Oganesson (Og) – Definition, Preparation, Properties, Uses, Compounds, Reactivity
Dive into the enigmatic world of Oganesson, a synthetic chemical marvel that sits at the pinnacle of the periodic table. This complete guide demystifies the essence of Oganesson, providing insightful examples that illuminate its properties, uses, and the fascinating science behind its creation. Uncover the intriguing role of Oganesson in advancing chemical research and its potential implications for future scientific breakthroughs. Engage with a keyword-rich narrative designed for optimal SEO and NLP compatibility, ensuring a comprehensive understanding of this extraordinary element.
What is Oganesson?
Oganesson is a superheavy, synthetic element with the chemical symbol Og and atomic number 118. It is known for being produced in particle accelerators through the fusion of atomic nuclei. Oganesson does not occur in nature and has a very short lifespan before it decays, which presents challenges for its study. The element’s discovery is crucial for nuclear physics research, especially in probing the properties and behaviors of superheavy elements in the periodic table. Because of its significant instability and radioactivity, oganesson has no practical applications beyond scientific inquiry, where it plays a role in investigating the conjectural “island of stability” and the boundaries of the periodic table.
Oganesson Formula
- Formula: Og
- Composition: Consists of a single oganesson atom.
- Bond Type: As a synthetic element, oganesson does not naturally form bonds as it exists only for extremely short durations before decaying. In theoretical compounds, it might form covalent or ionic bonds, though its full chemical behavior is largely unknown due to its instability.
- Molecular Structure: Oganesson, in its elemental form, does not have a traditional molecular structure due to its extreme instability and short half-life. It is not observed in large enough quantities to determine its solid-state structure or how it would interact in a molecular environment.
- Electron Sharing: Theoretical predictions suggest that oganesson could potentially share electrons covalently or engage in ionic interactions in compounds. However, no stable compounds involving oganesson have been synthesized, and its full range of chemical properties remains speculative.
