Chlorine (Cl) – Definition, Preparation, Properties, Uses, Compounds, Reactivity
Chlorine, a highly reactive halogen, holds paramount importance in both organic and inorganic chemistry. This guide delves into chlorine’s multifaceted role, from water purification to the production of everyday household products. The element’s reactivity with hydrogen and other elements forms a myriad of compounds essential for various industrial applications. Discover the expansive world of chlorine, understand its interactions, especially with hydrogen, and explore its crucial role in maintaining public health and safety.

What is Chlorine?
Chlorine is a greenish-yellow gas at room temperature, known for its powerful disinfectant properties and pungent odor. As a member of the halogen group, it is highly reactive, especially with hydrogen, forming hydrochloric acid, a substance fundamental to various industries. Chlorine is widely used in water treatment, bleaching, and the production of plastics and other chemicals. Its ability to eliminate bacteria and purify makes it essential in maintaining clean water supplies worldwide.
Other Reactive Nonmetals
| Hydrogen | Phosphorus |
| Carbon | Sulfur |
| Nitrogen | Selenium |
| Oxygen | Bromine |
| Fluorine | Iodine |
Chlorine Formula
Formula: Cl₂
Composition: Two chlorine atoms.
Bond Type: A single covalent bond connecting the atoms.
Molecular Structure: Diatomic molecule.
