Sulfuric Acid (H₂SO₄) – Definition, Structure, Preparation, Uses, Benefits, Side Effects
Sulfuric acid is a powerful substance that plays a big role in the world of chemistry. It’s a covalent compound, which means it’s made up of atoms that share electrons to stick together. This clear, colorless liquid is known for its unique ability to react with other substances, making it super important in making lots of things we use every day.
What is Sulfuric Acid?
Chemical Names and Formulas
| Property | Value |
|---|---|
| Formula | H₂SO₄ |
| Hill formula | H₂O₄S |
| Name | Sulfuric acid |
| Alternate names | Anhydrous sulfuric acid, Dihydrogen sulfate, Oil of vitriol, Sulphuric acid |
Structure of Sulfuric Acid
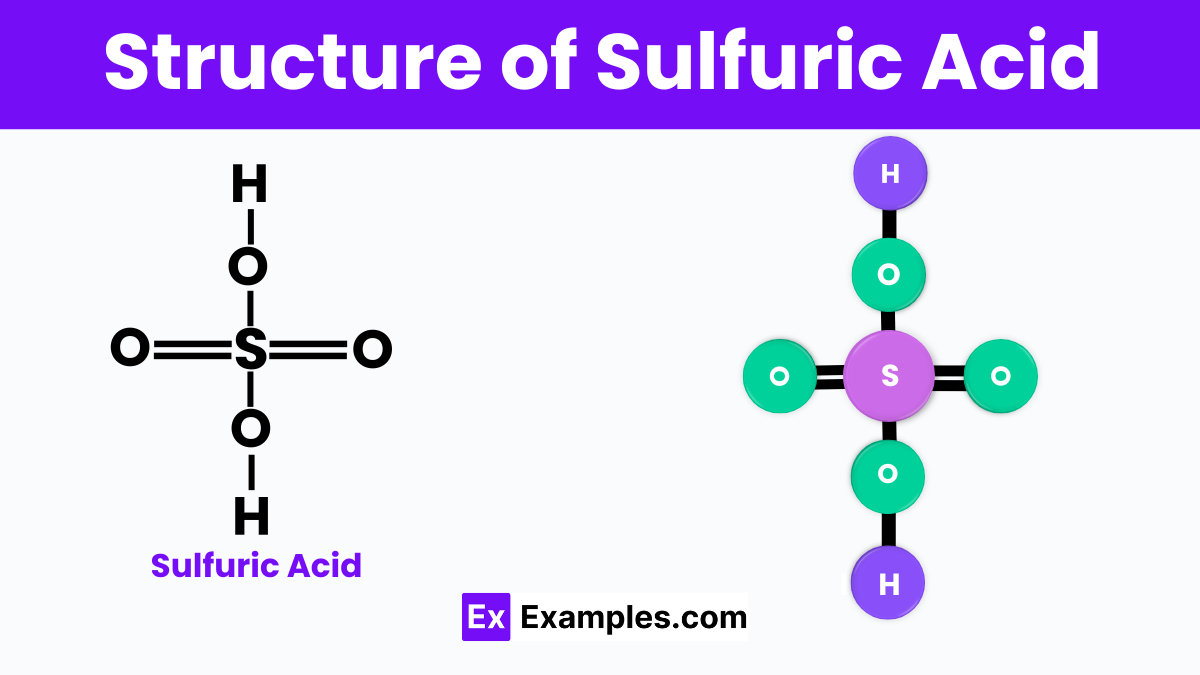
Sulfuric acid is a chemical compound with a complex and interesting structure. It has the chemical formula H₂SO₄. This means it is made up of two hydrogen atoms (H), one sulfur atom (S), and four oxygen atoms (O). The structure of sulfuric acid can be thought of as a central sulfur atom surrounded by four oxygen atoms in a tetrahedral arrangement. Two of these oxygen atoms are double-bonded to the sulfur atom, and the other two are single-bonded but also attached to hydrogen atoms.This arrangement makes sulfuric acid a very strong acid. It’s able to donate two protons (the hydrogen atoms) when it reacts with other substances. This ability to give away protons easily makes it a very important and widely used chemical in many industries. Its structure is key to its reactivity and usefulness in making a wide variety of products.
Preparation of Sulfuric Acid
Sulfuric acid is made using a method called the Contact Process. This process involves a few key steps. First, sulfur (S) is burned in air to create sulfur dioxide (SO₂). This is represented by the equation:
Next, the sulfur dioxide is mixed with more oxygen (O₂) in the presence of a catalyst, typically vanadium(V) oxide (V₂O₅). This helps convert the sulfur dioxide into sulfur trioxide (SO₃), shown by the equation:
Once sulfur trioxide is formed, it’s not directly combined with water to make sulfuric acid because that reaction is too violent. Instead, the sulfur trioxide is first absorbed into a pre-existing concentration of sulfuric acid. This forms oleum (H₂S₂O₇), which is then carefully diluted with water to form sulfuric acid (H₂SO₄). The final step can be represented by the equation:
This careful process allows for the production of highly concentrated sulfuric acid, which is essential for many industrial applications.
