Methanol (CH₃OH) – Definition, Structure, Preparation, Properties, Uses, Side Effects
Methanol, often referred to in chemistry as a simple yet fascinating molecular compound, is like a building block in the vast world of science. Think of it as a kind of alcohol, but not the kind you find in beverages. Instead, methanol is used in making various products, from plastics to paints, and even as fuel for some racing cars. Its structure is quite straightforward, composed of one carbon atom connected to three hydrogen atoms and one oxygen atom, which is also bonded to a hydrogen atom. This neat arrangement makes methanol a key player in many chemical reactions, showcasing the incredible versatility and importance of molecular compounds in our daily lives and the broader field of chemistry.
What is Methanol?
Chemical Names and Formulas
| Property | Value |
|---|---|
| Formula | CH₃OH |
| Hill formula | CH₄O |
| Name | Methanol |
| Alternate Names | Carbinol, Colonial spirit, Methyl alcohol, Methyl hydroxide, Methylol, Pyroxylic spirit, Wood alcohol, Wood naphtha, Wood spirit |
Structure of Methanol
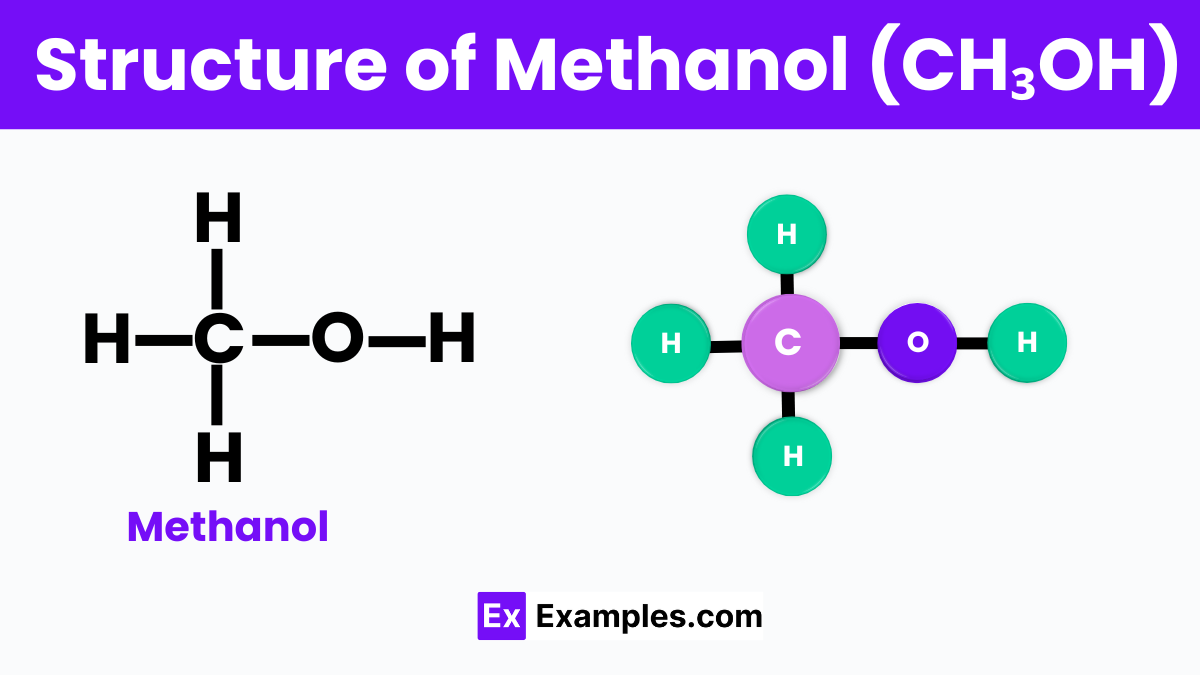
Methanol, also known as wood alcohol, has a simple structure that makes it a key player in the world of chemistry. At its core, methanol is made up of one carbon atom linked to three hydrogen atoms and one oxygen atom that’s also connected to a hydrogen atom. Think of it as a little family, with the carbon atom being the parent and the hydrogen and oxygen atoms as the kids holding hands. This arrangement gives methanol its unique properties, allowing it to mix well with water and be used in a variety of products, from fuel to solvents. Its simple structure is a gateway to understanding more complex chemical compounds, making methanol a fundamental molecule in both science classrooms and industries.
Preparation of Methanol
Methanol, a simple but vital chemical, is made through a process that transforms natural gas into something much more versatile. This transformation is like a magic trick; starting with methane, the main component of natural gas, we add a little oxygen to the mix. When methane (CH₄) and oxygen (O₂) are heated up together in the presence of a catalyst—a substance that speeds up the reaction without being consumed—they undergo a chemical change. The end result? Methanol (CH₃OH) and water (H₂O). The chemical equation for this fascinating process looks like this:
In simpler terms, think of it as baking a cake. Just as you mix ingredients like flour, eggs, and sugar to create a delicious treat, chemists mix methane with oxygen under the right conditions to ‘bake’ methanol. This process, known as steam reforming, is widely used in industries around the world to produce methanol on a large scale. Methanol is then used in making various products, from fuels to plastics, showcasing its importance in our daily lives and industries.