Electron Configuration: Seven valence electrons per atom, fourteen in total for Cl₂.
Significance: Essential for water purification, disinfectants, and bleach production.
Role in Chemistry: Widely used in the production of PVC, solvents, and many other organic compounds. Exists as a greenish-yellow gas at room temperature and has a strong, pungent odor.
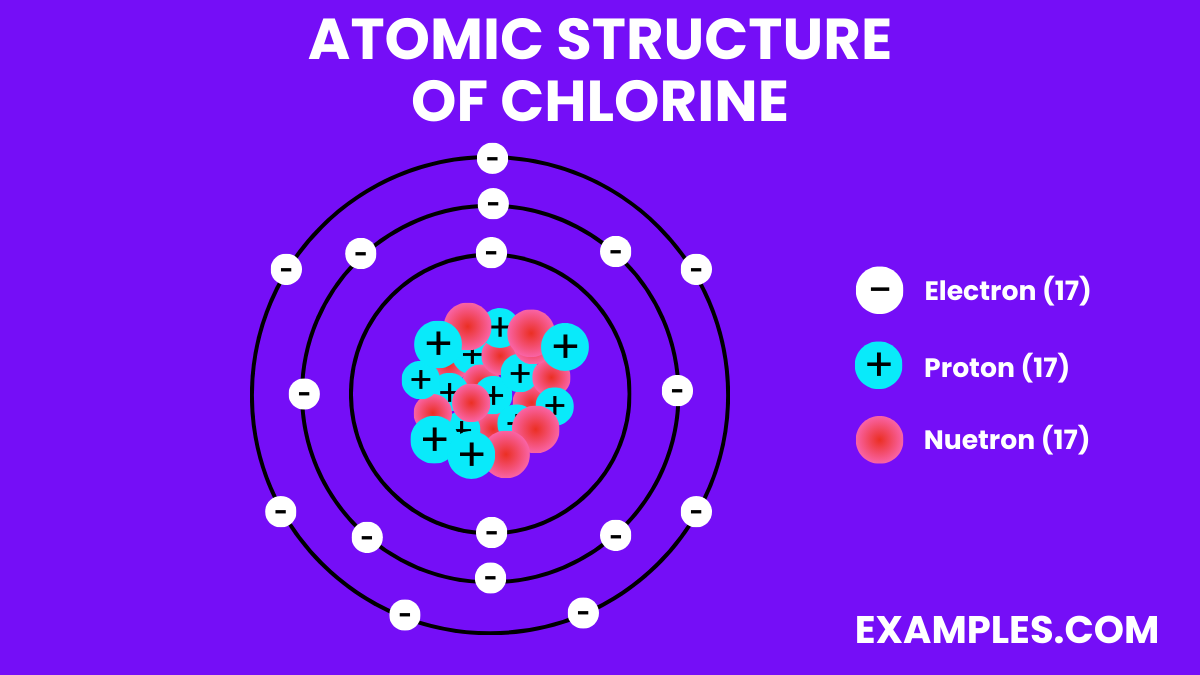
Atomic Structure of Chlorine
Properties of Chlorine
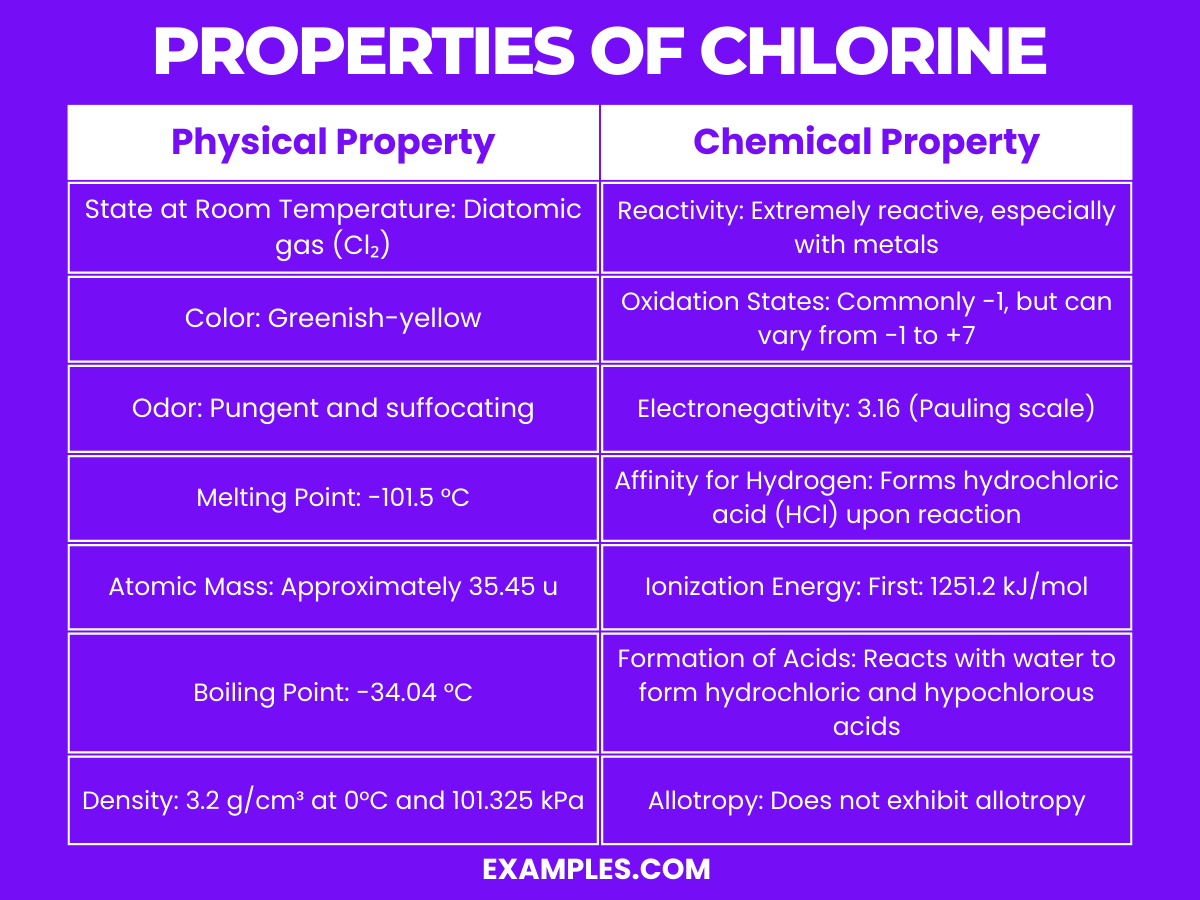
Physical Properties of Chlorine
| Physical Property | Description |
|---|---|
| State at Room Temperature | Diatomic gas (Cl₂) |
| Color | Greenish-yellow |
| Odor | Pungent and suffocating, bleach-like |
| Melting Point | -101.5 °C |
| Boiling Point | -34.04 °C |
| Density | 3.2 g/cm³ at 0°C and 101.325 kPa (denser than air) |
| Solubility | Soluble in water, forming chloric acid and hydrochloric acid |
Chemical Properties of Chlorine
Chlorine is known for its high reactivity and potent oxidizing properties. Here are detailed explanations of its key chemical properties:
- Reactivity: Chlorine is extremely reactive, especially with metals, to form chlorides. For example, it reacts vigorously with sodium to form sodium chloride (common table salt). This high reactivity is primarily due to its electron configuration, where it’s one electron short of a complete octet and readily accepts an electron.
