Beryllium (Be) – Definition, Preparation, Properties, Uses, Compounds, Reactivity
Beryllium, a fascinating element, is often overlooked in educational discussions. This guide aims to change that. We delve into the essence of Beryllium, exploring its unique properties and relevance in various fields. Our focus is on making this information accessible and engaging for teachers, enhancing their ability to convey complex concepts with ease. Beryllium’s multifaceted nature offers a rich context for learning, making it an invaluable addition to any curriculum.
What is Beryllium?
Beryllium is a chemical element with the symbol Be and atomic number 4. It is a hard, grayish metal naturally found in mineral rocks, coal, soil, and volcanic dust. This lightweight yet strong element is notable for its high melting point and excellent thermal conductivity. Beryllium is used in various applications, including aerospace, telecommunications, and nuclear reactors, due to its unique physical properties. Understanding Beryllium’s characteristics and applications can enhance teaching related to chemistry, physics, and industrial applications.
Other Alkaline Earth Metals
| Magnesium(Mg) |
| Calcium(Ca) |
| Strontium(Sr) |
| Barium(Ba) |
| Radium(Ra) |
Beryllium Formula
- Formula: Be
- Composition: A single beryllium atom.
- Bond Type: Beryllium forms covalent bonds and can also exhibit ionic characteristics.
- Molecular Structure: Hard, gray metal.
- Electron Configuration: Four electrons, with the configuration 1s² 2s².
- Significance: Beryllium is used in aerospace materials, nuclear reactors, and as a component in various alloys.
- Role in Chemistry: It’s known for its stiffness, lightweight, and ability to slow down neutrons, making it valuable in nuclear applications.
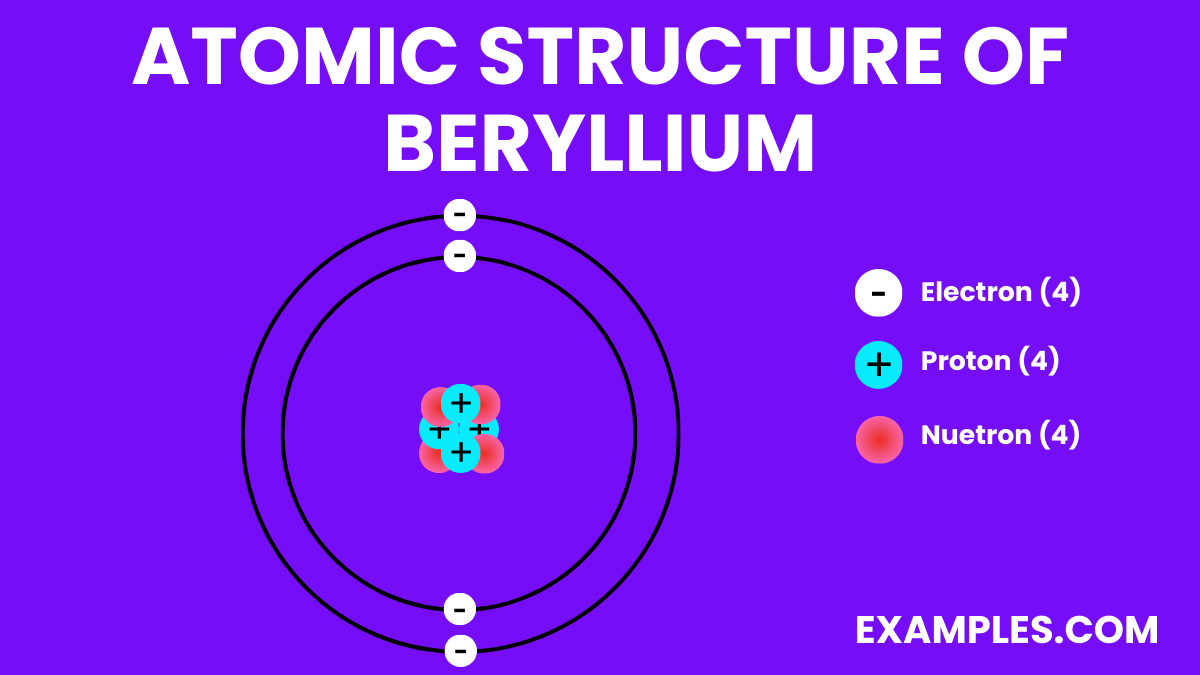
Atomic Structure of Beryllium
Properties of Beryllium

Physical Properties of Beryllium
| Property | Description |
|---|---|
| Appearance | Silvery-gray, hard, lightweight metal. |
| Atomic Number | 4 |
| Atomic Mass | Approximately 9.0122 u |
| Density | About 1.85 g/cm³ |
| Melting Point | 1287°C (2349°F) |
| Boiling Point | 2469°C (4476°F) |
| State at Room Temperature | Solid |
| Thermal Conductivity | High thermal conductivity |
| Electrical Conductivity | Good electrical conductor, though not as much as some metals |
Chemical Properties of Beryllium
- Reactivity: Beryllium is relatively inert at room temperature but becomes more reactive with oxygen or air upon heating, forming beryllium oxide (BeO). It doesn’t readily react with water or acids, except for nitric acid.
