Einsteinium (Es) – Definition, Preparation, Properties, Uses, Compounds, Reactivity
Embark on an exploratory journey into the atomic world of Einsteinium, a synthetic element shrouded in the mysteries of nuclear science. This complete guide unveils the enigmatic properties, production methods, and the pivotal role of Einsteinium in advancing scientific knowledge. With examples ranging from its discovery in the aftermath of nuclear tests to its contributions to research in chemistry and physics, we delve into how Einsteinium serves as a key to unlocking profound insights into the structure of the atomic nucleus and the potential for new chemical innovations. Discover the intriguing applications and challenges of harnessing Einsteinium, an element that encapsulates the relentless pursuit of scientific discovery.
What is Einsteinium?
Einsteinium has no stable isotopes, with Einsteinium-252 being one of its most studied isotopes due to its relatively longer half-life of about 471.7 days. The element exhibits chemical properties typical of the actinide series and is primarily used for research purposes. Scientists study einsteinium to learn more about its properties and to synthesize new elements and compounds in the lab. Due to its scarcity and radioactivity, practical applications of einsteinium outside of scientific research are limited
Other Actinides
| Actinium | Berkelium |
| Thorium | Californium |
| Protactinium | Fermium |
| Uranium | Curium |
| Neptunium | Mendelevium |
| Plutonium | Nobelium |
| Americium | Lawrencium |
Einsteinium Formula
- Formula: Es
- Composition: Consists of a single einsteinium atom.
- Bond Type: In its elemental form, einsteinium does not form bonds as it is a pure element. However, einsteinium can form covalent or ionic bonds when reacting with other elements.
- Molecular Structure: As a pure element, einsteinium does not form a molecular structure in the same way as compounds like H₂. At room temperature, einsteinium is in a metallic state with a face-centered cubic (fcc) crystalline structure, typical for actinides.
- Electron Sharing: In compounds, einsteinium typically shares electrons covalently or transfers electrons ionically, depending on the nature of the other element(s) it is bonding with. It often exhibits a +3 oxidation state in its compounds, facilitating various types of chemical bonds.
- Significance: Einsteinium is notable primarily for its use in scientific research rather than for industrial or commercial applications. Its discovery was a significant milestone in the exploration of transuranic elements, contributing to our understanding of the actinide series and nuclear chemistry.
