Ozone (O₃) – Definition, Structure, Preparation, Uses, Benefits, Side Effects
Ozone is a special kind of gas. This bond forms what scientists call a covalent compound, which means the atoms share electrons to stay connected. In the world of chemistry, ozone plays a big role in protecting our planet by absorbing the sun’s harmful rays high up in the atmosphere. This gas is not only crucial for life on Earth but also fascinating to study because of its unique properties and the way it interacts with other elements in the air.
What is Ozone ?
Ozone (O₃) is a type of gas found in the Earth’s atmosphere, made up of three oxygen atoms linked together. Unlike the oxygen we breathe, which has two oxygen atoms, ozone has an extra oxygen atom that makes it quite special. This unique structure allows ozone to absorb most of the sun’s harmful ultraviolet rays, providing a protective layer high above us that shields all living things from the sun’s intense energy. In simple terms, ozone acts like the Earth’s sunscreen, playing a crucial role in keeping our planet and its inhabitants safe.
Chemical Names and Formulas
| Property | Value |
|---|---|
| Formula | O₃ |
| Name | ozone |
| Alternate Names | triatomic oxygen, trioxygen |
Structure Of Ozone(O₃)
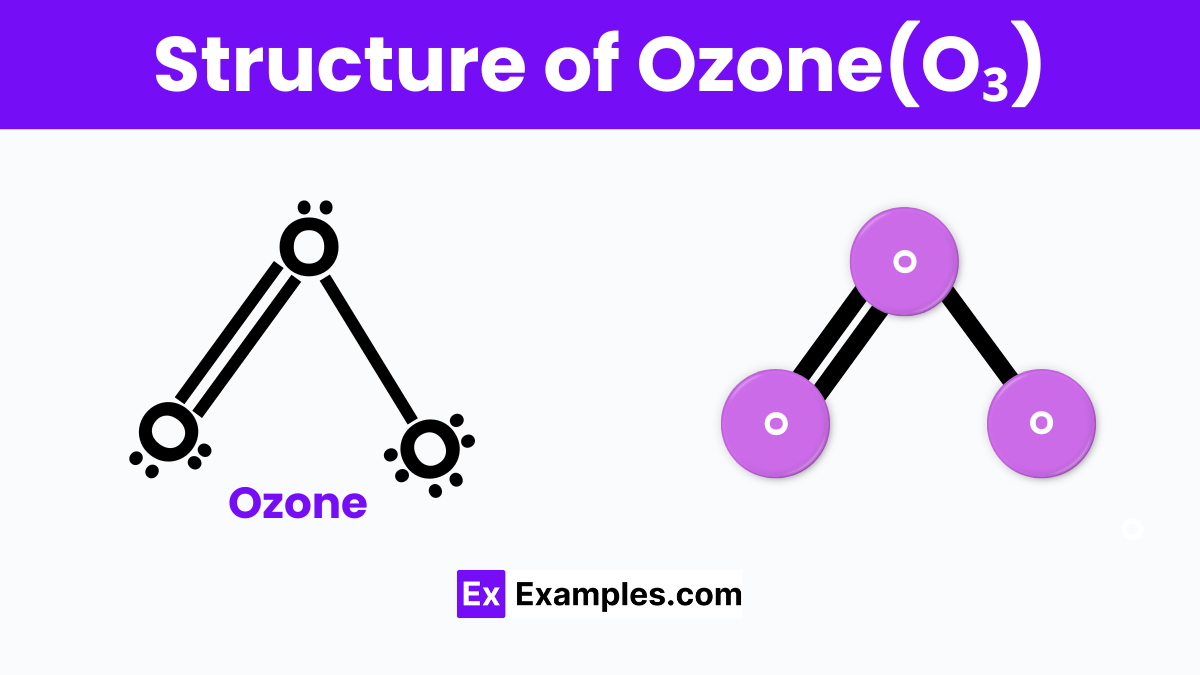
Ozone, or O₃, is a type of oxygen molecule that’s different from the oxygen we breathe, which has two oxygen atoms (O₂). In an ozone molecule, three oxygen atoms are linked together in a way that forms a bent shape. Imagine a boomerang, where the middle oxygen atom is at the tip of the boomerang, and the other two atoms are at the ends. This shape is because of the way electrons are shared between the oxygen atoms, making it a little bit like a stretched-out triangle.
This bent shape of ozone is really important for how it behaves in our atmosphere, especially in the ozone layer, which protects us from the sun’s harmful UV rays. Unlike regular oxygen, ozone can absorb these UV rays, acting like a shield for the Earth. The unique structure of ozone, with its three oxygen atoms, makes all this possible, helping to keep our planet a safe place for all of us to live.