- Electron Configuration: The electron configuration of beryllium is 1s² 2s². Its two valence electrons make it prone to forming covalent bonds.
- Electronegativity: It has a relatively high electronegativity due to its small atomic size, making it quite electronegative among metals.
- Ionization Energy: Beryllium has a high ionization energy, which makes it less reactive than other alkaline earth metals.
- Compounds: Beryllium forms a variety of compounds, most notably beryllium oxide (BeO) and beryllium chloride (BeCl₂). These compounds are characterized by their high melting points and strength.
- Alloys: Beryllium forms alloys with many metals, including aluminum, copper, and nickel. These alloys are lightweight, strong, and have significant thermal stability.
- Toxicity: Beryllium compounds are toxic and can cause lung diseases when inhaled. Therefore, safety precautions are necessary when handling them.
- Role in Organic Chemistry: Beryllium has limited roles in organic chemistry due to its toxicity and reactivity. However, it is used in some high-tech applications, such as in aerospace components and nuclear reactors, due to its stiffness, light weight, and transparency to X-rays.
Thermodynamic Properties of Beryllium
| Property | Description / Value |
|---|---|
| Melting Point | 1287°C (2349°F) |
| Boiling Point | 2469°C (4476°F) |
| Thermal Conductivity | 200 W/(m·K) at 25°C |
| Specific Heat | 1.82 J/(g·K) at 25°C |
| Thermal Expansion | 11.3 µm/(m·K) at 25°C |
Material Properties of Beryllium
| Property | Description / Value |
|---|---|
| Density | 1.85 g/cm³ |
| Young’s Modulus | 287 GPa |
| Tensile Strength | 370 MPa |
| Mohs Hardness | 5.5 |
| Elastic Modulus | 300 GPa |
Electromagnetic Properties of Beryllium
| Property | Description / Value |
|---|---|
| Electrical Conductivity | 36% IACS (International Annealed Copper Standard) |
| Magnetic Susceptibility | -9.0·10⁻⁶ cm³/mol (diamagnetic) |
| Reflectivity | Approximately 67% at 600 nm (visible light spectrum) |
Nuclear Properties of Beryllium
| Property | Description / Value |
|---|---|
| Atomic Number | 4 |
| Atomic Mass | 9.012182 u |
| Neutron Cross Section | 0.0092 barns (thermal neutrons) |
| Isotopes | Mainly ⁹Be; ¹⁰Be (trace, cosmogenic) |
| Radioactivity | ⁷Be (radioactive isotope, half-life: 53.22 days) |
Chemical Compounds of Beryllium

- Beryllium Oxide (BeO)
- Explanation: Beryllium oxide is a white crystalline compound, notable for its high thermal conductivity and electrical resistivity. It’s used in electronics, microwave ovens, and as a ceramic for high-performance electronics.
- Equation: Be + ½O₂ → BeO
- Beryllium Chloride (BeCl₂)
- Explanation: This compound is formed by the reaction of beryllium with chlorine. It’s a hygroscopic solid used in organic chemistry for various reactions, including Friedel-Crafts acylations.