- Oxidation States: Chlorine typically exhibits an oxidation state of -1 in its compounds, such as in hydrochloric acid (HCl) and metal chlorides. However, it can exhibit a range of oxidation states from -1 to +7 due to its ability to accept or donate electrons, which is evident in compounds like chlorine dioxide (ClO₂) and perchlorates (ClO₄⁻).
- Electronegativity: With a value of 3.16 on the Pauling scale, chlorine is one of the most electronegative elements, meaning it has a strong tendency to attract electrons towards itself when forming chemical bonds. This characteristic is fundamental in its ability to form ionic and covalent bonds.
- Affinity for Hydrogen: Chlorine has a high affinity for hydrogen, with which it forms hydrochloric acid (HCl). This reaction is highly exothermic and forms the basis for the industrial production of hydrochloric acid.
- Formation of Acids: When dissolved in water, chlorine can react to form a mixture of hydrochloric acid (HCl) and hypochlorous acid (HOCl). The latter is an important component in bleach and disinfectants due to its ability to kill bacteria and other pathogens.
- Displacement Reactions: Chlorine can displace less reactive halogens such as bromine and iodine from their compounds due to its higher reactivity. This property is used in the extraction and purification of these elements.
Thermodynamic Properties of Chlorine
| Property | Value with Unit |
|---|---|
| Boiling Point | -34.04 °C |
| Melting Point | -101.5 °C |
| Critical Temperature | 144 °C |
| Critical Pressure | 7.99 MPa |
| Heat of Vaporization | 20.41 kJ/mol |
| Heat of Fusion | 6.40 kJ/mol |
| Specific Heat Capacity (at 25°C) | 0.479 J/g·K |
| Thermal Conductivity | 0.0089 W/m·K |
Material Properties of Chlorine
| Property | Value with Unit |
|---|---|
| Density (at 0°C and 1 atm) | 3.214 kg/m³ (Gas) |
| Viscosity (at -34°C) | 0.0135 mPa·s (Gas) |
