Hydrochloric Acid (HCl) – Definition, Structure, Preparation, Properties, Uses, Side Effects
Hydrochloric acid, often found in the fascinating world of chemistry, is a strong and important acid with a formula HCl. This clear, colorless solution is not just any acid; it plays a crucial role in our daily lives, from aiding in digestion within our stomachs to being used in various industries for cleaning and processing materials. Its unique properties make it a staple in science experiments and industrial applications alike, showcasing the power and versatility of chemistry in action. Whether it’s breaking down food or cleaning metal surfaces, hydrochloric acid demonstrates the incredible ways acids contribute to both nature and technology.
What is Hydrochloric Acid?
Chemical Names and Formulas
| Property | Value |
|---|---|
| Formula | HCl |
| Name | Hydrogen Chloride |
| Alternate Names | Anhydrous Hydrochloric Acid, Anhydrous Hydrogen Chloride, Chlorane |
Structure of Hydrochloric Acid
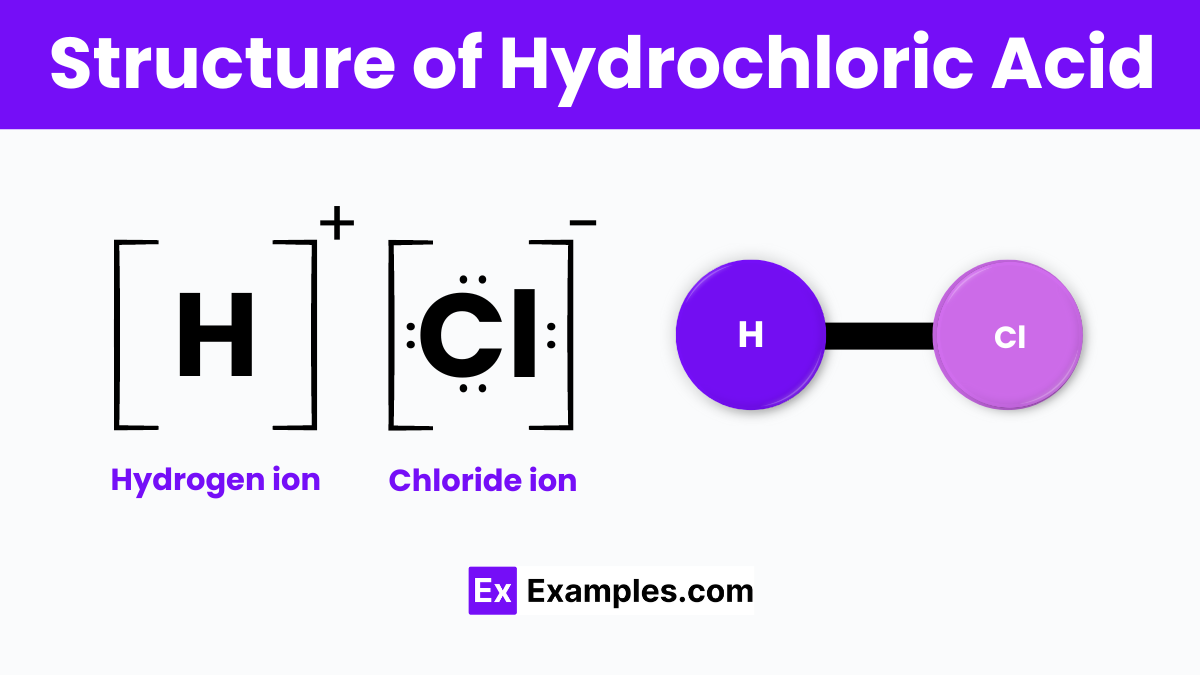
Hydrochloric Acid, commonly known as HCl, is a simple but powerful substance found in many scientific and industrial applications. Imagine it as a dance between two very different partners: a hydrogen atom (H) and a chlorine atom (Cl). These two atoms come together to form a molecule of HCl. In this molecular dance, the hydrogen atom donates its single electron to the chlorine atom, creating a strong bond that keeps them closely linked. This bonding results in HCl being a highly reactive, colorless gas under standard conditions, which, when dissolved in water, creates a strong and corrosive acid.
Preparation of Hydrochloric Acid
Hydrochloric Acid is like a recipe that combines two main ingredients: hydrogen gas (H₂) and chlorine gas (Cl₂). When these two gases are brought together, they react in an energetic dance to form Hydrochloric Acid. Picture it as if the hydrogen and chlorine atoms are eager to join hands, and when they do, they create HCl as a result. This reaction is not only fascinating but also quite straightforward. The chemical equation that represents this process looks like this:
In simpler terms, when one molecule of hydrogen gas reacts with one molecule of chlorine gas, two molecules of Hydrochloric Acid gas are formed. This reaction is typically carried out in a controlled environment, as it requires careful handling due to the reactive nature of chlorine gas and the corrosive property of Hydrochloric Acid. Once formed, Hydrochloric Acid can be dissolved in water to produce a solution that’s widely used in various applications, from cleaning to food processing.