- Significance: Oganesson’s significance is primarily in the realm of scientific research, particularly in exploring the limits of the periodic table and the properties of superheavy elements. Its synthesis and subsequent decay provide insights into nuclear physics, the behavior of superheavy elements, and the theoretical boundaries of element stability.
- Role in Chemistry: Oganesson’s role in chemistry is largely theoretical and centered on research. It offers insights into the potential chemical behaviors and properties of superheavy elements, despite the fact that it has no practical applications due to its very short half-life and the challenges associated with producing it in observable quantities.
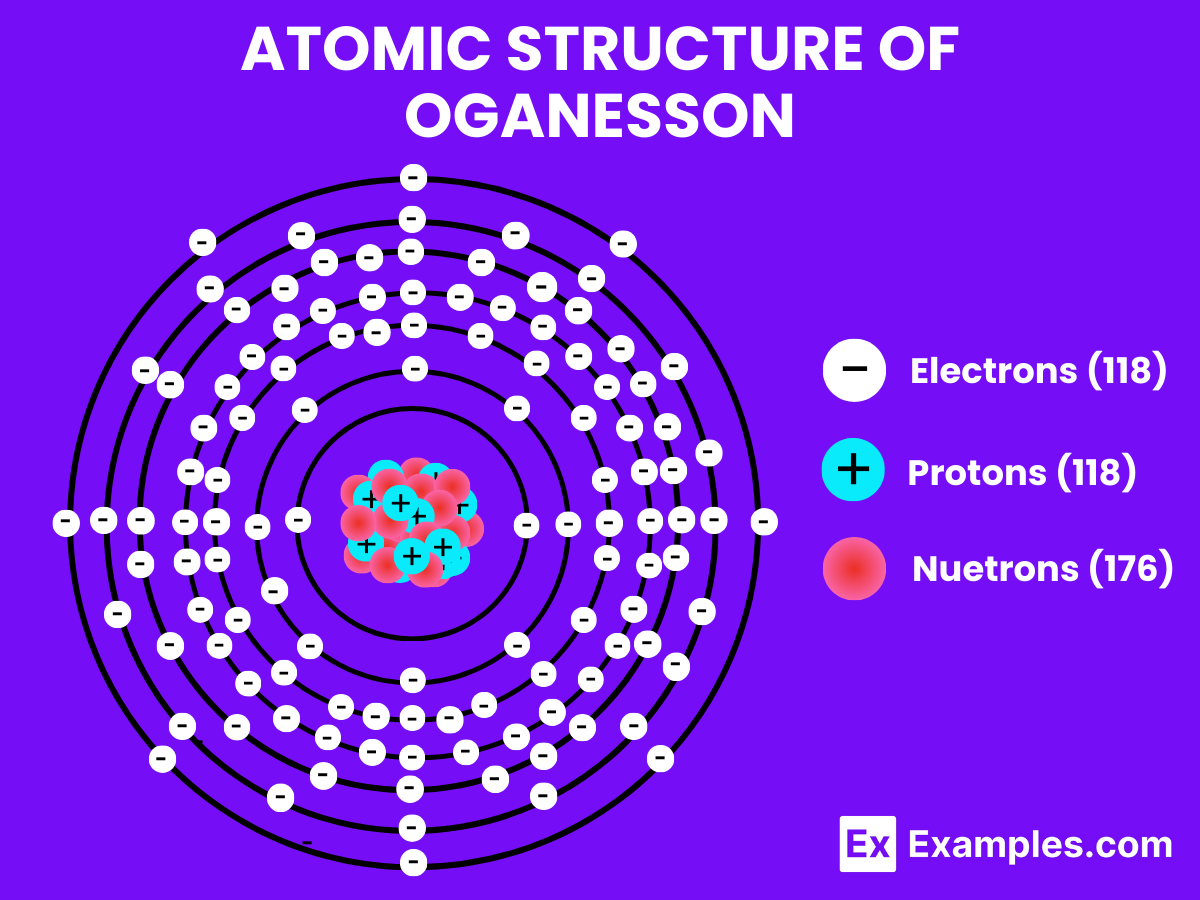
Atomic Structure of Oganesson
Oganesson, a synthetic element that stands out significantly from lighter, more commonly encountered elements such as hydrogen or gallium, is a superheavy element that occupies a unique position in nuclear chemistry due to its placement in the periodic table and its categorization as a noble gas in the post-actinide series.
Atomic Level: Each atom of Oganesson (Og) is characterized by having 118 protons in its nucleus, defining its atomic number as 118. The theoretical electron configuration of Oganesson is [Rn]5f¹⁴ 6d¹⁰ 7s² 7p⁶, indicating it has a full 5f and 6d orbital, with six electrons in its 7p orbital, setting the foundation for chemical interactions. However, relativistic effects are anticipated to significantly influence its actual electron configuration, potentially altering its chemical properties.
Molecular Formation: Unlike simpler elements that can form diatomic molecules (such as H₂), Oganesson does not naturally form molecules or exhibit a stable molecular structure due to its extremely short half-life and high instability. The element exists for only milliseconds before decaying into lighter elements, making the study of its bonding characteristics and molecular formation largely theoretical. In the hypothetical scenario where Oganesson atoms could persist long enough to interact chemically, their behavior would likely be influenced by their electron configuration, but this remains speculative.
The stability and phase of Oganesson under various temperatures and pressures are subjects of theoretical speculation, as its brief existence precludes the observation of solid, liquid, or gaseous states under normal conditions. The term “Oganesson Gas” does not apply in the same way it might for compounds like uranium hexafluoride (UF₆) in the context of uranium.