| Solubility in Water (at 20°C) | 7.3 g/100 mL of water |
| Color | Greenish-yellow |
| Phase at Room Temperature | Gas |
Electromagnetic Properties of Chlorine
| Property | Value with Unit |
|---|---|
| Electrical Conductivity | Non-conductive (Gas) |
| Electronegativity (Pauling scale) | 3.16 |
| Ionization Energy | 12.967 eV |
| Electron Affinity | 3.617 eV |
Nuclear Properties of Chlorine
| Property | Value with Unit |
|---|---|
| Atomic Number | 17 |
| Atomic Mass | 35.453 amu (Average) |
| Isotopes | ^35Cl (75.77%), ^37Cl (24.23%) |
| Nuclear Spin (for ^35Cl) | 3/2 ℏ |
| Nuclear Spin (for ^37Cl) | 3/2 ℏ |
| Neutron Cross Section (for ^35Cl) | 43.6 barns |
| Neutron Cross Section (for ^37Cl) | 0.43 barns |
| Nuclear Magnetic Moment (for ^35Cl) | 0.821 µN |
| Nuclear Magnetic Moment (for ^37Cl) | 0.684 µN |
Chemical Compounds of Chlorine
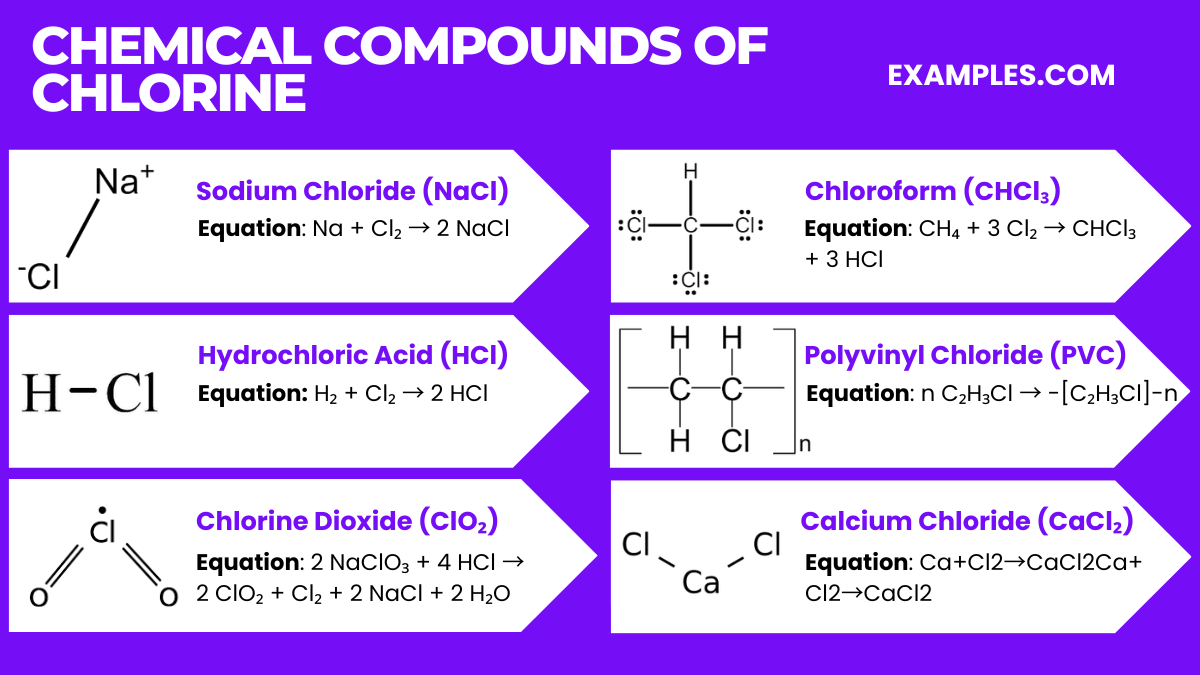
Chlorine, a highly reactive halogen, forms a wide array of significant chemical compounds, each with unique properties and applications. Some of the popular chlorine compounds are:
- Sodium Chloride (NaCl)
- Description: Commonly known as table salt, it is the most abundant chloride mineral.
- Uses: Essential for human health, widely used in food, and in the chemical industry for producing chlorine and caustic soda.
- Equation: Na + Cl₂ → 2 NaCl
- Hydrochloric Acid (HCl)
- Description: A strong acid formed by dissolving hydrogen chloride in water.
- Uses: Utilized in cleaning, pickling of steel, and as a reagent in various chemical reactions.
- Equation: H₂ + Cl₂ → 2 HCl
- Chlorine Dioxide (ClO₂)
- Description: A yellow to reddish-yellow gas, used as a bleaching agent.
- Uses: Applied in paper bleaching, water treatment, and disinfection.
- Equation: 2 NaClO₃ + 4 HCl → 2 ClO₂ + Cl₂ + 2 NaCl + 2 H₂O
- Chloroform (CHCl₃)
- Description: A dense, colorless liquid with a sweet smell, historically used as an anesthetic.