Preparation Of Ozone(O₃)
Ozone (O₃) is typically produced by using electric sparks or ultraviolet light to split ordinary oxygen molecules (O₂) into single oxygen atoms. These highly reactive atoms quickly combine with other oxygen molecules to form ozone. The process can be summarized by the equation:
which shows that three molecules of oxygen are converted into two molecules of ozone. In laboratories or industrial settings, this is often done using devices called ozone generators, where a high voltage electric arc is passed through oxygen gas. The electric sparks from the arc break the oxygen molecules into individual atoms, which then combine with other oxygen molecules to form ozone. This method is widely used because it can produce a concentrated amount of ozone efficiently, which is useful for various applications such as water treatment, air purification, and in chemical synthesis.
Physical Properties of Ozone(O₃)
| Property | Description |
|---|---|
| Chemical Formula | O₃ |
| Physical State | Gas at room temperature |
| Color | Pale blue |
| Odor | Sharp, chlorine-like |
| Melting Point | -192.2 °C (-313.96 °F) |
| Boiling Point | -111.9 °C (-169.42 °F) |
| Density | 2.144 g/L (at 0 °C and 101.325 kPa) |
| Solubility in Water | Soluble, but less so than oxygen. The solubility decreases with increasing temperature. |
| Magnetic Susceptibility | Paramagnetic, meaning it is attracted by a magnetic field due to its unpaired electrons. |
Chemical Properties of Ozone(O₃)
Highly Reactive
Ozone is much more reactive than dioxygen (O₂) due to its molecular structure. It readily reacts with other molecules, making it a powerful oxidizing agent. This property is utilized in water treatment and air purification to break down pollutants.
Oxidizing Agent
As an oxidizing agent, ozone can cause metals to corrode and rubber to crack. It reacts with most metals to form metal oxides. For instance:
This equation shows iron (Fe) reacting with ozone to form iron(III) oxide (rust).
Decomposition into Oxygen
Ozone is unstable and tends to decompose back into dioxygen:
This decomposition can be accelerated by the presence of catalysts or high temperatures.
Reaction with Organic Compounds
Ozone reacts with many organic compounds in a process known as ozonolysis, breaking down complex molecules into simpler ones. This is particularly useful in organic synthesis and pollution control. For example, in the ozonolysis of alkenes:
This equation illustrates how an alkene reacts with ozone to form two carbonyl compounds.
Ozone and Living Tissues
Ozone can have harmful effects on living tissues. Inhaling ozone can lead to respiratory problems because it reacts with compounds in the respiratory tract. This property underlines the importance of monitoring and regulating ozone levels in the air.
Ozone(O₃) Chemical Compound Information
Chemical Identifiers
| Property | Value |
|---|---|
| CAS Registry Number | 10028-15-6 |
| PubChem Compound ID | 24823 |
| SMILES Identifier | [O-][O+]=O |
| InChI Identifier | InChI=1/O3/c1-3-2 |
| EU Number | 233-069-2 |
| Gmelin Number | 1101 |
| RTECS Number | RS8225000 |
Uses of Ozone(O₃)
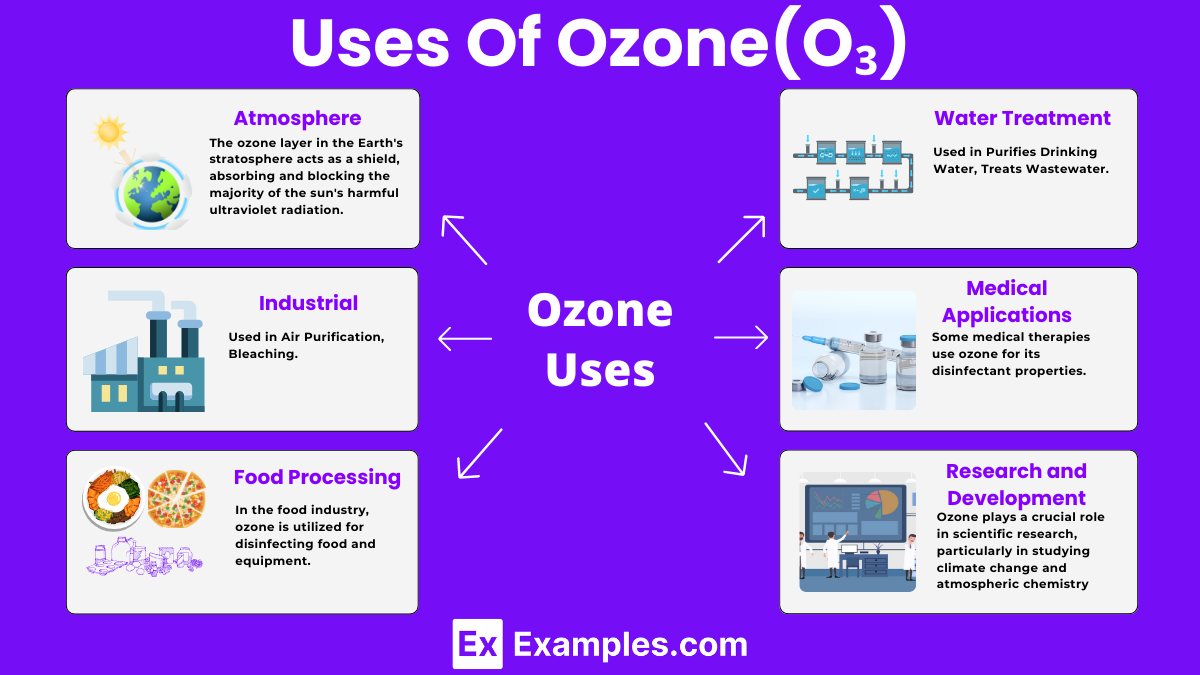
In the Atmosphere
- Protects from UV Rays: The ozone layer in the Earth’s stratosphere acts as a shield, absorbing and blocking the majority of the sun’s harmful ultraviolet radiation. This protection reduces the risk of skin cancer and cataracts in humans, and prevents damage to plants and marine ecosystems.