- Equation: Be + Cl₂ → BeCl₂
- Beryllium Hydroxide (Be(OH)₂)
- Explanation: Beryllium hydroxide is an amphoteric hydroxide, soluble in both acids and alkalis. It is used in the extraction of beryllium from its ores.
- Equation: Be(OH)₂ (s) ⇌ Be²⁺ (aq) + 2OH⁻ (aq)
- Beryllium Sulfate (BeSO₄)
- Explanation: This compound is a white crystalline solid used in the production of beryllium and its compounds. It’s also used in analytical chemistry for its properties as a flame retardant.
- Equation: Be + H₂SO₄ → BeSO₄ + H₂↑
- Beryllium Nitrate (Be(NO₃)₂)
- Explanation: Beryllium nitrate is used in pyrotechnics for the brilliant white color it imparts to flames. It is also used in the laboratory for various chemical syntheses.
- Equation: Be + 2HNO₃ → Be(NO₃)₂ + H₂↑
- Beryllium Fluoride (BeF₂)
- Explanation: Beryllium fluoride is used in nuclear reactors as a high-temperature liquid fuel. It is highly stable and forms a tetrahedral structure similar to that of silica.
- Equation: Be + F₂ → BeF₂
Isotopes of Beryllium
| Isotope | Half-Life | Natural Abundance | Mode of Decay | Usage/Significance |
|---|---|---|---|---|
| Be-7 | 53.12 days | Trace | Electron capture to Li-7 | Used in radiological dating and studies. |
| Be-9 | Stable | 100% | – | The only stable isotope, used in most applications of beryllium. |
| Be-10 | 1.39 million years | Trace | Beta decay to B-10 | Used in cosmogenic radionuclide dating of soils and sediments. |
| Be-8 | 6.7×10⁻¹⁷ seconds | None (synthetic) | Alpha decay to two He-4 nuclei | Important in nuclear physics studies, particularly in demonstrating the triple-alpha process in stars. |
| Be-11 | 13.81 seconds | None (synthetic) | Beta decay to B-11 | Used in nuclear physics experiments. |
| Be-12 | 21.49 milliseconds | None (synthetic) | Beta decay to C-12 | Studied in nuclear physics for its unusual decay properties. |
Uses of Beryllium

- Aerospace Applications
- Beryllium is prized in the aerospace industry due to its combination of light weight and high stiffness. It’s significantly lighter than aluminum and more rigid than steel, making it ideal for aerospace components where weight and performance are crucial. Beryllium is used in the manufacture of satellites, spacecraft, and various aerospace vehicles. Its thermal stability allows components made from beryllium to maintain their shape across a range of temperatures, which is essential in space applications.
- Nuclear Applications
- Beryllium is a critical component in nuclear reactors as a neutron reflector and moderator. It effectively slows down neutrons to speeds necessary for sustaining nuclear chain reactions. Additionally, its low absorption cross-section for thermal neutrons makes it an excellent reflector and moderator, significantly improving the efficiency of nuclear reactors. Beryllium is also used in the production of nuclear weapons.
- X-ray Windows
- Beryllium is transparent to X-rays, making it an ideal material for X-ray equipment windows. In medical and analytical X-ray equipment, beryllium windows allow the X-ray to pass through with minimal scattering and attenuation. This property ensures a clearer image or analysis result, which is vital in medical diagnostics and scientific research where precision is crucial.
- Electronic and Telecommunication Components
- Due to its high thermal conductivity, beryllium is used in electronic connectors, relays, switches, and other telecommunications network components. It effectively dissipates heat in electronic devices, preventing overheating and ensuring the reliability of these components. Beryllium’s non-magnetic nature is also beneficial in applications where magnetic interference can be a problem, like in certain electronic and telecommunication systems.
- Alloys and Ceramics
- Beryllium is alloyed with other metals such as copper to create materials that are more robust, corrosion-resistant, and better conductors of electricity and heat. These beryllium-copper alloys are used in various applications, including automotive systems, computer components, and electrical connectors. Beryllium oxide is used in ceramics for electronics and high-temperature applications due to its excellent heat conductivity and electrical insulation properties. These ceramics are crucial in electronics, where they provide both insulation and heat dissipation.
