Borax (Na₂B₄O₇·10H₂O) – Definition, Structure, Preparation, Properties, Uses, Side Effects
Borax, also known as sodium borate, is a versatile inorganic compound that plays a significant role in chemistry and various industrial applications. This white powder is not only fundamental in creating cleaning products and detergents but also serves as an essential ingredient in glassmaking and ceramics. Easily soluble in water, borax exhibits unique properties that make it valuable in both household and laboratory settings. Its chemical stability and low toxicity provide a safe and effective solution for many chemical reactions and processes, making it a staple in the study of chemistry and the production of various inorganic materials.
What is Borax?
Chemical Names and Formulas
| Property | Value |
|---|---|
| Formula | Na₂B₄O₇·10H₂O |
| Hill Formula | B₄H₂₀Na₂O₁₇ |
| Name | Sodium borate decahydrate |
| IUPAC Name | Disodium 3,7-dioxido-2,4,6,8,9-pentaoxa-1,3,5,7-tetraborabicyclo[3.3.1]nonane decahydrate |
Structure of Borax
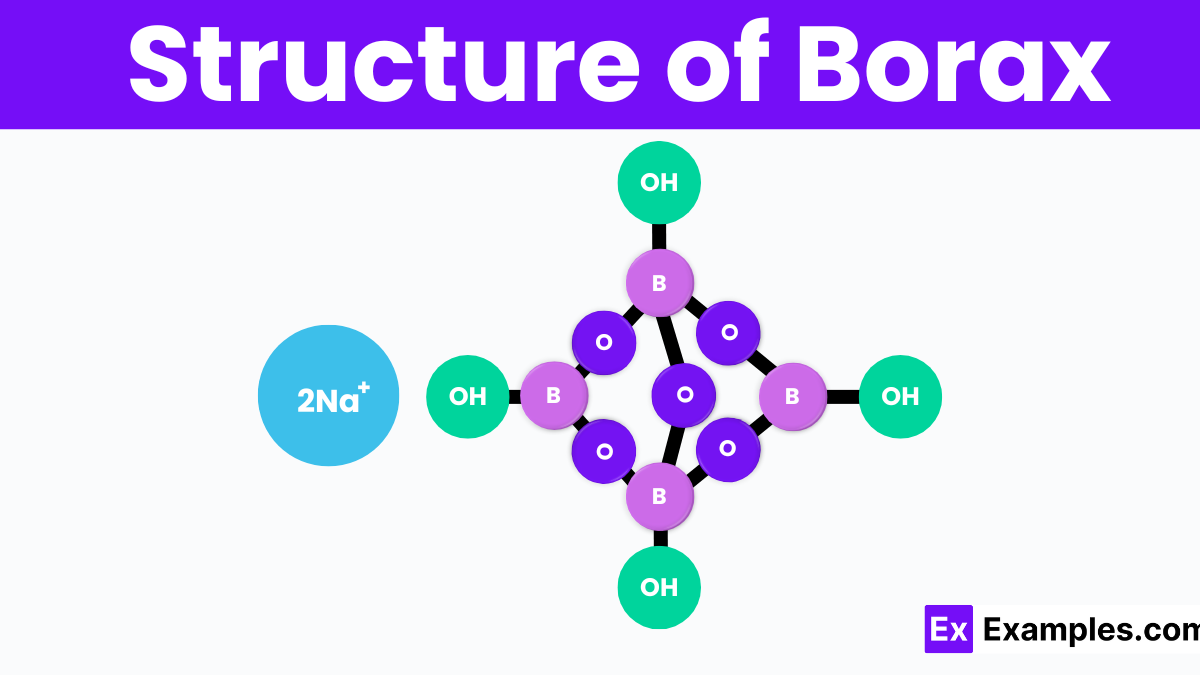
Borax, scientifically known as sodium tetraborate decahydrate, has a unique crystal structure that includes four boron atoms, ten water molecules, and two sodium atoms. In this structure, the boron atoms are linked together in a symmetrical pattern, forming a part of the compound called the borate ion. Each boron atom is connected to oxygen atoms, creating a network that holds the sodium ions in place. This arrangement results in a solid, yet easily dissolvable, structure in water. Understanding borax’s structure helps explain its effectiveness in various cleaning and household tasks.
Preparation of Borax
Borax is typically prepared through a process involving the mineral colemanite, a borate mineral rich in calcium. This process starts with the reaction of colemanite with sodium carbonate (soda ash). During this reaction, colemanite reacts with sodium carbonate in hot water, resulting in the formation of borax and calcium carbonate as a by-product. The chemical equation for this reaction is:
Here, Na₂B₄O₇·10H₂O represents borax (sodium tetraborate decahydrate), and 𝐶𝑎𝐶𝑂₃ stands for calcium carbonate, which precipitates out. The mixture is then filtered to separate the borax from the calcium carbonate sediment. The resulting solution is cooled, allowing the borax to crystallize. This method is widely used in industry due to its efficiency in producing borax from natural sources.
Physical Properties of Borax
| Property | Description |
|---|---|
| Appearance | White, crystalline powder or granular solid |
| Solubility in Water | Soluble, especially at higher temperatures |
| Melting Point | 743°C (dehydrates), 1575°C (anhydrous) |
| Density | 1.73 g/cm³ (solid) |
| pH | About 9.3 (alkaline when dissolved in water) |
| Odor | Odorless |
Chemical Properties of Borax
Reacts with Acids
Borax reacts with strong acids such as hydrochloric acid to produce boric acid and sodium chloride. The chemical equation for this reaction is:
This reaction is particularly useful for producing boric acid, which is employed in various applications including antiseptics and insecticides.
