Carbon Dioxide (CO2) – Definition, Structure, Preparation, Uses, Benefits, Side Effects
Carbon dioxide is a gas that you can’t see or smell, but it’s all around us. When we breathe out, we release CO₂ into the air. It’s also made when things like cars and factories burn fuel. Trees and plants need Carbon dioxide to grow, taking it from the air and giving us oxygen in return. But, too much Carbon dioxide in the air can cause problems for our planet, like making the Earth warmer than it should be. This gas plays a big role in our world, from helping plants grow to affecting our climate.
What is Carbon Dioxide?
Carbon dioxide, often abbreviated as CO₂, is a covalent compound made of carbon and oxygen. In simpler terms, it’s a kind of gas that forms when carbon atoms bond closely with two oxygen atoms. This bonding happens in a special way called covalent bonding, where the atoms share electrons with each other. You can’t see or smell carbon dioxide, but it’s a big part of the air around us and plays a crucial role in life on Earth. Plants use CO₂ to make food through a process called photosynthesis, turning CO₂ and sunlight into energy. However, too much CO₂ in the atmosphere can lead to climate change, making it an important substance to understand in chemistry and environmental science.
Chemical Names and Formulas
| Property | Value |
|---|---|
| Formula | CO₂ |
| Name | Carbon Dioxide |
| Alternate Names | Anhydride Carbonique, Carbon Dioxide, Compressed, Carbonic Anhydride, Freon 744 R-744 |
Structure Of Carbon Dioxide (CO₂)
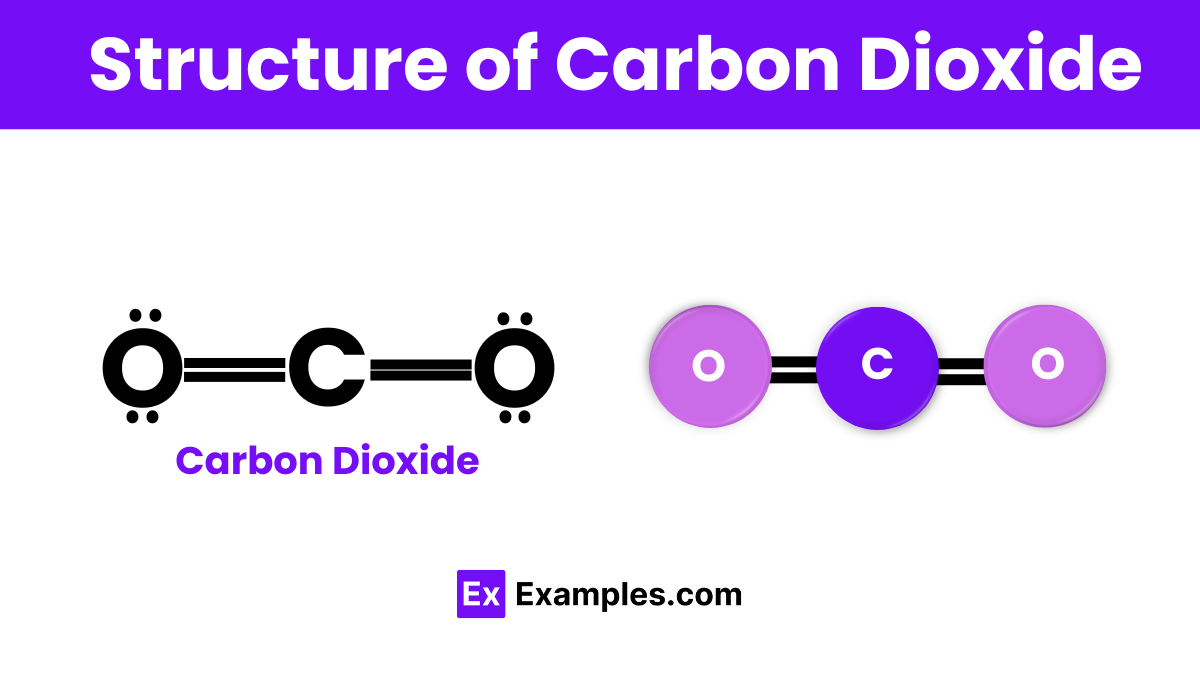
The structure of carbon dioxide (CO₂) is straightforward yet fascinating. At its core, CO₂ consists of one carbon atom that forms double bonds with two oxygen atoms. This configuration gives CO₂ a linear shape, meaning all three atoms lie in a straight line. The double bonds between the carbon and oxygen atoms are strong and stable, making CO₂ a non-polar molecule. This structural simplicity allows CO₂ to be a gas at room temperature, easily mixing with the air around us. Its linear, symmetrical form is key to its behavior and interactions in the environment and industrial processes.
Preparation Of Carbon Dioxide (CO₂)
Carbon dioxide (CO₂) can be prepared through several methods, one of the most common being the chemical reaction between a carbonate or bicarbonate and an acid. For example, when calcium carbonate (CaCO₃), commonly found in chalk or limestone, reacts with hydrochloric acid (HCl), carbon dioxide gas is released as a byproduct. The equation for this reaction is:
Another way to produce CO₂ is by heating carbonate compounds. When magnesium carbonate (MgCO₃) is heated, it decomposes to form magnesium oxide (MgO) and releases carbon dioxide gas. The equation for this process is:
These reactions not only illustrate how CO₂ can be generated in a laboratory setting but also reflect the compound’s involvement in natural geological processes and its role in the carbon cycle.
