Magnesium Hydroxide (Mg(OH)2) – Definition, Structure, Preparation, Properties, Uses
Magnesium Hydroxide is a compound in chemistry that belongs to the category of bases. It’s made up of magnesium, a shiny gray metal found in many minerals, and hydroxide, a combination of hydrogen and oxygen ions. When magnesium reacts with water or an acid, it forms magnesium hydroxide. This compound is commonly known as milk of magnesia and is often used in medicines to treat indigestion and heartburn because of its ability to neutralize stomach acids. Additionally, magnesium hydroxide is used in various industrial processes, such as water treatment and as a flame retardant in plastics.
What is Magnesium Hydroxide?
Chemical Names and Formulas
| Property | Value |
|---|---|
| Formula | Mg(OH)₂ |
| Hill Formula | H₂MgO₂ |
| Name | Magnesium hydroxide |
| IUPAC Name | Magnesium dihydroxide |
| Alternate Names | Magnesia magma, Magnesia, Magnesium dihydroxide, Magnesium hydrate , Milk of magnesia |
Structure of Magnesium Hydroxide
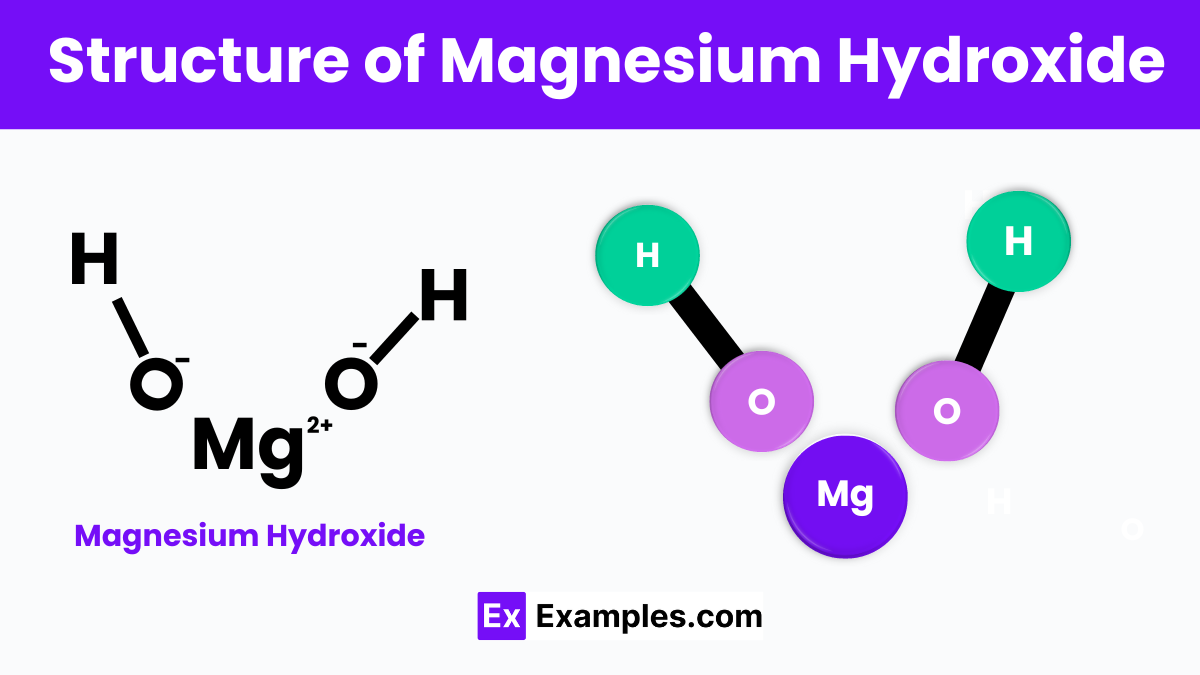
Magnesium Hydroxide is composed of magnesium ions (Mg²⁺) and hydroxide ions (OH⁻) arranged in a specific pattern. In its solid form, this compound forms layers where each magnesium ion is surrounded by six hydroxide ions, creating a sheet-like structure. These layers are held together by ionic bonds, which are forces of attraction between positively charged magnesium and negatively charged hydroxide ions. This arrangement results in a stable, crystalline structure that is typical of many minerals found in nature.
Preparation of Magnesium Hydroxide
Magnesium hydroxide can be prepared using a simple chemical reaction involving magnesium chloride (MgCl₂) and sodium hydroxide (NaOH). When these two compounds are mixed in water, they react to form magnesium hydroxide and sodium chloride (table salt). The chemical equation for this reaction is:
In this reaction, the magnesium chloride solution is gradually mixed with a solution of sodium hydroxide. As they react, magnesium hydroxide, which is only slightly soluble in water, forms as a white, solid precipitate. This precipitate can then be separated from the remaining liquid by filtration or settling and drying. This method is commonly used in laboratories and industrial processes to produce magnesium hydroxide for various applications, including medical uses and as a fire retardant.