Commercial Production of Beryllium
Beryllium is commercially produced primarily from the mineral beryl, which is a beryllium aluminum silicate. The process of extracting and refining beryllium from beryl involves several steps:
- Mining: Beryl is mined through conventional mining techniques in various countries, including the United States, China, and Kazakhstan.
- Extraction: Once beryl ore is obtained, it undergoes a chemical process to extract beryllium. One common method is the sintering or fusing of beryl into a mix, usually with sodium fluorosilicate, at high temperatures.
- Refining: The fused mixture is then cooled and leached with sulfuric acid, which dissolves the beryllium and aluminum compounds. Beryllium hydroxide is precipitated out of this solution by adjusting the pH.
- Conversion to Metal: The beryllium hydroxide is then converted into beryllium fluoride or chloride and finally reduced with magnesium, producing beryllium metal.
- Purification: The beryllium metal is then further purified, typically by vacuum melting or by electrolysis.
The final product is a lightweight metal with a high melting point, significant stiffness, and excellent thermal stability, making it valuable for various industrial applications, including aerospace, electronics, and nuclear reactors.
Health Effects of Beryllium
Beryllium has several health effects, particularly when beryllium dust or fumes are inhaled:
- Beryllium Sensitization (BeS): This is an immune response that some people develop when exposed to beryllium. Sensitized individuals may not have symptoms but are at risk for developing chronic beryllium disease (CBD).
- Chronic Beryllium Disease (CBD): A serious lung disease caused by inhaling beryllium particles. Symptoms include coughing, shortness of breath, fatigue, and, in severe cases, can lead to lung failure.
- Lung Cancer: Beryllium is classified as a human carcinogen, and exposure can increase the risk of developing lung cancer.
- Acute Beryllium Disease: Though now rare due to improved industrial hygiene, it’s an acute form of chemical pneumonia caused by high exposure levels.
- Dermatitis: Skin contact with beryllium can cause rashes or ulcers, known as contact dermatitis.
Proper safety measures, including protective equipment, ventilation, and exposure monitoring, are essential to minimize these health risks in workplaces where beryllium is used.
Environmental Effects of Beryllium
The environmental impact of beryllium is relatively limited due to its low concentration in the Earth’s crust and in water. However, there are some concerns:
- Soil Contamination: Beryllium can contaminate soil near mining and processing sites, potentially affecting plant life.
- Water Pollution: Industrial processes may lead to the release of beryllium compounds into water sources, though this is typically at low levels.
- Airborne Particles: Industrial emissions can release beryllium particles into the air, which can be a health hazard for humans and animals if inhaled.
- Bioaccumulation: Beryllium does not tend to bioaccumulate in the food chain, reducing the risk of widespread ecological damage.
- Impact on Wildlife: Direct impact on wildlife is minimal, but in areas with significant soil or water contamination, there could be localized effects.
What Beryllium is Used For?
Beryllium is primarily used in aerospace, telecommunications, and defense, making lightweight, strong alloys and electrical conductors.
How is Beryllium Toxic to Humans?
Beryllium is toxic when inhaled as dust or fumes, causing lung diseases such as chronic beryllium disease and lung cancer.
Where is Beryllium Found in Everyday Life?
Beryllium is found in everyday items like electronics, golf clubs, and aerospace materials due to its strength and lightweight properties.
Is Beryllium the Most Toxic Metal?
While highly toxic, beryllium isn’t the most toxic metal; however, its inhalation is particularly dangerous due to lung disease risks.
What Does Beryllium Do to Your Body?
Inhaled beryllium can damage lungs, leading to chronic beryllium disease (CBD), scarring of lung tissue, and potential lung cancer.
What is Beryllium Good For?
Beryllium is valued for improving physical properties in alloys, enhancing conductivity in electrical components, and its use in nuclear reactors.
What Happens if You Touch Beryllium?
Touching beryllium metal is not typically harmful, but inhaling its dust or fumes can cause serious lung conditions. Skin contact may cause irritation.
Understanding beryllium’s unique properties, uses, and associated health risks is crucial. When writing about beryllium, emphasize its industrial significance and toxicity precautions. Highlight its applications in various fields while underscoring the importance of safe handling. This guide offers a balanced view, blending beryllium’s benefits with its potential hazards, providing comprehensive insights for various audiences.