Physical Properties of Carbon Dioxide (CO₂)
| Property | Description |
|---|---|
| Molecular Weight | 44.01 g/mol |
| Phase at Room Temperature | Gas |
| Color | Colorless |
| Odor | Odorless |
| Density | 1.977 kg/m³ (at 0°C and 1 atm) |
| Melting Point | -56.6°C (sublimes) |
| Boiling Point | -78.5°C (at 1 atm, sublimation point) |
| Solubility in Water | 1.45 g/L (at 20°C and 1 atm) |
| Critical Temperature | 31.1°C |
| Critical Pressure | 7.38 MPa |
Chemical Properties of Carbon Dioxide (CO₂)
Reactivity with Water
Carbon dioxide dissolves in water to form carbonic acid (H₂CO₃), albeit weakly, affecting the water’s pH level. This reaction is crucial for the carbon cycle and aquatic life balance. CO₂+H₂O→H₂CO₃
Reactivity with Bases
CO₂ reacts with bases (alkalis) to produce carbonates or bicarbonates. For example, when CO₂ is passed through a solution of sodium hydroxide (NaOH), sodium carbonate (Na₂CO₃) is formed.
CO₂+2NaOH→Na₂CO₃+H2O
Formation of Carbonates
In the presence of water and calcium ions, CO₂ can lead to the formation of calcium carbonate (CaCO₃), a key component of limestone, through a series of reactions. This process is vital in geological formations and the ocean’s carbon cycle.
Photosynthesis
Carbon dioxide is a key reactant in the photosynthesis process by which plants convert CO₂ and water into glucose and oxygen, using sunlight as energy. This reaction is fundamental to life on Earth.
6CO₂+6H₂O→C₆H₁₂O₆+6O₂
Dry Ice to Gas (Sublimation)
At temperatures below its sublimation point (-78.5°C), solid CO₂ (dry ice) converts directly into CO₂ gas without becoming liquid. This property makes dry ice useful for refrigeration and creating fog effects.
Carbon Dioxide (CO₂) Chemical Compound Information
Chemical Identifiers
| Property | Value |
|---|---|
| CAS Registry Number | 124-38-9 |
| Beilstein Number | 1900390 |
| PubChem Compound ID | 280 |
| PubChem Substance ID | 24857758 |
| SMILES Identifier | C(=O)=O |
| InChI Identifier | InChI=1/CO2/c2-1-3 |
| RTECS Number | FF6400000 |
| MDL Number | MFCD00011491 |
Uses of Carbon Dioxide (CO₂)

In Beverages
CO₂ is used to carbonate soft drinks, sparkling water, and beer, giving them their fizz and enhancing their taste.
Fire Extinguishers
Carbon dioxide extinguishers are effective against electrical fires and flammable liquids because CO₂ displaces oxygen, smothering the fire.
Plant Growth
In greenhouses, CO₂ enrichment can accelerate plant growth, improving yield in controlled agricultural settings.
Refrigeration
Dry ice, the solid form of CO₂, is used for cooling and refrigeration. It’s particularly useful for transporting perishable goods and creating fog effects in entertainment.
Medical
Carbon Dioxide (CO₂) is used in medical procedures, including minimally invasive surgeries, as it is non-toxic and dissolves in the body without leaving residues.
Photosynthesis Research
Scientists use CO₂ in photosynthesis studies to understand plant growth and improve crop production strategies.
Oil Recovery
CO₂ is injected into oil wells to increase pressure and reduce the viscosity of the oil, enhancing its recovery.
Carbonated Concrete
Carbon dioxide is used to cure concrete, speeding up the hardening process and potentially trapping CO₂ permanently in buildings and structures.
Side Effects of Carbon Dioxide (CO₂)
Health Effects
- Breathing Problems: High levels of CO₂ can make it hard to breathe, especially for people with asthma or lung conditions.
- Dizziness: Being in an area with too much CO₂ can make you feel dizzy or light-headed.
- Headaches: Excessive CO₂ levels can cause headaches and fatigue, making it hard to concentrate.
- Increased Heart Rate: Your heart may beat faster as it tries to supply enough oxygen to your body in high CO₂ conditions.
Environmental Effects
- Global Warming: CO₂ is a greenhouse gas that traps heat in the atmosphere, leading to global warming and climate change.
- Ocean Acidification: CO₂ absorbed by oceans makes them more acidic, harming marine life and coral reefs.
- Altered Plant Growth: High CO₂ levels can change how plants grow, affecting food crops and ecosystems.
- Weather Changes: Increased CO₂ contributes to more extreme weather patterns, including hotter temperatures, storms, and droughts.
FAQ’S
What Does Carbon Dioxide Do to Your Body?
Excess carbon dioxide can lead to health issues like headaches, dizziness, breathing difficulties, and an increased heart rate, affecting overall well-being.
What Are CO₂ Levels in a Person?
Normal CO₂ levels in a person range between 35-45 mmHg, indicating healthy respiratory and metabolic function.
Is Carbon Dioxide Flammable?
Carbon dioxide is not flammable. It is used in fire extinguishers because it suffocates flames by displacing oxygen.
What Are CO₂ Levels in Air?
Current CO₂ levels in the air average about 417 ppm, a significant increase from pre-industrial levels, influencing global climate patterns.