Physical Properties of Hydrochloric Acid
| Property | Description |
|---|---|
| State at Room Temperature | Hydrochloric acid is typically found as a colorless to slightly yellow liquid when it is in a water solution. |
| Odor | It has a sharp, pungent smell, reminiscent of vinegar, but much stronger. |
| Taste | Not applicable and highly dangerous to try, but it is extremely sour. |
| Boiling Point | The boiling point of concentrated hydrochloric acid solution is around 110°C (230°F). |
| Melting Point | The melting point is about -27.32°C (-17.18°F) for the concentrated acid. |
| Solubility in Water | It is highly soluble in water, capable of forming a wide range of concentrations. |
| Density | The density of a 38% solution in water is approximately 1.19 g/cm³ at 25°C. |
| pH Value | A 1 M solution of HCl in water has a pH of about 0, making it a very strong acid. |
Chemical Properties of Hydrochloric Acid
Reactivity with Metals
- Hydrochloric Acid reacts with most metals to produce hydrogen gas (H₂) and a metal chloride. For instance, when it reacts with zinc (Zn), we get zinc chloride (ZnCl₂) and hydrogen gas.
- Equation: Zn (s) + 2HCl (aq) → ZnCl₂ (aq) + H₂ (g)
Reactivity with Carbonates
- When Hydrochloric Acid is added to carbonates, such as calcium carbonate (CaCO₃), it produces carbon dioxide (CO₂), water (H₂O), and a chloride salt.
- Equation: CaCO₃ (s) + 2HCl (aq) → CaCl₂ (aq) + CO₂ (g) + H₂O (l)
Reactivity with Bases
- HCl reacts with bases (alkalis) to form a salt and water, in a process known as neutralization. For example, when HCl reacts with sodium hydroxide (NaOH), sodium chloride (table salt, NaCl) and water are formed.
- Equation: HCl(aq) + NaOH(aq) → NaCl(aq) + H₂O(l)
Conductivity
Hydrochloric Acid is an excellent conductor of electricity when dissolved in water. This is because it dissociates into ions (H⁺ and Cl⁻), which move freely in the solution, allowing it to conduct electricity.
pH Level
HCl has a very low pH, making it a strong acid. A 1 Molar (1M) solution of HCl in water has a pH of around 0, indicating its high acidity.
Stability
While Hydrochloric Acid is stable under normal conditions, it can release dangerous chlorine gas when mixed with other chemicals, highlighting the importance of handling it with care.
Hydrochloric Acid (HCl) Chemical Compound Information
Chemical Identifiers
| Property | Value |
|---|---|
| CAS Registry Number | 7647-01-0 |
| PubChem Compound ID | 313 |
| PubChem Substance ID | 24857783 |
| SMILES Identifier | Cl |
| InChI Identifier | InChI=1/ClH/h1H |
| RTECS Number | MW4025000 |
| MDL Number | MFCD00011324 |
NFPA Label
| Property | Value |
|---|---|
| NFPA Health Rating | 3 |
| NFPA Fire Rating | 0 |
| NFPA Reactivity Rating | 1 |
Uses of Hydrochloric Acid
Cleaning Agent
Hydrochloric Acid is widely used to clean metals before they are plated with another metal. Its strong acidic nature helps remove rust and other impurities from the surface of metals, ensuring that they are clean and ready for further processing.
Food Industry
In the food industry, HCl plays a crucial role in the production of various food products. It is used to make gelatin and other food additives. Hydrochloric Acid helps in breaking down proteins and is used in the process of making corn syrups.
Medicine
Hydrochloric Acid is also an important component in the pharmaceutical industry. It is used to produce a variety of medicines and as a pH regulator in the formulation of some medications, ensuring they are effective when used.
Water Treatment
In water treatment processes, Hydrochloric Acid is used to adjust the pH level of water. This is important in ensuring that drinking water is safe and in maintaining the effectiveness of the water purification process.
Leather Processing
The leather industry uses Hydrochloric Acid in the tanning process of leather. It helps in removing impurities from the raw hide and preparing it for further processing, ensuring the leather is clean and ready for use.
Laboratory Use
In laboratories, HCl is a staple reagent. It is used in various chemical reactions and experiments due to its reactive nature. Hydrochloric Acid is also used in titrations to determine the concentration of basic (alkaline) solutions.
Household Cleaning
Diluted Hydrochloric Acid is found in many household cleaning products. It is effective in removing tough stains from toilets and tiles, making it a powerful cleaning agent for maintaining cleanliness in homes.
Side Effects of Hydrochloric Acid (HCl)
- Skin Irritation and Burns: Direct contact with Hydrochloric Acid can cause severe skin irritation and chemical burns. Even low concentrations can irritate the skin, while higher concentrations can cause burns, sometimes leading to permanent damage.
- Respiratory Issues: Inhaling the fumes of Hydrochloric Acid can irritate the respiratory tract, leading to coughing, choking, and in severe cases, pulmonary edema, where the lungs fill with fluid.
- Eye Damage: Exposure to HCl fumes or splashes can severely damage the eyes, potentially leading to blindness. Even brief contact can cause significant irritation and redness.
- Digestive System Damage: Accidentally ingesting Hydrochloric Acid can cause severe damage to the mouth, throat, esophagus, and stomach, leading to nausea, vomiting, stomach pain, and in extreme cases, perforation of the stomach or intestines.
- Environmental Harm: When not handled properly, Hydrochloric Acid can be harmful to the environment. It can damage soil, water sources, and aquatic life, highlighting the need for careful disposal and handling.
FAQ’S
Is Hydrochloric Acid Harmful to Humans?
Yes, Hydrochloric Acid (HCl) can be harmful, causing skin burns, respiratory issues, and eye damage. Proper safety measures are essential when handling it.
What is Hydrochloric Acid Commonly Found In?
Hydrochloric Acid is commonly found in industrial cleaners, the food industry for processing, and naturally in the stomach to aid digestion.
Does Vinegar Make Hydrochloric Acid?
No, vinegar does not make Hydrochloric Acid. Vinegar mainly contains acetic acid, which is chemically different from Hydrochloric Acid (HCl).