- Role in Chemistry: Einsteinium plays a significant role in research and the study of the actinide series. Its radioactive properties and ability to form compounds with various oxidation states make it a subject of interest for studying the chemistry of heavy elements and their potential applications in fields like nuclear science.
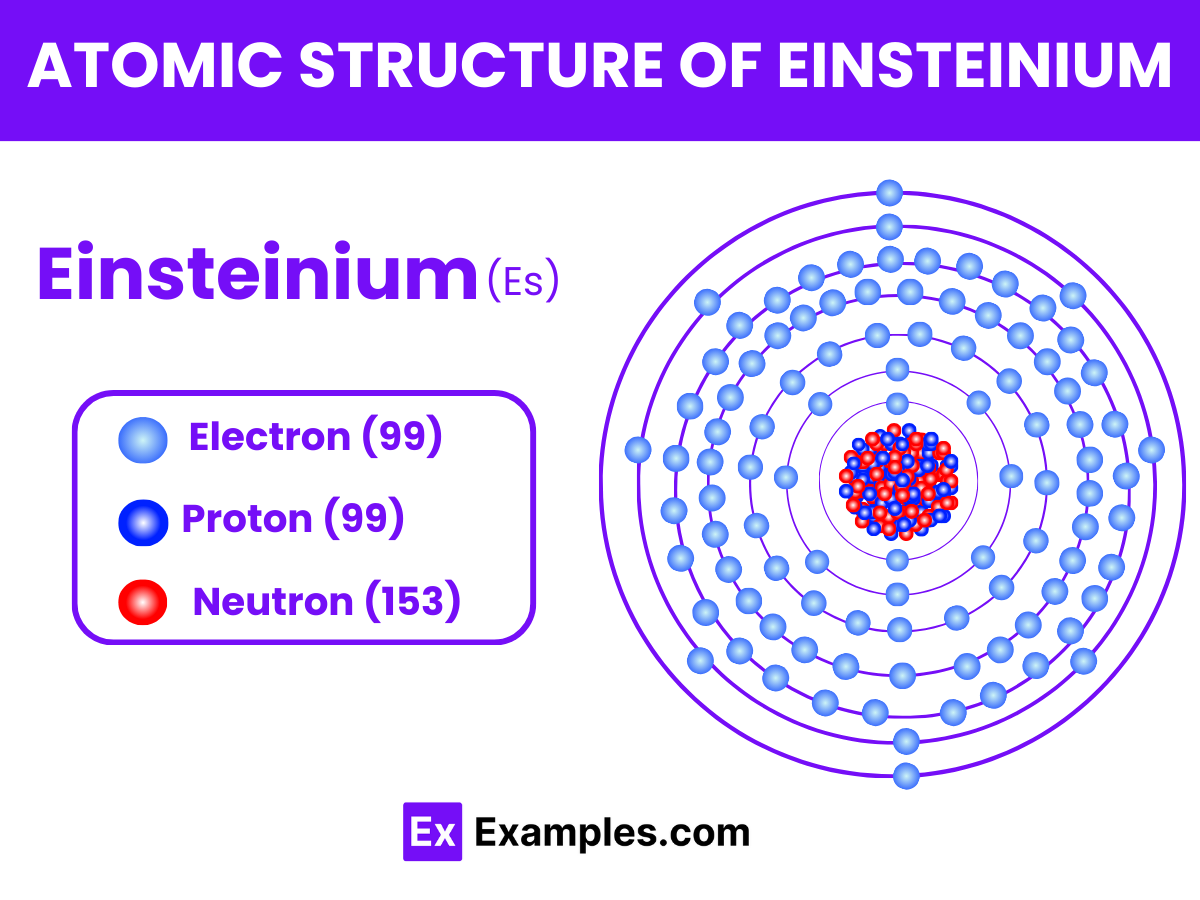
Atomic Structure of Einsteinium
Einsteinium, with the symbol Es and atomic number 99, is a synthetic element that belongs to the actinide series of the periodic table. Known for its radioactivity and synthetic origin, Einsteinium has a complex atomic structure that contributes to its unique chemical and physical properties. Here’s a breakdown of the atomic structure of Einsteinium:
Atomic Number and Symbol
- Atomic Number (Z): 99, indicating the presence of 99 protons in its nucleus.
- Symbol: Es
Atomic Mass
- Typical Isotopes: Einsteinium has no stable isotopes. The most commonly studied isotope is Einsteinium-253, with an atomic mass of approximately 253 atomic mass units (amu).
Electronic Configuration
- Ground State Configuration: The electron configuration of Einsteinium in its ground state is [Rn]5f¹¹7s². This shows that it has electrons in the 5f and 7s orbitals, similar to other actinides.
Isotopes
- Number of Isotopes: Einsteinium has over 19 isotopes, all of which are radioactive.
- Most Stable Isotope: Einsteinium-252, with a half-life of about 471.7 days, is one of its most stable isotopes.
Nuclear Properties
- Radioactivity: All isotopes of Einsteinium are radioactive, primarily undergoing alpha decay.
- Neutrons: The number of neutrons varies across its isotopes, affecting their stability and half-life.
Chemical Properties
- Oxidation States: Einsteinium commonly exhibits a +3 oxidation state, similar to other actinides.
- Reactivity: It reacts with oxygen, halogens, and acids, forming compounds like Einsteinium oxide (Es2O₃), Einsteinium chloride (EsCl₃), and others.
Physical Properties
- Appearance: Einsteinium is expected to be a silvery metal, though only microscopic amounts have been produced, making its physical properties difficult to observe directly.
- Phase at Room Temperature: Solid, assuming sufficient quantity for observation.
Safety and Handling
- Radiation Precautions: Due to its high radioactivity, Einsteinium requires specialized handling procedures to protect researchers from radiation exposure.
- Use in Controlled Environments: Its use and study are restricted to highly controlled environments, like nuclear facilities and research laboratories specializing in radioactive materials.
