Formaldehyde (CH₂O) – Definition, Structure, Preparation, Properties, Uses, Side Effects
Formaldehyde is a fascinating substance that plays a big role in the world of chemistry. At its core, it’s a simple molecular compound, known scientifically as CH₂O. This means it’s made up of carbon (C), hydrogen (H), and oxygen (O) atoms arranged in a specific way. Even though it might sound complex, it’s actually found in everyday life, from preserving biological specimens to being used in making various household products. Its unique properties make it incredibly useful, but also something that chemists handle with care due to its strong odor and effects on health. So, when we dive into the chemistry of formaldehyde, we’re exploring a molecule that’s small in size but huge in impact!
What is Formaldehyde?
Chemical Names and Formulas
| Property | Value |
|---|---|
| Formula | HCHO |
| Hill formula | CH₂O |
| Name | Formaldehyde |
| Alternate Names | Formalin, Formol, Superlysoform |
Structure of Formaldehyde
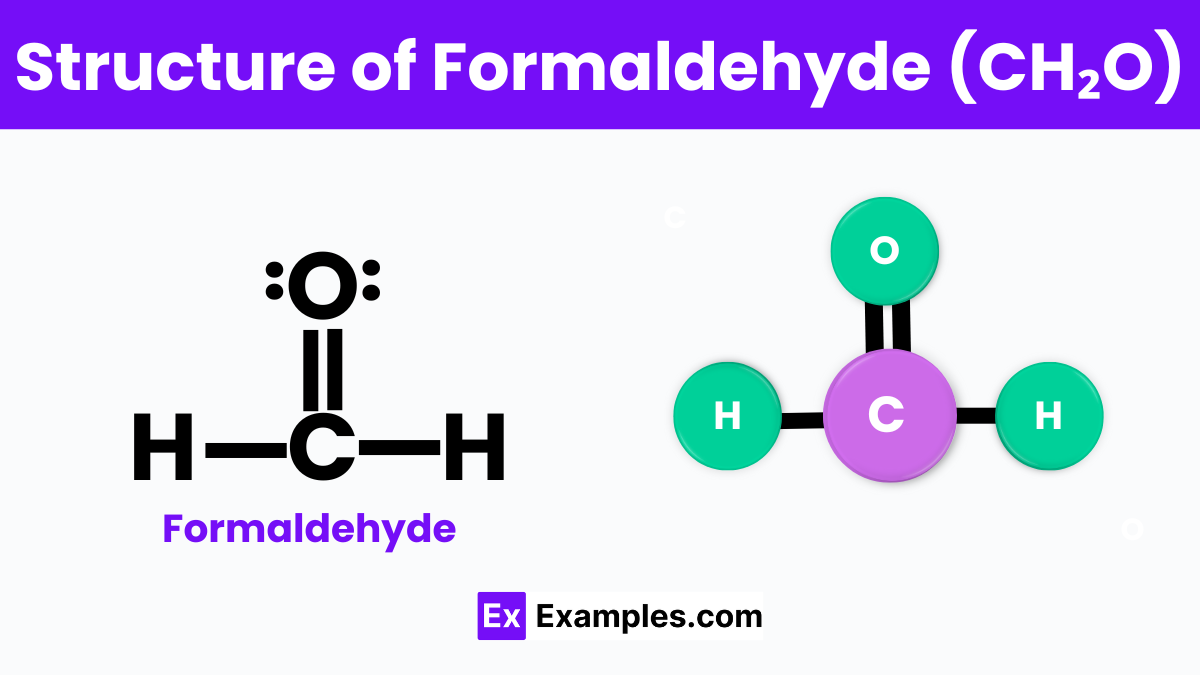
Formaldehyde, a common chemical in the science world, has a simple yet interesting structure that’s easy to understand. Picture it as a central carbon atom (C) holding hands with two hydrogen atoms (H) on one side and forming a double bond with an oxygen atom (O) on the other. This double bond with oxygen is what makes formaldehyde special. It looks like the letter “V,” with the carbon atom at the bottom point. This V-shaped molecule is not only easy to picture but also crucial for its ability to combine with other substances, leading to its wide use in making resins, plastics, and textiles. Understanding the basic “V” structure of formaldehyde helps us see how it fits into so many parts of our daily lives, from the construction of our homes to the clothes we wear.
Preparation of Formaldehyde
Formaldehyde is like a magic trick in the world of chemistry, created through a fascinating process. The main method to make formaldehyde is by using a gas called methanol, which scientists turn into formaldehyde through a reaction with oxygen from the air. Imagine methanol as a little building block that, when it meets oxygen, transforms into formaldehyde and water as a byproduct. This transformation happens in the presence of a catalyst, a special substance that speeds up the reaction without being consumed by it.The chemical equation for this process is quite straightforward:
This means two molecules of methanol (CH₃OH) react with one molecule of oxygen (O₂), resulting in two molecules of formaldehyde (CH₂O) and two molecules of water (H₂O). This process is widely used in industries to create formaldehyde, which then goes on to be a part of many products we use every day, from building materials to textiles. Understanding this reaction gives us a glimpse into the fascinating world of chemical transformations.