Properties of Oganesson
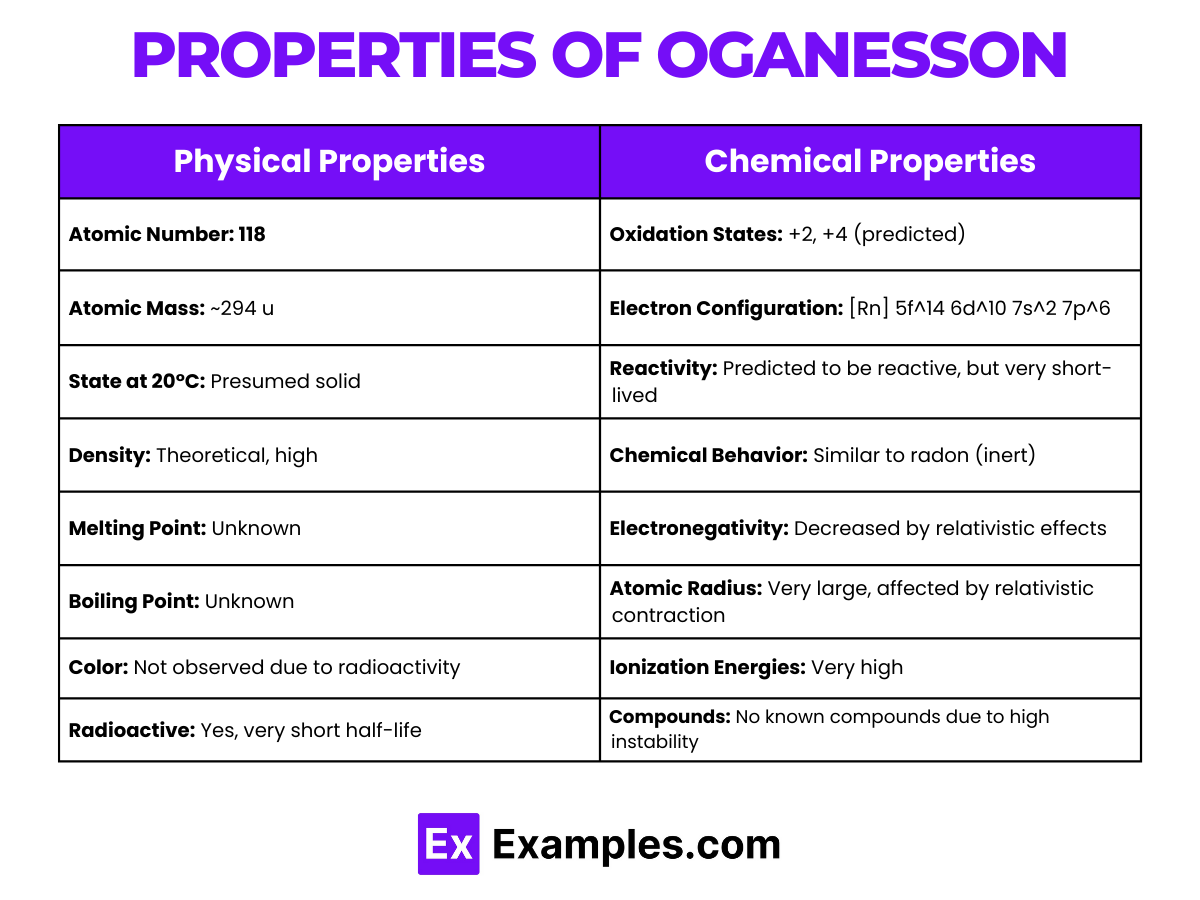
Physical Properties of Oganesson
| Property | Description |
|---|---|
| Appearance | Not observed directly; presumed to have no stable form due to extreme radioactivity |
| Atomic Number | 118 |
| Density (at 20°C) | 13.65 g·cm³ |
| Melting Point | Unknown; expected to be high based on periodic trends (theoretical) |
| Boiling Point | Unknown; predicted to be high, specific values not estimated (theoretical) |
| State at Room Temperature | Expected to be solid (based on theoretical calculations) |
| Electron Configuration | [Rn] 5f¹⁴ 6d¹⁰ 7s² 7p⁶ |
| Common Oxidation States | +2, +4 (predicted, but not co |
Chemical Properties of Oganesson
Oganesson, with the atomic number 118, is a synthetic element located in group 18 of the periodic table.
- States: Oganesson is anticipated to exhibit oxidation states of 0, +2, +4, +6, and possibly +8, diverging from the typically inert nature of other group 18 elements. The +6 state, although speculative, underscores the element’s unique position, where relativistic effects enable a broader range of chemical behavior compared to its lighter noble gas counterparts.
- Predicted Compounds: As a member of group 18, Oganesson is a noble gas, traditionally expected to be chemically inert. However, its theoretical ability to form compounds, such as with highly electronegative elements or metals, would challenge the conventional understanding of noble gases due to relativistic effects enhancing its chemical reactivity.
- Oxides: Oganesson oxides, if they could exist, would represent a significant deviation from the norm for noble gases, which typically do not form stable oxides. Such compounds would likely exhibit extraordinary properties, influenced by Oganesson’s unique electron configuration and the impact of relativistic effects.
- Hydrides: The hypothetical formation of Oganesson hydride (OgH) would be groundbreaking, suggesting possible reactivity patterns that differ markedly from the inertness associated with lighter noble gases. This compound would provide invaluable insights into the influence of relativistic effects on chemical bonding in superheavy elements.