Physical Properties of Sulfuric Acid
| Property | Description |
|---|---|
| Appearance | Colorless, oily liquid |
| Odor | Odorless |
| Molecular Weight | 98.079 g/mol |
| Density | 1.84 g/cm³ at 20°C |
| Melting Point | 10°C (50°F) |
| Boiling Point | 337°C (639°F) |
| Solubility in Water | Completely soluble, releases heat upon mixing |
| pH | Highly acidic (pH < 1 for concentrated solutions) |
| Viscosity | High, varies with concentration |
| Conductivity | Good conductor of electricity due to ionization in water |
Chemical Properties of Sulfuric Acid
Strong Acid
Sulfuric acid is a very strong acid. It can donate two protons (H⁺ ions) in reactions. This makes it very reactive with bases and capable of causing burns.
Hygroscopic
It has the ability to absorb water from the air. This property makes it useful for drying out substances.
Dehydrating Agent
Sulfuric acid can remove water from other compounds. For example, when it reacts with sugar (C₁₂H₂₂O₁₁), it removes water, leaving a black mass of carbon and releasing water vapour.
Equation: C₁₂H₂₂O₁₁ + H₂SO₄ → 12C + 11H₂O + Heat
Exothermic Reactions
Mixing sulfuric acid with water releases a lot of heat. This reaction should be done carefully by adding acid to water, not the other way around, to avoid splashing of the hot acid.
Oxidizing Agent
In concentrated form, sulfuric acid can act as a powerful oxidizing agent. It can take electrons from other substances, leading to oxidation. This property allows it to react with metals, non-metals, and carbon compounds, often producing sulfur dioxide (SO₂) or sulfates.
Reaction with Metals
Sulfuric acid reacts with most metals, producing hydrogen gas and metal sulfate. For example, zinc reacts with sulfuric acid to produce zinc sulfate and hydrogen gas.
Reaction with Bases
It reacts with bases (like sodium hydroxide) to form water and a salt. This reaction with sodium hydroxide (NaOH) produces water and sodium sulfate.
Equation: 2NaOH → Na₂SO₄ + 2H₂O
Sulfuric Acid (H₂SO₄) Chemical Compound Information
Chemical Identifiers
| Property | Value |
|---|---|
| CAS registry number | 7664-93-9 |
| Beilstein number | 2037554 |
| PubChem compound ID | 1118 |
| PubChem substance ID | 24859176 |
| SMILES identifier | OS(=O)(=O)O |
| InChI identifier | InChI=1/H2O4S/c1-5(2,3)4/h(H2,1,2,3,4)/f/h1-2H |
| MDL number | MFCD00064589 |
NFPA label
| Property | Value |
|---|---|
| NFPA health rating | 3 |
| NFPA fire rating | 0 |
| NFPA reactivity rating | 2 |
| NFPA hazards | Water reactive |
Uses of Sulfuric Acid (H₂SO₄)
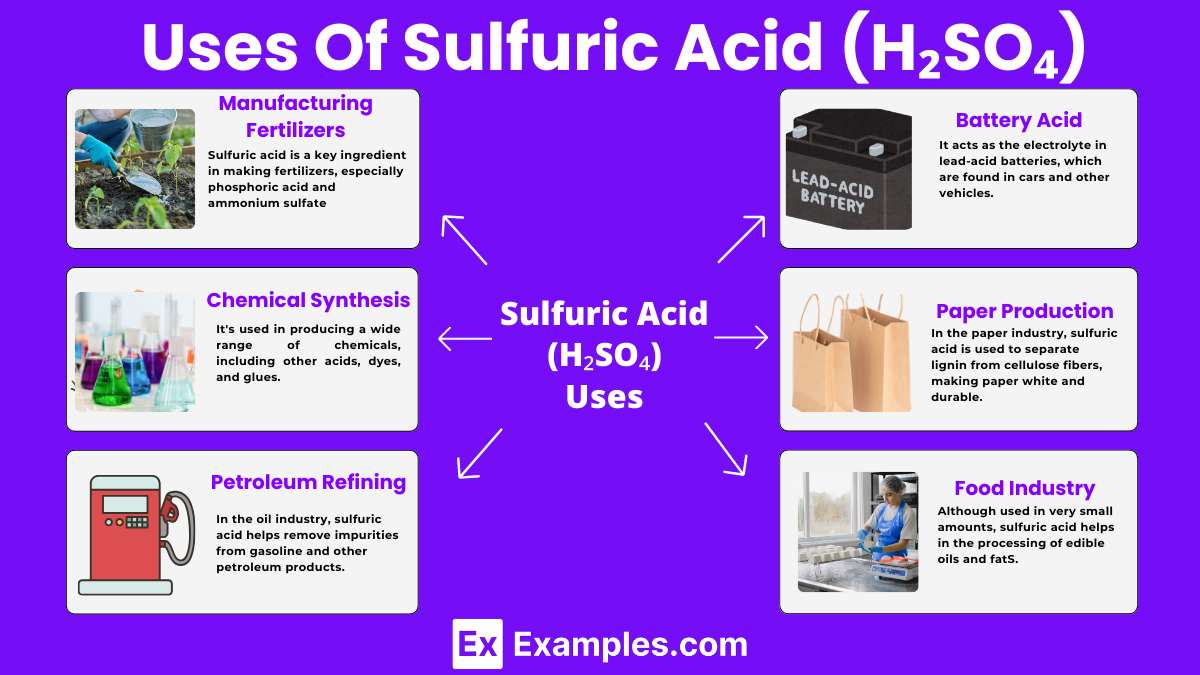
Manufacturing Fertilizers
Sulfuric acid is a key ingredient in making fertilizers, especially phosphoric acid and ammonium sulfate. These fertilizers help plants grow by providing them essential nutrients.