Physical Properties of Formaldehyde
| Property | Description |
|---|---|
| Appearance | Colorless gas; it’s invisible in the air and has a very strong, pungent smell that is easily noticeable. |
| Chemical Formula | CH₂O; This shows that formaldehyde is made up of carbon (C), hydrogen (H), and oxygen (O) atoms in a specific arrangement. |
| State at Room Temperature | Gas; Formaldehyde is usually found as a gas at room temperature, making it present in the air around us, especially indoors. |
| Solubility in Water | Highly soluble; It dissolves easily in water, which means it can mix well with water without separating. |
| Boiling Point | -19°C (-2.2°F); This is the temperature at which formaldehyde changes from a liquid to a gas. It has a low boiling point, indicating it turns into a gas at temperatures lower than water does. |
| Melting Point | -92°C (-133.6°F); This is the temperature at which formaldehyde changes from a solid to a liquid. It shows that formaldehyde freezes at a much colder temperature compared to water. |
Chemical Properties of Formaldehyde
Reactivity with Oxygen
- Formaldehyde can react with oxygen in a process known as oxidation. This reaction often results in the formation of formic acid. It demonstrates formaldehyde’s ability to change and react under specific conditions, highlighting its reactive nature.
- Equation: 2 CH₂O + O₂ → 2 HCOOH
Polymerization Ability
- One of the most fascinating aspects of formaldehyde is its capacity to polymerize. This means it can join with itself to form larger molecules. This property is utilized in the creation of various resins and plastics, such as when formaldehyde molecules link to form paraformaldehyde, a solid polymer.
- Equation: nCH₂O → (CH₂O)n
Chemical Synthesis Component
- Formaldehyde serves as a crucial building block in the synthesis of many other chemicals and compounds. For instance, it reacts with phenol to produce phenol-formaldehyde resins. These resins are essential in adhesives, coatings, and insulation materials, showcasing the wide-ranging utility of formaldehyde in industrial applications.
- Equation: CH₂O + C₆H₅OH → Phenol-Formaldehyde Resins
Reactivity with Ammonia
- Another interesting reaction of formaldehyde is with ammonia, resulting in the production of urotropin (hexamethylenetetramine). This example further illustrates the chemical’s adaptability in reactions, producing compounds used in medicine, explosives, and as solid fuel.
- Equation: 6 CH₂O + 4 NH₃ → C₆H₁₂N₄ + 6 H₂O
Formaldehyde (CH₂O) Chemical Compound Information
Chemical Identifiers
| Property | Value |
|---|---|
| CAS Registry Number | 50-00-0 |
| Beilstein Number | 1209228 |
| PubChem Compound ID | 712 |
| PubChem Substance ID | 3367 |
| SMILES Identifier | C=O |
| InChI Identifier | InChI=1/CH2O/c1-2/h1H2 |
| InChI Key | WSFSSNUMVMOOMR-UHFFFAOYAT |
| MDL Number | MFCD00003274 |
NFPA Label
| Property | Value |
|---|---|
| NFPA Health Rating | 3 |
| NFPA Fire Rating | 2 |
| NFPA Reactivity Rating | 0 |
Uses of Formaldehyde
Preserving Biological Specimens
In science labs, formaldehyde is often used as a preservative. It helps keep biological specimens, like frogs or plants, in their original state for a long time, making it easier for students and researchers to study them.
Making Building Materials
Formaldehyde plays a crucial role in producing building materials, including particleboard, plywood, and fiberglass insulation. It acts as a binder, helping these materials hold their shape and maintain their strength.
Producing Household Products
Many household products, such as glues, paints, and even some cosmetics, contain formaldehyde. It helps these products perform better, like making glues more adhesive or ensuring paints dry properly.
In the Textile Industry
Formaldehyde is used in treating fabrics to prevent wrinkles and reduce mildew. This means your clothes can look crisp and fresh without needing to iron them frequently.
As a Disinfectant
Due to its ability to kill bacteria and fungi, formaldehyde is also used in some disinfectants and household cleaners. This makes it valuable in maintaining cleanliness and hygiene in various settings.
In Vaccine Production
Interestingly, formaldehyde has a role in creating certain vaccines. It inactivates viruses, making them safe to use in vaccines that help our bodies build immunity without causing the disease.
Side Effects of Formaldehyde
- Irritation to Eyes, Nose, and Throat: Formaldehyde can cause a stinging sensation in the eyes, along with coughing and sore throat, especially in poorly ventilated areas.
- Skin Irritation: Coming into direct contact with formaldehyde can lead to skin rashes and itching, signaling irritation or an allergic reaction.
- Respiratory Problems: Prolonged inhalation of formaldehyde vapors can aggravate respiratory conditions, leading to difficulty breathing and potentially exacerbating asthma symptoms.
- Nausea and Headache: High levels of formaldehyde exposure can cause nausea and headaches, making it uncomfortable for individuals to remain in affected environments.
- Cancer Risk: Long-term exposure to formaldehyde has been linked to an increased risk of certain types of cancer, particularly nasopharyngeal cancer and leukemia, as classified by the International Agency for Research on Cancer (IARC).
- Sensitization: Repeated exposure to formaldehyde can make individuals more sensitive to it over time, meaning even low levels could eventually trigger allergic reactions.
FAQ’S
What Does Formaldehyde Do to the Body?
Formaldehyde exposure can irritate the skin, eyes, nose, and throat, potentially leading to respiratory issues and worsening asthma symptoms in sensitive individuals.
Is Formaldehyde Harmful to Humans?
Yes, formaldehyde is harmful, especially with prolonged exposure. It can cause respiratory problems, skin irritation, and is linked to an increased risk of certain cancers.
Where is Formaldehyde Found in the Home?
Formaldehyde is found in pressed wood products, glues, paints, fabrics, and cosmetics, particularly in poorly ventilated indoor spaces.
What Are 4 Everyday Items That Contain Formaldehyde?
Everyday items containing formaldehyde include pressed wood furniture, adhesives, certain fabrics, and cosmetics like nail polish and hair smoothing products.