Water Treatment
- Purifies Drinking Water: Ozone is used in water treatment plants to kill bacteria and other pathogens. It’s an effective disinfectant that helps ensure our drinking water is safe.
- Treats Wastewater: Besides purifying drinking water, ozone is also used to treat wastewater. It breaks down pollutants and organic waste, making the water safer to release into the environment.
Industrial Uses
- Food Processing: In the food industry, ozone is utilized for disinfecting food and equipment. It kills bacteria and degrades pesticides on fruits and vegetables, making them safer for consumption.
- Air Purification: Ozone generators are used to clean the air in some industrial and commercial settings. They help eliminate odors, kill microbes, and reduce air pollutants.
- Bleaching: Ozone acts as a bleaching agent in the production of paper and textiles. It’s an environmentally friendly alternative to chlorine-based bleaches.
Medical Applications
- Treatment of Medical Conditions: Some medical therapies use ozone for its disinfectant properties. It’s applied in treating wounds, ulcers, and certain skin diseases, promoting faster healing.
Research and Development
- Scientific Research: Ozone plays a crucial role in scientific research, particularly in studying climate change and atmospheric chemistry. Researchers use ozone measurements to understand environmental impacts and develop strategies for pollution control.
Function Of Ozone (O₃)
UV Radiation Protection
Ozone in the stratosphere absorbs most of the sun’s harmful ultraviolet radiation, protecting living organisms from UV damage, which includes reducing risks of skin cancer and cataracts in humans and safeguarding ecosystems.
Atmospheric Role
- Climate Influence: Ozone contributes to the Earth’s temperature regulation, essential for energy balance.
- Air Quality: At ground level, it affects air quality, with high concentrations being harmful, while also playing a role in pollutant removal.
- Greenhouse Effect: In the troposphere, ozone acts as a greenhouse gas, influencing the climate by trapping heat.
Benefits of Ozone (O₃)
Protects Against UV Radiation
- Natural Sunscreen: Shields the Earth from harmful UV radiation, reducing the risk of skin cancer and cataracts.
Water Treatment
- Purifies Water: Destroys bacteria and pathogens in drinking water without leaving harmful residues.
Air Quality Improvement
- Eliminates Odors: Removes unpleasant odors in air and water by breaking down pollutants.
Food Preservation
- Extends Shelf Life: Slows down the spoilage of food by killing surface microorganisms.
Medical Applications
- Disinfectant: Used in ozone therapy to treat infections and promote wound healing.
Industrial Applications
- Bleaching Agent: Serves as an eco-friendly bleaching option in textiles and paper manufacturing.
Environmental Cleanup
- Detoxifies Contaminants: Cleans soil and groundwater from toxic substances.
Agricultural Benefits
- Crop Protection: Helps in pest control and reduces chemical pesticide use, improving crop yield.
Side Effects Of Ozone(O₃)
- Respiratory Issues: Can cause coughing, throat irritation, and breathing difficulty.
- Asthma Aggravation: May worsen asthma symptoms and increase hospital visits.
- Lung Damage: Long-term exposure can harm lung tissue and decrease lung function.
- Immune System Impact: Could make individuals more susceptible to respiratory infections.
- Air Quality: Contributes to smog and poor outdoor air quality, affecting vulnerable groups like the elderly and children.
- Indoor Air Quality: Using ozone generators indoors improperly can lead to harmful air pollutants.
FAQ’S
Is Ozone Harmful to Humans?
Ozone can be harmful, causing respiratory issues and aggravating lung diseases like asthma. High concentrations damage the lungs and throat.
Why is Ozone Bad for Earth?
At ground level, ozone is a pollutant, damaging crops, forests, and harming wildlife. It contributes to the greenhouse effect and global warming.
Why is Ozone Therapy Illegal?
Ozone therapy is illegal in some places due to lack of FDA approval and potential health risks like blood vessel damage and lung problems.
What Does Ozone Smell Like?
Ozone has a distinct sharp, clean smell, often described as similar to chlorine bleach. It’s noticeable after thunderstorms or around electrical equipment