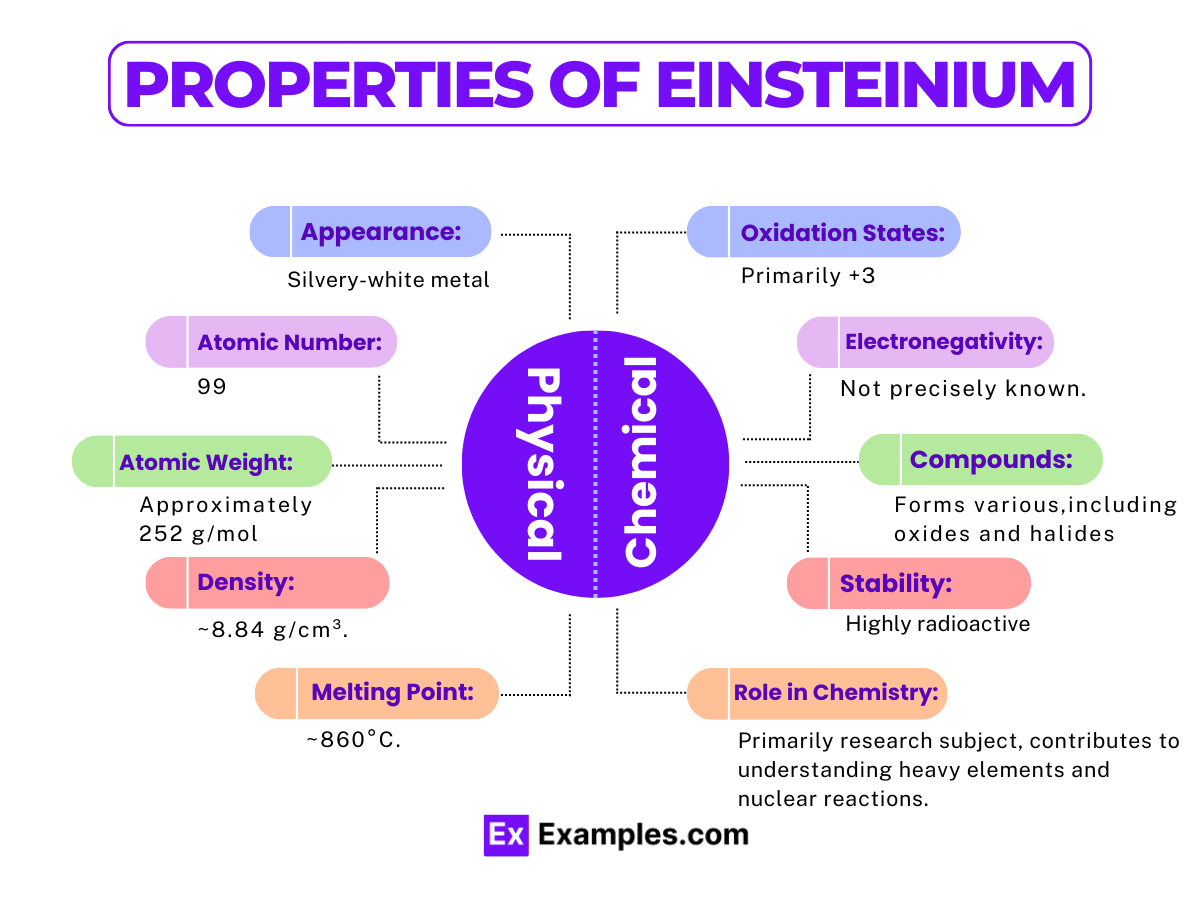
Properties of Einsteinium
Physical Properties of Einsteinium
| Property | Value |
|---|---|
| Appearance | Silvery, lustrous, almost white metal |
| Phase at Room Temperature | Solid |
| Density | Approx. 8.84 g/cm³ (estimated) |
| Melting Point | 860°C (estimated) |
| Boiling Point | Estimated to be around 996°C |
| Atomic Mass | 252 u (most stable isotope, Einsteinium-252) |
| Crystal Structure | Face-centered cubic (fcc) |
| State of Matter | Radioactive metal |
Chemical Properties of Einsteinium
Einsteinium, with the symbol Es and atomic number 99, is a synthetic, highly radioactive element belonging to the actinide series. Its chemical properties share similarities with other actinides, exhibiting complex behavior due to its radioactivity and position in the periodic table. Here we explore the detailed chemical properties of Einsteinium, focusing on its oxidation states, reactions, and notable compounds.
Oxidation States
- Primary Oxidation State: +3
- Einsteinium most commonly exhibits a +3 oxidation state in its compounds, similar to many actinides.
- Possible Oxidation States: It is theorized that Einsteinium can also exhibit +2 and +4 oxidation states under certain conditions, although these are less stable and less commonly observed.
