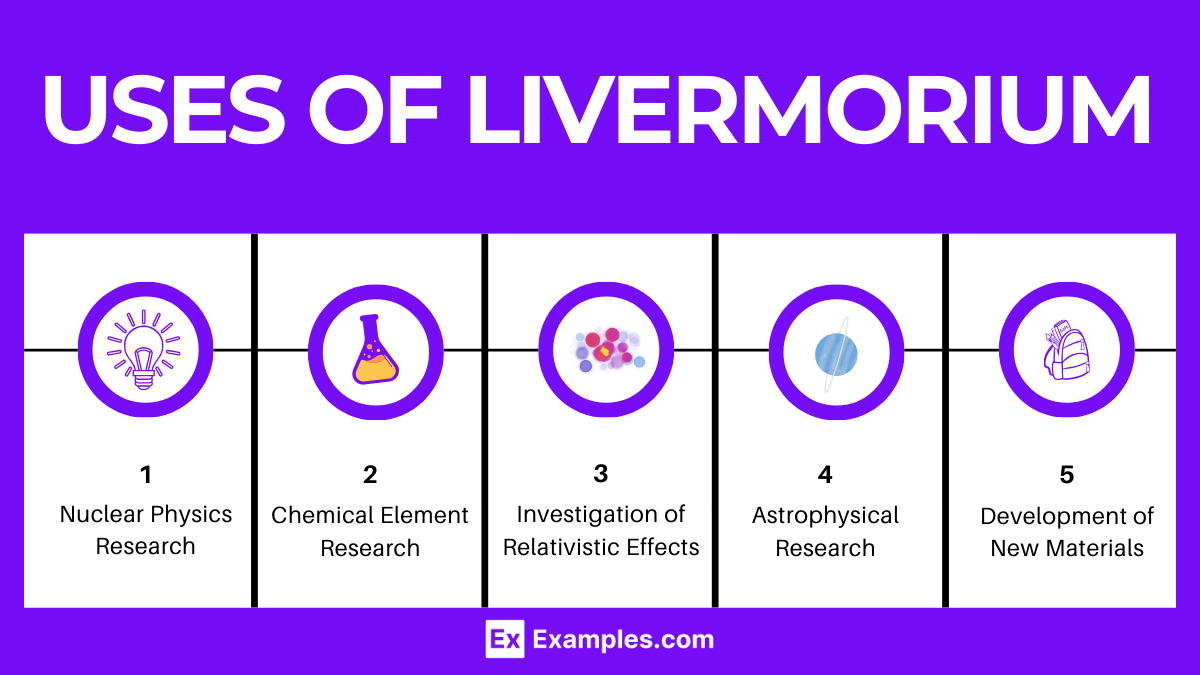
- Nuclear Physics Research:
- Livermorium plays a crucial role in the study of nuclear physics, particularly in understanding the properties of superheavy elements. Researchers study livermorium’s decay patterns, half-life, and the possibility of reaching the “island of stability,” a theoretical region where superheavy elements might have longer half-lives.
- Chemical Element Research:
- Though livermorium’s chemical properties are largely unknown due to its instability, theoretical studies and comparison with lighter homologs provide insights into the chemical behavior of superheavy elements. Research in this area expands our knowledge of the periodic table and chemical bonding.
- Investigation of Relativistic Effects:
- The study of livermorium and other super heavy elements allows scientists to investigate relativistic effects on electrons. These effects become significant in elements with very high atomic numbers and can alter chemical and physical properties in unexpected ways.
- Astrophysical Research:
- While livermorium is not found naturally and is unlikely to exist in observable quantities in the universe, its synthesis and study can contribute to astrophysical models, particularly those related to the process of nucleosynthesis in extreme cosmic environments.
- Development of New Materials and Technologies:
- Although livermorium itself is not directly used in developing new materials or technologies, the techniques developed for its synthesis and study can lead to advancements in particle accelerator technology and nuclear chemistry.
Production of Livermorium
Livermorium, with the symbol Lv and atomic number 116, is a synthetic element produced in extremely small amounts through nuclear reactions involving heavy ions.
- Selection of Target and Projectile Materials:
- The production of livermorium typically involves bombarding a heavy target element with lighter ions. A common choice for the target is curium (Cm), specifically isotopes like Cm-248, while the projectile often involves calcium (Ca) ions, such as Ca-48.
- Acceleration of Projectile Ions:
- The calcium ions are accelerated to high velocities using a particle accelerator. This acceleration is crucial for overcoming the electrostatic repulsion between the projectile and the target nuclei.
- Collision and Fusion:
- The accelerated calcium ions collide with the curium target. Under the right conditions, a calcium ion can fuse with a curium nucleus, forming a compound nucleus that is an excited state of the new element, livermorium.
- Nuclear Decay:
- The compound nucleus may lose its excess energy by emitting one or more neutrons, resulting in the formation of a livermorium isotope.
- Detection and Identification:
- The newly formed livermorium nuclei are highly unstable and decay rapidly. Detection systems equipped with devices for tracking alpha decay, spontaneous fission, or gamma radiation are used to identify the presence of livermorium and to study its properties.
- Challenges in Production:
- The synthesis of livermorium is a challenging process, with low production rates and the difficulty of isolating the element due to its rapid decay. The success rate of producing livermorium atoms is extremely low, making it one of the most challenging elements to study.
Applications of Livermorium
Livermorium is a synthetic element with the atomic number 116, identified by its symbol, Lv.
- Scientific Research:
- The primary application of livermorium is in scientific research, particularly in nuclear physics and chemistry. Studies focus on investigating the element’s nuclear stability, decay patterns, and potential chemical properties, contributing valuable data to theoretical models of super heavy elements.
- Periodic Table and Elemental Behavior:
- Research on livermorium helps scientists understand the behavior of elements in the periodic table’s extreme regions. Insights gained from studying livermorium and its isotopes enhance knowledge of the periodic table’s structure and the forces that govern elemental properties.
- Search for the Island of Stability:
- Livermorium’s synthesis and study are part of the broader scientific quest to find the “island of stability,” a theoretical group of super heavy elements that are hypothesized to have relatively long half-lives. Discovering such elements would be a significant breakthrough in nuclear physics, potentially leading to new materials with novel properties.
- Advancements in Nuclear Chemistry and Physics:
- The techniques developed to produce and detect livermorium and other super heavy elements drive technological and methodological advancements in nuclear chemistry and physics. These advancements can have broader applications, including in medical imaging, radiation therapy, and materials science.
- Educational and Theoretical Significance:
- Livermorium’s discovery and the challenges associated with its study serve as an educational tool, illustrating the complexities of chemical synthesis, nuclear stability, and the theoretical limits of the periodic table. Its study aids in refining theoretical models of atomic structure and elemental behavior.
livermorium is a synthetic, super heavy element marked by its elusive nature and scarcity of data. Primarily used for scientific research, its synthesis offers insights into the periodic table’s limits and nuclear physics. Despite lacking practical applications, livermorium’s study propels advancements in understanding super heavy elements and contributes to the quest for the island of stability.