- Noble Gas Compounds: Oganesson may be capable of forming noble gas compounds with other elements, including possible intergroup reactions that defy the inert nature traditionally ascribed to group 18 elements. These compounds would likely showcase a range of stoichiometries and properties, reflecting the theoretical oxidation states of Oganesson.
- Relativistic Effects: The chemical behavior of Oganesson is profoundly affected by relativistic effects, which modify its electron orbitals and lead to potential chemical properties distinct from those of its group 18 predecessors.
- Experimental Challenges: The synthesis and study of Oganesson and its compounds are hindered by the element’s extreme radioactivity and brief half-life. These factors complicate the experimental observation of its chemical properties, limiting current knowledge to theoretical predictions.
Investigating the chemical properties of Oganesson remains a largely speculative endeavor, dependent on future advancements in experimental methods and the synthesis of more stable isotopes for comprehensive analysis.
| Property | Value with Unit |
|---|---|
| Atomic Number | 118 |
| Atomic Mass | Most stable isotope: Oganesson-294 (294 u) |
| Isotopes | Various, including ^294Og (predicted to be the most stable) |
| Half-Life (for ^294Og) | Less than a millisecond (estimated) |
| Nuclear Spin | Not precisely determined due to short half-lives |
| Neutron Cross Section | Not determined (extremely short-lived isotopes make measurement challenging) |
Preparation of Oganesson
Oganesson is a superheavy, synthetic element that does not occur naturally and can only be synthesized in a laboratory setting. The preparation of oganesson involves highly specialized equipment, including advanced nuclear reactors and ion accelerators. Here is an outline of the general process used to create oganesson:
Selection of Target and Projectile:
- The preparation of oganesson typically involves a nuclear reaction between a lighter element (the projectile) and a heavier element (the target). A common combination used is californium (Cf) as the target and calcium (Ca) as the projectile.
Acceleration:
- The calcium ions are accelerated to high speeds using a particle accelerator. This process imparts the necessary kinetic energy to the calcium ions to overcome the electrostatic repulsion between the nuclei of the target and projectile.
Collision and Fusion:
- The accelerated calcium ions are directed towards a thin layer of californium. Upon colliding with the californium atoms, they can fuse together, resulting in the formation of a heavier, compound nucleus. This process is known as hot fusion.
Nucleus Cooling and Decay:
- The compound nucleus thus formed is usually in an excited state and tends to lose its excess energy by emitting one or more neutrons, cooling down to a more stable state. This process may lead to the formation of oganesson.
Detection and Identification:
- The newly formed oganesson atoms are highly unstable and undergo radioactive decay in a very short time. Researchers detect and identify these atoms by observing their decay products and patterns, often using detectors that are sensitive to alpha particles, gamma rays, and spontaneous fission events.
Isolation of Isotopes:
- Various isotopes of oganesson can be produced depending on the specific nuclear reaction and the energy involved in the process. Identifying these isotopes is crucial for studying the chemical and physical properties of oganesson.
Chemical Compounds of Oganesson
-
Oganesson Oxide Formation
A theoretical compound suggesting the interaction between oganesson and oxygen to form an oxide.
Equation: 2Og + O₂ → OgO₂
-
Oganesson Fluoride Reactivity
Predicts the formation of a fluoride compound when oganesson reacts with fluorine.
Equation: 2Og + F₂ → OgF₂
-
Oganesson Chloride Interaction
Suggests the possibility of oganesson combining with chlorine to form a chloride compound.
Equation: 2Og + Cl₂ → OgCl₂
-
Oganesson Bromide Chemistry
Indicates the theoretica₂ l reaction between oganesson and bromine to produce a bromide.
Equation: 2Og + Br₂ → OgBr₂
-
Oganesson Iodide Inference
Inference about oganesson’s ability to react with iodine to form an iodide compound.
Equation: 2Og + I₂ → OgI₂
-
Oganesson Hydride Speculation
Speculates on the reaction between oganesson and hydrogen to create a hydride.
Equation: 2Og + H₂ → OgH₂
Isotopes of Oganesson
Oganesson is a synthetic element with no stable isotopes. Its isotopes have been created in laboratory settings through nuclear reactions, showcasing distinct decay characteristics.
| Isotope | Half-Life | Decay Mode |
|---|---|---|
| Og-293 | Less than 0.69 ms | Alpha decay to Lv-289 |
| Og-294 | Predicted, not observed | Predicted alpha decay |
| Og-295 | Predicted, not observed | Predicted alpha decay |
| Og-296 | Predicted, not observed | Predicted alpha decay to Lv-292 |
| Og-297 | Predicted, not observed | Predicted alpha decay |
| Og-298 | Predicted, not observed | Predicted alpha decay |
| Og-299 | Predicted, not observed | Predicted alpha decay |
Uses of Oganesson
- Frontier of Scientific Research: Oganesson’s synthesis pushes the boundaries of chemistry, enhancing our understanding of atomic structure and the limits of the periodic table.