Chemical Reactions
- Reaction with Oxygen:
- Equation:
- Einsteinium reacts with oxygen to form einsteinium(III) oxide (Es2O₃), demonstrating its stable +3 oxidation state.
- Reaction with Halogens:
- Equation:
- In reactions with halogens, Einsteinium forms halides such as einsteinium(III) fluoride (EsF3), again indicating the +3 oxidation state. Similar reactions can occur with chlorine, bromine, and iodine to form the respective halides.
- Reaction with Acids:
- Equation:
- Einsteinium dissolves in hydrochloric acid to produce einsteinium(III) chloride (EsCl3) and hydrogen gas, showcasing its reactivity with acids.
Complexation and Solubility
- Complex Formation: Like other actinides, Einsteinium can form complex ions with various ligands, which is important in its separation and purification processes.
- Solubility: Einsteinium compounds, particularly the halides and oxides, have specific solubilities in water and other solvents, which are crucial for their chemical manipulation and study.
Radioactivity and Stability
- The radioactive decay of Einsteinium (predominantly alpha decay) significantly affects its chemical stability and properties. The handling and study of Einsteinium compounds require stringent safety measures due to its high radioactivity
Thermodynamic Properties of Einsteinium
| Property | Value |
|---|---|
| Melting Point | Approximately 860°C |
| Boiling Point | Estimated to be around 996°C |
| Heat of Fusion | Data not available |
| Heat of Vaporization | Data not available |
| Specific Heat Capacity | Data not available |
Material Properties of Einsteinium
| Property | Value |
|---|---|
| State at Room Temperature | Solid |
| Density | Estimated to be around 8.84 g/cm³ |
| Appearance | Silver-colored, radioactive metal |
| Crystal Structure | Face-centered cubic (estimated) |
Electromagnetic Properties of Einsteinium
| Property | Value |
|---|---|
| Electrical Resistivity | High (specific value not available) |
| Magnetic Ordering | Paramagnetic |
| Thermal Conductivity | Data not available |
Nuclear Properties of Einsteinium
| Property | Value |
|---|---|
| Most Stable Isotope | Einsteinium-252 (half-life of 471.7 days) |
| Primary Decay Modes | Alpha decay |
| Neutron Cross Section | Data not available |
| Critical Mass | Not applicable due to production in microgram quantities |
Preparation of Einsteinium
The preparation of Einsteinium (Es) involves complex nuclear reactions, primarily occurring within high-flux nuclear reactors or during the detonation of nuclear weapons. Here’s a step-by-step overview of how Einsteinium is typically produced:
- Neutron Bombardment: The process begins with the bombardment of heavy elements like plutonium, americium, or curium with neutrons in a nuclear reactor. This environment provides the intense neutron flux necessary for the successive neutron capture required to produce heavier elements.
- Nuclear Reactions: Through a series of neutron capture reactions, the bombarded atoms gain neutrons and undergo beta decay, transforming into heavier elements. For instance, the bombardment of Uranium-238 can eventually lead to the formation of Einsteinium after multiple neutron captures and decay processes.
- Chemical Separation: Following irradiation, the resulting material contains a mixture of different actinides, including the newly formed Einsteinium. Chemical separation techniques are employed to isolate Einsteinium from this mixture. Processes such as ion exchange chromatography are commonly used, taking advantage of the distinct chemical properties of each element to achieve separation.
- Purification: The isolated Einsteinium is then further purified to remove any residual contaminants. This step is crucial for obtaining pure samples for research, as the presence of other elements can interfere with experimental results.
- Formulation: Finally, the purified Einsteinium is converted into a form suitable for study and application, such as Einsteinium oxide (Es2O₃) or Einsteinium chloride (EsCl₃). The choice of compound depends on the intended use in research or experimental applications
Chemical Compounds of Einsteinium
1. Einsteinium Oxide (Es₂O₃)
Description: A basic oxide, demonstrating Einsteinium’s ability to form compounds with oxygen. Equation:
2. Einsteinium Chloride (EsCl₃)
Description: Forms by reacting Einsteinium with chlorine, showing +3 oxidation state.
Equation: 3
3. Einsteinium Iodide (EsI₃)
Description: A compound indicating Einsteinium’s reactivity with halogens.
Equation: 3
4. Einsteinium Fluoride (EsF₃)
Description: Showcases Einsteinium’s ability to form stable halide compounds.