- Uses: Now primarily used in the production of refrigerant and as a solvent in the chemical industry.
- Equation: CH₄ + 3 Cl₂ → CHCl₃ + 3 HCl
- Polyvinyl Chloride (PVC)
- Description: A polymer widely used in construction materials.
- Uses: Used for pipes, cables, containers, and in construction.
- Equation: n C₂H₃Cl → -[C₂H₃Cl]-n (Polymerization reaction)
- Calcium Chloride (CaCl₂)
- Description: A white, crystalline, highly soluble substance known for its moisture-absorbing properties.
- Uses: De-icing roads, dust control, desiccant, and in food as a firming agent.
- Equation: Ca+Cl2→CaCl2Ca+Cl2→CaCl2
Isotopes of Chlorine
Chlorine has two stable isotopes, which are widely used in various applications. Below is a table detailing these isotopes:
| Isotope | Abundance (%) | Atomic Mass | Half-Life | Uses/Significance |
|---|---|---|---|---|
| Chlorine-35 | 75.77 | 34.969 amu | Stable | Used in the study of molecular structures via NMR spectroscopy. Predominant in nature, contributing to the average atomic mass of chlorine. |
| Chlorine-37 | 24.23 | 36.966 amu | Stable | Useful in scientific research and in nuclear reactions. It helps in understanding the chemical and physical properties of chlorine. |
These isotopes play crucial roles in both the natural environment and various scientific fields. Chlorine-35, being more abundant, is often the focus in studies related to chlorine’s behavior and reactions, while Chlorine-37’s unique properties make it valuable in specific research contexts.
Uses of Chlorine
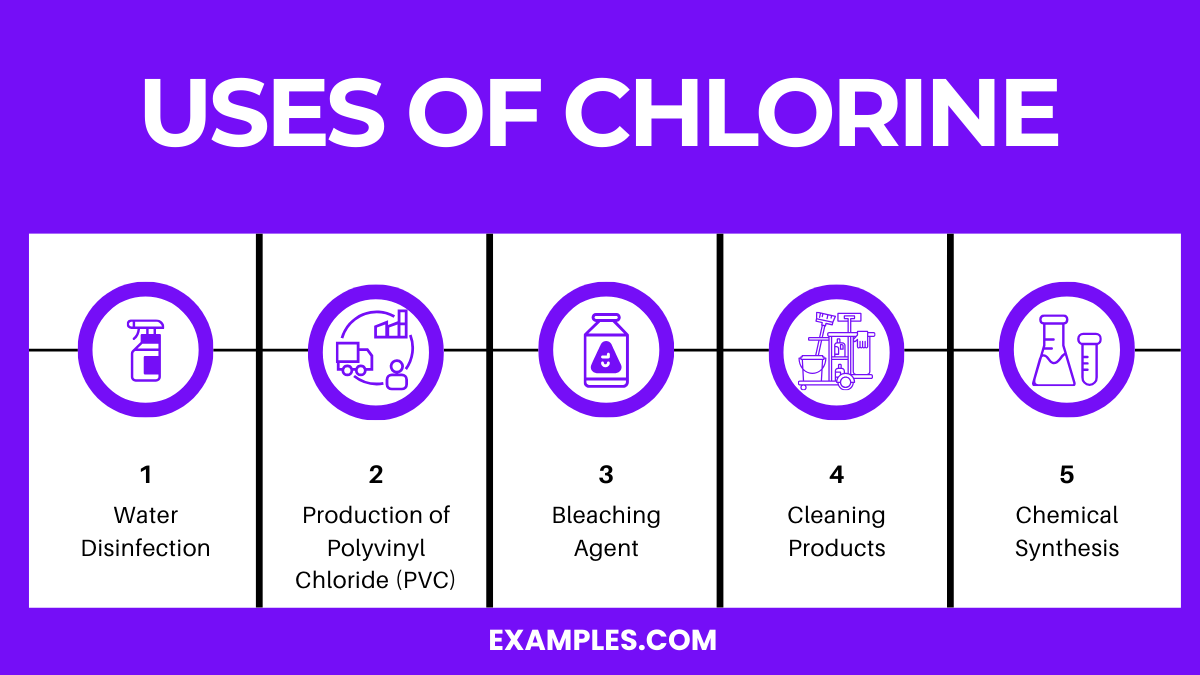
Chlorine is a chemical element with the symbol Cl and atomic number 17. It is a yellow-green gas under standard conditions, where it forms diatomic molecules. This element is an essential reagent in the chemical industry and has various applications, both industrial and household. Here are some of the most prominent uses of chlorine:
1. Water Disinfection: Chlorine is widely used for disinfecting drinking water and swimming pools. It kills bacteria, viruses, and other microbes that can cause disease. Chlorine is added to municipal water systems to make water safe for drinking. Its ability to eliminate pathogens has greatly reduced the incidence of waterborne diseases.