Physical Properties of Methanol
| Property | Description |
|---|---|
| Appearance | Methanol is a clear, colorless liquid. It looks a lot like water, so you can’t tell them apart just by looking. |
| Smell | It has a slightly sweet odor, but it’s usually hard to notice unless the concentration is high. |
| Boiling Point | Methanol boils at about 64.7°C (148.5°F). This means it turns into a gas at temperatures lower than boiling water. |
| Freezing Point | It freezes at -97.6°C (-143.7°F), which is way colder than water’s freezing point. So, it stays liquid in extremely cold temperatures. |
| Solubility in Water | Methanol mixes perfectly with water in all proportions. It’s like how sugar dissolves in tea, blending completely. |
| Density | It’s less dense than water, with a density of about 0.791 g/cm³ at 20°C (68°F). This means if you had a bottle of water and a bottle of methanol, the methanol would be lighter. |
Chemical Properties of Methanol
Flammability
- Methanol is highly flammable, making it a valuable fuel but also requiring careful handling. It can catch fire easily when exposed to an open flame or high heat. During combustion, methanol reacts with oxygen to produce carbon dioxide and water, releasing energy in the process.
- Equation: 2 CH₃OH + 3 O₂ → 2 CO₂ + 4 H₂O
Reactivity with Acids and Bases
- Methanol reacts with both acids and bases, showcasing its versatility. With acids, it forms esters, which are compounds often used in perfumes and flavors due to their sweet smell. With bases, methanol can form methoxide ions, which have various applications in chemical synthesis.
- Equation: CH₃OH + CH₃COOH → CH₃COOCH₃ + H₂O
Toxicity upon Oxidation
- When oxidized, methanol transforms into formaldehyde and then into formic acid. This oxidation process highlights methanol’s toxic nature when ingested, as both formic acid and formaldehyde can harm the human body, particularly affecting the nervous system and vision.
- Equation: CH₃OH + O₂ → HCHO + H₂O, followed by HCHO + O₂ → HCOOH.
Solubility in Water
Methanol is completely miscible with water, meaning it can be mixed with water in any amount, leading to a clear solution. This property makes methanol an excellent solvent for a wide range of substances, including organic compounds and salts. This solubility plays a critical role in its various applications in industrial and chemical processes.
Methanol (CH₃OH) Chemical Compound Information
Chemical Identifiers
| Property | Value |
|---|---|
| CAS Registry Number | 67-56-1 |
| Beilstein Number | 1098229 |
| PubChem Compound ID | 887 |
| PubChem Substance ID | 24850836 |
| SMILES Identifier | CO |
| InChI Identifier | InChI=1/CH4O/c1-2/h2H, 1H3 |
| RTECS Number | PC1400000 |
| MDL Number | MFCD00004595 |
NFPA Label
| Property | Value |
|---|---|
| NFPA Health Rating | 1 |
| NFPA Fire Rating | 3 |
| NFPA Reactivity Rating | 0 |
Uses of Methanol

Fuel for Vehicles
Methanol is a clean-burning fuel that can power cars and other vehicles, offering a more environmentally friendly alternative to traditional gasoline. It’s used in special racing cars due to its high octane rating and ability to burn cooler, which improves performance.
Solvent in Laboratories
In labs around the world, methanol serves as a crucial solvent. Its ability to dissolve a wide range of chemical compounds makes it indispensable for scientists creating medicines, testing samples, or conducting research.
Antifreeze in Winter Products
Methanol’s low freezing point makes it an excellent choice for antifreeze products. It helps keep car engines and other machinery running smoothly in cold weather, preventing water-based fluids from freezing.
Ingredient in Windshield Washer Fluid
Thanks to its cleaning power and antifreeze properties, methanol is a key component in windshield washer fluid. It cuts through dirt and grime on car windshields and prevents the fluid from freezing in winter.
Production of Formaldehyde and Other Chemicals
Methanol is a primary ingredient in the production of formaldehyde, a chemical used in making building materials, textiles, and many household products. It also serves as a starting point for synthesizing a variety of other important chemicals.
Energy Carrier
Methanol holds promise as an energy carrier for the future. It can store hydrogen energy, making it easier to transport and use hydrogen fuel, a potential key to unlocking cleaner energy sources.
Side Effects of Methanol
- Blurred Vision or Blindness: Methanol can harm the optic nerve, leading to vision problems or even permanent blindness. This is one of the most serious risks of ingesting or being exposed to methanol.
- Headaches and Dizziness: Breathing in methanol vapors can cause headaches and dizziness. It’s like feeling sick after spinning around too fast, but it doesn’t go away quickly.
- Nausea and Vomiting: Swallowing methanol, even in small amounts, can upset your stomach, leading to nausea and vomiting. It’s as if your body is trying to get rid of something harmful.
- Difficulty Breathing: High methanol vapor exposure can cause severe breathing difficulties, akin to intense breathlessness experienced after strenuous exercise.
- Skin and Eye Irritation: Methanol can irritate your skin and eyes. Imagine chopping onions, but the sting doesn’t stop when you walk away.
- Fatigue and Weakness: Methanol exposure can induce extreme fatigue, resembling days of sleep deprivation.
FAQ’S
Is Methanol Poisonous to Humans?
Yes, methanol is toxic to humans. Ingesting or inhaling it can lead to serious health issues, including blindness, organ failure, and even death.
Why is Methanol Banned?
Methanol is banned in consumer products due to its toxicity. Accidental ingestion or exposure can result in fatal poisoning, prompting strict regulatory measures.
Is Methanol and Alcohol the Same Thing?
Methanol and ethanol (drinking alcohol) are different. Methanol is toxic and used industrially, while ethanol is safe in moderation and found in beverages.
What Alcohol has the Least Methanol?
Beverages like beer and wine have the least methanol, thanks to strict distillation and fermentation processes that minimize methanol content for safe consumption.