- Insights into Relativistic Effects: Studying Oganesson helps scientists explore the effects of relativistic physics on superheavy elements, contributing to advancements in theoretical chemistry and quantum mechanics.
- Potential for Future Technologies: While currently speculative, the knowledge gained from Oganesson could inform the development of new materials with unique properties, potentially impacting electronics, energy, and materials science.
- Contribution to Nuclear Science: Oganesson’s production and decay offer valuable insights into nuclear reactions and decay processes, aiding in the advancement of nuclear physics models and potentially influencing nuclear energy and safety.
- Symbol of Human Curiosity: Oganesson represents the pinnacle of human ingenuity in the pursuit of scientific knowledge, showcasing our ability to explore and understand the most elusive aspects of the natural world.
Production of Oganesson
Oganesson, element 118, is the heaviest element currently recognized by the International Union of Pure and Applied Chemistry (IUPAC).
- Synthetic Creation: Oganesson is synthesized through a meticulously controlled process of nuclear fusion, where lighter elements are combined under extreme conditions to form this superheavy element.
- Nuclear Fusion Reactions: The most common method involves bombarding a target material, usually made of a lighter element like californium (Cf), with accelerated ions of another lighter element, such as calcium (Ca). The fusion of these atomic nuclei leads to the formation of Oganesson.
- Precursor Elements: Typically, the production process utilizes isotopes of californium (Cf) as the target and isotopes of calcium (Ca) as the projectile. For example, the reaction has been used to synthesize Oganesson, where “3n” denotes the release of three neutrons.
- Experimental Facilities: The synthesis of Oganesson requires state-of-the-art nuclear research facilities equipped with powerful particle accelerators, capable of achieving the immense energies necessary for initiating and sustaining the nuclear fusion process.
- Challenges and Limitations: The production of Oganesson is an exceedingly rare and complex process, limited by the availability of target materials, the efficiency of particle accelerators, and the element’s extremely short half-life, which challenges detection and study.
Applications of Oganesson
Oganesson, with atomic number 118, represents one of the latest frontiers in the exploration of the periodic table. Due to its position as a superheavy element and its incredibly short half-life, practical applications of Oganesson outside theoretical research are yet to be realized. However, its discovery and the ongoing investigations into its properties hold significant implications across various scientific domains:
- Theoretical Chemistry and Physics: The primary application of Oganesson currently lies in the realm of theoretical research. Studying its properties helps scientists deepen their understanding of quantum mechanics, nuclear physics, and the behavior of superheavy elements. These insights are pivotal for refining the theoretical models that describe atomic structure and stability.
- Advancement in Material Science: Though direct applications are speculative at this stage, the study of Oganesson could eventually influence material science. Understanding the properties of superheavy elements might lead to the theoretical design of new materials with unique electronic, magnetic, or chemical properties, opening new pathways for innovation in technology and industry.
- Nuclear Research: Oganesson’s synthesis and decay processes offer valuable data for nuclear research, contributing to the development of safer and more efficient methods of nuclear energy production and waste management. Insights gained from studying its radioactive decay could improve models of nuclear stability and fission processes.
- Exploration of the Periodic Table’s Limits: Oganesson stands at the edge of the known periodic table, and its study challenges and expands our understanding of the periodic trends and the limits of chemical element stability. This could lead to the discovery of even heavier elements and the potential expansion of the periodic table.
- Educational and Inspirational Role: The discovery and exploration of Oganesson serve as powerful educational tools, inspiring curiosity and interest in the natural sciences. Its production is a testament to human ingenuity and the relentless pursuit of knowledge, underscoring the importance of scientific research and collaboration.
Oganesson stands as a monumental achievement in the field of chemistry, symbolizing the zenith of human curiosity and scientific exploration. Though its practical applications remain speculative, its discovery pushes the boundaries of the periodic table and enhances our understanding of the atomic world. Oganesson’s study promises to inspire future scientific breakthroughs, underscoring the endless pursuit of knowledge.