2. Production of Polyvinyl Chloride (PVC): PVC is a versatile plastic used in a wide range of products, including pipes, cables, frames, and medical devices. Chlorine is a key ingredient in producing PVC, making it an integral component in the plastics industry.
3. Bleaching Agent: Chlorine is a powerful bleaching agent used in the paper and textile industries. It is used to bleach wood pulp to produce white paper and to remove stains and colors from textiles.
4. Cleaning Products: Many household cleaning products and disinfectants contain chlorine or its compounds. These products are effective in killing germs, removing stains, and whitening surfaces.
5. Chemical Synthesis: Chlorine is involved in synthesizing various organic and inorganic chemicals, including solvents, dyes, and pharmaceuticals. It is used in producing hydrochloric acid, chloroform, and other important compounds.
6. Public Health and Sanitation: Chlorine compounds are used in sanitizing food processing equipment, controlling odor in waste treatment, and acting as an insecticide and a preservative. Its antimicrobial properties help maintain hygiene standards in various industries.
Commercial Production of Chlorine
Chlorine is predominantly produced through a method known as the chlor-alkali process. This industrial process involves the electrolysis of sodium chloride (salt) solution, producing chlorine gas, sodium hydroxide, and hydrogen gas. Here are the key aspects of the commercial production of chlorine:
1. Chlor-Alkali Process: There are three main methods used in the chlor-alkali process: the mercury cell, the diaphragm cell, and the membrane cell. Each method uses a different technique to separate chlorine from the other products formed during electrolysis.
- Mercury Cell: The mercury cell process produces very pure chlorine but has declined in use due to environmental concerns related to mercury emissions.
- Diaphragm Cell: This method uses a diaphragm to separate the chlorine gas and sodium hydroxide produced during electrolysis. It is more environmentally friendly than the mercury cell process.
- Membrane Cell: The membrane cell process is the most modern and efficient method. It uses a selective membrane to separate the products and is considered the best in terms of energy efficiency and environmental impact.
2. Raw Materials: The primary raw material for chlorine production is sodium chloride (NaCl), commonly known as table salt. This can be sourced from mined rock salt or evaporated from brine wells and seawater.
3. Energy Requirements: The chlor-alkali process is energy-intensive, requiring a significant amount of electricity to induce the necessary electrolysis reaction. As such, advancements in technology aim to make the process more energy-efficient and environmentally friendly.
4. Safety and Handling: Chlorine is a toxic and corrosive gas. During its production, transport, and use, strict safety measures must be observed to prevent exposure and environmental contamination. This includes the use of specialized equipment and protocols for handling and storage.
Understanding the uses and commercial production of chlorine allows for a greater appreciation of this element’s role in modern society, from ensuring clean drinking water to being a fundamental building block in various industries. Its versatile applications and the evolving methods of its production reflect its integral position in the chemical industry.