Physical Properties of Magnesium Hydroxide
| Property | Description |
|---|---|
| Appearance | White, odorless powder |
| Solubility in Water | Slightly soluble, more soluble in water containing CO₂ |
| Density | 2.34 grams per cubic centimeter |
| Melting Point | Decomposes at about 350°C (does not melt) |
| Boiling Point | Not applicable as it decomposes before boiling |
| Thermal Conductivity | Low, similar to other ceramic materials |
| pH | Highly alkaline (around 10.5 in a saturated solution) |
Chemical Properties of Magnesium Hydroxide
Reactivity with Acids
- Mg(OH)₂ reacts with acids in a neutralization reaction, which results in the formation of a magnesium salt and water. This reaction is commonly used in medicine to neutralize stomach acid. For example, when magnesium hydroxide reacts with hydrochloric acid (HCl), commonly found in the stomach, magnesium chloride (MgCl₂) and water are produced.
- Equation: Mg(OH)₂ + 2HCl → MgCl₂ + 2H₂O
Thermal Decomposition
- When heated, Mg(OH)₂decomposes into Magnesium oxide (MgO) and water vapor. This decomposition starts at about 350°C, making it useful in fire retardant applications because it releases water vapor which helps to extinguish flames.
- Equation: Mg(OH)₂ → MgO + H₂O (steam)
Reaction with Carbon Dioxide
- Mg(OH)₂ can also react with carbon dioxide (CO₂) to form MgCO₃ (Magnesium Carbonate). This reaction can occur naturally over time, especially when Mg(OH)₂ is exposed to air.
- Equation: Mg(OH)₂ + CO₂ → MgCO₃ + H₂O
Magnesium Hydroxide (Mg(OH)₂) Chemical Compound Information
Chemical Identifiers
| Property | Value |
|---|---|
| CAS Registry Number | 1309-42-8 |
| PubChem Compound ID | 14791 |
| PubChem Substance ID | 24882320 |
| SMILES Identifier | [OH-].[OH-].[Mg+2] |
| InChI Identifier | InChI=1/Mg.2H2O/h;21H2/q+2;;/p-2/fMg.2HO/h;21h/qm;2*-1 |
| RTECS Number | OM3570000 |
| MDL Number | MFCD00011104 |
NFPA Label
| Property | Value |
|---|---|
| NFPA Health Rating | 1 |
| NFPA Fire Rating | 0 |
| NFPA Reactivity Rating | 0 |
Uses of Magnesium Hydroxide
Antacid and Laxative
Magnesium Hydroxide is widely used as an antacid to relieve heartburn, upset stomach, and indigestion by neutralizing stomach acid. It also acts as a laxative to alleviate constipation. Its gentle action on the bowels makes it a popular choice for regular use, helping to maintain digestive comfort.
Fire Retardant
In the production of materials that require fire resistance, Magnesium Hydroxide is used as a fire retardant. It decomposes at high temperatures to release water vapor, which helps to cool and slow the spread of fire, making it an essential component in safety equipment and construction materials.
Wastewater Treatment
Magnesium Hydroxide is employed in environmental applications, particularly in the treatment of wastewater. It adjusts the pH of waste water to neutralize acids and remove heavy metals through precipitation, improving the safety and cleanliness of water before it is released back into the environment.
Pharmaceutical Ingredient
Magnesium Hydroxide is used as an ingredient in various products, including antiperspirants and skin care items. Its ability to neutralize acid and soothe makes it beneficial in formulations designed to reduce body odor and treat skin conditions.
Side Effects of Magnesium Hydroxide
- Diarrhea: One of the most common side effects of Mg(OH)₂, especially when used as a laxative.
- Stomach Cramps: Some people may experience mild to moderate stomach cramps after taking magnesium hydroxide.
- Nausea: Feelings of nausea or upset stomach can occur, although they are usually temporary and mild.
- Electrolyte Imbalance: Long-term use can lead to an imbalance in body electrolytes, such as potassium and sodium, which can affect overall health.
- Decreased Absorption of Other Medications: Magnesium hydroxide can interfere with the absorption of other medications, reducing their effectiveness.
FAQs
Is Magnesium Hydroxide Safe for Humans?
Yes, It is safe for humans when used as directed for relieving indigestion and constipation.
What Does Magnesium Hydroxide Do for Your Health?
It acts as an antacid and laxative, easing heartburn, indigestion, and constipation effectively.
Who Cannot Take Magnesium Hydroxide?
Individuals with kidney disease, severe abdominal pain, or dehydration.
Is Magnesium Hydroxide Cancerous?
No, it is not cancerous. It is considered safe and is commonly used in medical treatments.