Thermal Decomposition
Upon heating, borax undergoes thermal decomposition. It first loses its water molecules to form anhydrous sodium tetraborate at about 320°C. Further heating results in the formation of boron oxide (B2O3), sodium oxide (Na2O), and additional water. The reactions involved are:
Na₂B₄O₇ → 2NaBO₂ + B₂O₃
This decomposition is crucial for its role as a flux in the manufacture of glass and ceramics.
Buffer Solution
Borax can create a buffer solution when dissolved in water. This buffered solution is valuable for maintaining a stable pH, which is crucial in both chemical and biochemical lab settings for sustaining the necessary conditions for various reactions.
Complexation
Borax can form complexes with different molecules. An important example is its ability to react with methylene blue to form a borate complex, used as a pH indicator. This property of complexation makes borax a useful component in laboratory experiments and tests.
Borax (Na₂B₄O₇·10H₂O) Chemical Compound Information
Chemical Identifiers
| Property | Value |
|---|---|
| CAS Registry Number | 1303-96-4 |
| PubChem Compound ID | 16211214 |
| SMILES Identifier | B1(OB2OB(OB(O1)O2)[O-])[O-].O.O.O.O.O.O.O.O.O.O.O.[Na+].[Na+] |
| InChI Identifier | InChI=1/B4O7.2Na.10H2O/c5-1-7-3-9-2(6)10-4(8-1)11-3;;;;;;;;;;;;/h;;;101H2/q-2;2+1;;;;;;;;;; |
| RTECS Number | VZ2275000 |
| MDL Number | MFCD00149193 |
NFPA Label
| Property | Value |
|---|---|
| NFPA Health Rating | 2 |
| NFPA Fire Rating | 0 |
| NFPA Reactivity Rating | 0 |
Function of Borax
Cleaning and Deodorizing
Borax is commonly used in household cleaning products due to its ability to enhance the effectiveness of detergents. It helps to break down stains and odors, making it excellent for laundry and general household cleaning.
Water Softening
In areas with hard water, borax can act as a water softener. It binds to the minerals in hard water, such as calcium and magnesium, preventing them from interfering with the cleaning power of soaps and detergents.
pH Buffering
Borax also functions as a pH buffer, maintaining a stable alkaline environment in solutions. This property is crucial in pools, cosmetics, and various industrial applications to control acidity and alkalinity.
Antifungal and Antiseptic Agent
Due to its antifungal properties, borax is used in some over-the-counter creams and sprays to treat fungal infections like athlete’s foot. It also has mild antiseptic qualities for preventing the growth of microorganisms.
Enamel Enhancer
In the ceramics industry, borax is used to improve the quality of glazes and enamels on pottery and ceramics. It helps to create smooth and glossy surfaces that are more durable.
Flame Retardant
Another significant function of borax is its role as a flame retardant. It is applied to materials like wood and fabric to reduce their flammability and slow the spread of fire in case of an emergency.
Uses of Borax
Household Cleaning
Borax is a popular ingredient in many cleaning products due to its ability to enhance the cleaning power of laundry detergents. It helps remove tough stains, neutralizes odors, and disinfects surfaces effectively.
Pest Control
Due to its toxic properties to insects, borax is often used in homemade pest control recipes. It is particularly effective against ants and cockroaches when mixed with sweeteners that attract these pests.
Fungicide and Herbicide
Borax serves as a fungicide and herbicide, controlling the growth of mold, mildew, and weeds. It’s especially useful in gardens and lawns to manage fungal diseases and unwanted plants.
Fire Retardant
In the realm of fire safety, borax is used as a fire retardant. It’s applied to wood and fabrics to reduce their flammability and slow down the spread of fire.
Glass and Ceramic Production
In manufacturing, borax is a vital component in the production of glass and ceramics. It lowers the melting point of glass and improves the gloss and durability of glazed ceramics.
Slime Creation
A fun and educational use of borax is in making slime. It acts as a cross-linking agent that binds polymer molecules in glue to create stretchy, gooey slime, which is a popular science activity for kids.
Side Effects of Borax
- Skin Irritation: Direct contact with borax powder can cause skin irritation, redness, or a burning sensation, especially for those with sensitive skin.
- Eye Irritation: Borax dust can irritate the eyes, causing pain, redness, and blurred vision. It is important to wear protective goggles when handling borax in its powdered form.
- Respiratory Issues: Inhaling borax dust can lead to respiratory problems, including sore throat, coughing, and shortness of breath. In severe cases, it can cause respiratory tract inflammation.
- Digestive Disturbance: Accidental ingestion of borax can lead to nausea, vomiting, and diarrhea. In larger amounts, it can be highly toxic and potentially fatal.
- Reproductive Health Concerns: There is evidence to suggest that prolonged exposure to borax can affect fertility and may cause harm to the unborn child if exposure occurs during pregnancy.
FAQs
Is Borax Harmful to Humans?
Yes, borax can be harmful if ingested, inhaled, or if it contacts skin, causing irritation, respiratory issues, and toxicity.
Is Borax Just Baking Soda?
No, borax and baking soda are different substances; borax is sodium tetraborate, while baking soda is sodium bicarbonate.
Why Do People No Longer Use Borax?
Borax is less used due to safety concerns about its potential toxicity and environmental impact, leading to restricted use in some countries.
What Bugs Does Borax Kill?
Borax effectively kills household pests like ants, cockroaches, fleas, and silverfish when used as a pesticide.