Chemical Synthesis
It’s used in producing a wide range of chemicals, including other acids, dyes, and glues. This process involves reactions where sulfuric acid helps create new compounds.
Petroleum Refining
In the oil industry, sulfuric acid helps remove impurities from gasoline and other petroleum products. This refining process ensures the fuel is clean and efficient.
Metal Processing
Sulfuric acid is used to clean metals before they are coated or plated with other metals. It removes rust and prepares the surface for further treatment.
Battery Acid
It acts as the electrolyte in lead-acid batteries, which are found in cars and other vehicles. The acid allows electricity to flow between the battery’s plates.
Water Treatment
Sulfuric acid is used in water treatment plants to adjust the pH levels of water, making it safe for drinking and use in industrial processes.
Paper Production
In the paper industry, sulfuric acid is used to separate lignin from cellulose fibers, making paper white and durable.
Pharmaceuticals
It plays a role in manufacturing various medicines, acting as a catalyst in the production processes of drugs and antibiotics.
Food Industry
Although used in very small amounts, sulfuric acid helps in the processing of edible oils and fats. It also acts as a preservative in some food products.
Benefits of Sulfuric Acid (H₂SO₄)
Agricultural Productivity
- Fertilizer Production: Sulfuric acid is crucial in manufacturing fertilizers, which enhance soil nutrition and boost crop yields significantly.
Industrial Applications
- Chemical Synthesis: It serves as a fundamental ingredient in the synthesis of various chemicals, facilitating the production of dyes, pigments, and many other industrial compounds.
- Metal Processing: The acid’s ability to clean metal surfaces ensures better adhesion of coatings, improving the longevity and quality of metal products.
Energy Sector
- Battery Efficiency: As the electrolyte in lead-acid batteries, sulfuric acid is essential for the storage and provision of electrical energy, particularly in vehicles and renewable energy storage systems.
Environmental Protection
- Water Treatment: By adjusting pH levels, sulfuric acid plays a pivotal role in water purification processes, making water safe for consumption and use in industries.
- Pollution Control: It aids in removing harmful substances from industrial emissions, contributing to cleaner air and a healthier environment.
Manufacturing
- Petroleum Refining: The acid helps refine petroleum products, ensuring cleaner burning fuels and reducing emissions from vehicles.
- Paper Production: Its use in the paper industry leads to higher quality paper by removing impurities and enhancing the brightness and strength of the paper.
Healthcare and Pharmaceuticals
- Drug Manufacturing: Sulfuric acid is used in the pharmaceutical industry to produce various medicines, helping in the fight against diseases and infections.
Food Industry
- Food Processing: Though used in minimal quantities, it assists in the processing and preservation of certain food products, maintaining their quality and safety.
Side Effects Of Sulfuric Acid (H₂SO₄)
- Health Hazards: Causes severe skin burns, eye damage, respiratory issues, and digestive tract harm upon contact or inhalation.
- Environmental Impact: Can lead to water and soil pollution, contributing to ecosystem damage and soil infertility.
- Material Corrosion: Corrodes metals and building materials, posing infrastructure risks.
- Safety Risks: Improper handling and storage can result in accidents and exposure.
FAQ’S
What Happens When You Add Water to Sulfuric Acid?
Adding water to sulfuric acid can cause violent reactions, emitting heat and potentially leading to splashes or explosions. Always add acid to water instead.
What Does Sulfuric Acid Do to Skin?
Sulfuric acid burns skin upon contact, causing severe chemical burns, tissue damage, and possible scarring. Immediate rinsing with water is crucial for affected areas.
What Does Sulfuric Acid Smell Like?
Sulfuric acid has a strong, pungent odor resembling burnt matches, due to the release of sulfur oxides when exposed to air, indicating its corrosive nature.
What Happens if Sulfuric Acid Touches You?
Contact with sulfuric acid results in chemical burns, skin irritation, and potential tissue damage. Immediate and thorough washing with water is necessary to mitigate injuries.