Equation: 3
5. Einsteinium Bromide (EsBr₃)
Description: Highlights Einsteinium’s consistent +3 oxidation state with halogens.
Equation: 3
6. Einsteinium Hydroxide (Es(OH)₃)
Description: Demonstrates Einsteinium’s reaction with water, forming hydroxides.
Equation:
Isotopes of Einsteinium
| Isotope | Half-Life | Decay Mode |
|---|---|---|
| Einsteinium-252 | 471.7 days | Alpha decay |
| Einsteinium-253 | 20.47 days | Beta decay |
| Einsteinium-254 | 275.7 days | Alpha decay |
| Einsteinium-255 | 39.8 days | Beta decay |
| Einsteinium-256 | 25.4 minutes | Beta decay |
| Einsteinium-257 | 7.7 days | Alpha decay |
Uses of Einsteinium
- Scientific Research: Einsteinium’s most prominent use is in the field of scientific research, particularly in studying the properties of heavy elements and their reactions.
- Production of Heavier Elements: It serves as a target material in particle accelerators for the synthesis of new elements. Einsteinium isotopes have been used to create even heavier elements, contributing to the expansion of the periodic table.
- Nuclear Physics: Research involving Einsteinium isotopes helps in understanding nuclear fission and fusion processes, providing insights into nuclear physics and the forces binding atomic nuclei.
- Medical Research: Though not widely used due to its scarcity and radioactivity, Einsteinium has potential applications in medical research, particularly in the development of new radiopharmaceuticals for cancer treatment, leveraging its radioactive properties for targeted therapy.
- Material Science: Einsteinium’s unique properties can contribute to material science, especially in studying the behavior of metals under extreme conditions. Its inclusion in alloys or compounds could help understand the effects of radiation on materials, potentially leading to advancements in materials used in space exploration or nuclear reactors
Production of Einsteinium
The production of Einsteinium (Es) involves highly specialized nuclear reactions, primarily occurring within nuclear reactors or during high-energy nuclear tests. As a synthetic element, Einsteinium does not naturally exist on Earth and is produced through a series of neutron capture reactions and subsequent beta decays of heavier elements.
Neutron Bombardment
- Starting Materials: Typically involves bombarding Plutonium (Pu) or Curium (Cm) isotopes with neutrons in a nuclear reactor.
- Process: The target material captures neutrons, increasing its atomic mass until it undergoes beta decay, transforming into heavier elements, including Einsteinium.
Nuclear Tests
- Historical Context: The first detection of Einsteinium was in the fallout from the first hydrogen bomb test in 1952, highlighting a method of production through intense neutron fluxes during nuclear explosions.
Chemical Separation
- Isolation: Following irradiation, Einsteinium is separated from other actinides and fission products through a series of chemical processes, including solvent extraction and ion exchange chromatography.
- Purification: The isolated Einsteinium is further purified to obtain specific isotopes, primarily Einsteinium-253, for research purposes.
Challenges
- Radioactivity: The extreme radioactivity of Einsteinium necessitates stringent safety protocols during production and handling.
- Short Half-lives: Many Einsteinium isotopes have relatively short half-lives, making their study and application challenging.
Applications of Einsteinium
The applications of Einsteinium are predominantly in the field of scientific research, given its scarcity and high radioactivity. Here are the primary uses:
Scientific Research
- Nuclear Physics: Research into the properties of Einsteinium and its isotopes contributes to our understanding of nuclear reactions and the stability of heavy elements.
- Chemistry: Studies on the chemical behavior of Einsteinium help in exploring the chemistry of actinides, enhancing our knowledge of these elements.
Material Science
- Target Material: Einsteinium serves as a target material in particle accelerators for the synthesis of heavier elements, such as those beyond element 100 (Fermium), expanding the periodic table.
Radiation Source
- Potential Uses: While practical applications are limited due to its radioactivity and scarcity, the intense alpha radiation emitted by Einsteinium isotopes could potentially be used in radiation sources for scientific instruments.
This article has systematically explored the intriguing world of Einsteinium, detailing its thermodynamic, material, electromagnetic, and nuclear properties through concise tables. Despite the challenges posed by its radioactivity and limited availability, the study of Einsteinium offers valuable insights into the actinide series and contributes significantly to scientific research, underscoring the element’s unique position in the periodic table and its potential for advancing our understanding of nuclear chemistry.