Health Effects of Sulfur
Sulfur in various forms has diverse implications for health, ranging from beneficial in small amounts to harmful in larger concentrations or particular compounds.
- Nutritional Role: Sulfur is a vital component of certain amino acids (cysteine and methionine) and vitamins (thiamine and biotin). It’s essential for the synthesis of proteins and enzymes, playing a critical role in metabolism, the structure of proteins, and the function of certain vitamins.
- Allergies and Sensitivity: Some individuals may be allergic or sensitive to sulfur or sulfur-containing compounds. Reactions can include skin rashes, hives, or respiratory issues, particularly from sulfites used as preservatives in foods and medications.
- Toxicity: While elemental sulfur is relatively non-toxic, certain sulfur compounds can be hazardous. Hydrogen sulfide (H₂S) is a toxic gas with a characteristic rotten egg smell and can be lethal at high concentrations. Sulfur dioxide (SO₂), a common air pollutant, can irritate the eyes, nose, throat, and lungs, particularly in individuals with asthma or other respiratory conditions.
- Therapeutic Uses: Sulfur is used in the therapeutic treatment of certain skin conditions like acne, dandruff, and psoriasis. Sulfur-containing products can have antimicrobial and keratolytic properties, helping to clear blocked pores and reduce inflammation.
Environmental Effects of Sulfur
Sulfur and its compounds also have significant environmental implications, affecting air quality, climate, and ecosystems.
- Acid Rain: Sulfur dioxide (SO₂) and sulfur trioxide (SO₃) from industrial processes and volcanic activity can combine with water vapor to form sulfuric acid (H₂SO₄), leading to acid rain. Acid rain can have devastating effects on aquatic ecosystems, forests, soil, and man-made structures, corroding metals and eroding buildings.
- Air Quality: Sulfur compounds, particularly sulfur dioxide (SO₂), are harmful air pollutants that affect human health and the environment. They can cause respiratory problems and aggravate cardiovascular diseases. SO₂ is also a precursor to particulate matter, which can reduce visibility (forming smog) and further impact respiratory health.
- Climate Change: While sulfur compounds can have a cooling effect on the atmosphere by reflecting sunlight (sulfate aerosols), they also contribute to global dimming, potentially disrupting ecosystems and climate patterns. The balance of these effects with the warming impact of other greenhouse gases is a subject of ongoing research and environmental concern.
- Ecosystem Impacts: Sulfur deposition from the atmosphere can acidify water bodies and soil, leading to reduced biodiversity and altering nutrient availability. Sulfur emissions can also lead to the formation of fine particulates that can be deposited far from the source, affecting environments and species over a wide area.
Understanding the health and environmental effects of sulfur is crucial for managing its use in industry, agriculture, and consumer products, as well as for regulating emissions to protect human health and the environment. This knowledge helps inform policies and personal decisions related to air quality, industrial processes, and dietary choices.
How Harmful is Chlorine to Humans?
Chlorine gas can be harmful; it irritates the respiratory system, eyes, and skin. Prolonged exposure may lead to severe health issues.
What is Chlorine Used For?
Chlorine is widely used for water purification, disinfection, and in making plastics, solvents, and other chemicals.
Why is Chlorine Banned?
Chlorine is banned in some applications due to its high toxicity and potential for harmful environmental and health effects.
What is Chlorine Made Out Of?
Chlorine is a chemical element with the symbol Cl and atomic number 17, primarily extracted from salt through electrolysis.
Is Chlorine in Bleach?
Yes, chlorine is a primary component in bleach, commonly used as sodium hypochlorite for disinfection and whitening.
Is Chlorine a Metal?
No, chlorine is a non-metal. It is a yellow-green gas at room temperature and belongs to the halogen group in the periodic table.
Chlorine is a critical element with vast applications and implications. This guide has illuminated its uses, handling, and safety tips, aiming to provide comprehensive knowledge for effective and cautious utilization. Always prioritize safety and environmental considerations to harness chlorine’s benefits while minimizing its risks, enhancing both understanding and application.


