Flerovium (FL) – Definition, Preparation, Properties, Uses, Compounds, Reactivity
Dive into the fascinating realm of superheavy elements with our comprehensive guide on Flerovium. This elusive element, known for its position in the periodic table, is shrouded in mystery and scientific intrigue. Flerovium stands at the forefront of nuclear research, offering insights into the synthesis and behavior of superheavy elements. Through detailed examples, we’ll explore Flerovium’s discovery, unique properties, potential uses, and the groundbreaking compounds it forms, enriching your knowledge on this cutting-edge scientific frontier.
What is Flerovium?
Flerovium is a synthetic chemical element with the symbol Fl and atomic number 114. It is part of the group known as the superheavy elements, specifically located in the seventh period and the p-block of the periodic table. Flerovium is named after the Flerov Laboratory of Nuclear Reactions of the Joint Institute for Nuclear Research in Dubna, Russia, where the element was first synthesized in 1998 by a Russian-American collaboration team.As a superheavy element, flerovium does not occur naturally and is produced in a laboratory through the fusion of lighter nuclei, such as bombarding plutonium targets with calcium ions. Its most stable known isotopes have very short half-lives, on the order of a few seconds, making it challenging to study. Due to its short-lived existence, research on flerovium is primarily focused on understanding its nuclear properties and its position in the periodic table, rather than on practical applications.
[/ns_callout]
Other Unknown properties
| Meitnerium | Darmstadtium | Roentgenium |
| Copernicium | Nihonium | Moscovium |
| Livermorium | Tennessine | Oganesson |
Flerovium Formula
Formula: Fl
Composition: Consists of a single flerovium atom.
Bond Type: In its elemental form, flerovium does not have bonds as it is a pure element. However, flerovium can form covalent or ionic bonds when reacting with other elements, although its chemical reactivity is not well-studied due to its rarity and short half-life.
Molecular Structure: As a pure element, flerovium does not form a molecular structure in the same sense as compounds. It is predicted to be a heavy, volatile metal with a possible close-packed crystalline structure, though its exact physical and chemical properties remain largely speculative.
Electron Sharing: In compounds, flerovium is expected to share electrons covalently or transfer electrons ionically, depending on the nature of the other element(s) it is bonding with. Its chemistry is predicted based on its position in the periodic table, but experimental data is limited.
Significance: Flerovium is notable for being a superheavy element synthesized in particle accelerators. Its study helps scientists understand the properties of elements at the edge of the periodic table and the stability of superheavy nuclei.
Role in Chemistry: Flerovium’s role in chemistry is primarily in the field of research, particularly in studies aimed at exploring the limits of the periodic table and the synthesis of new elements. Its potential applications outside of scientific research are currently unknown due to its extreme rarity and instability.
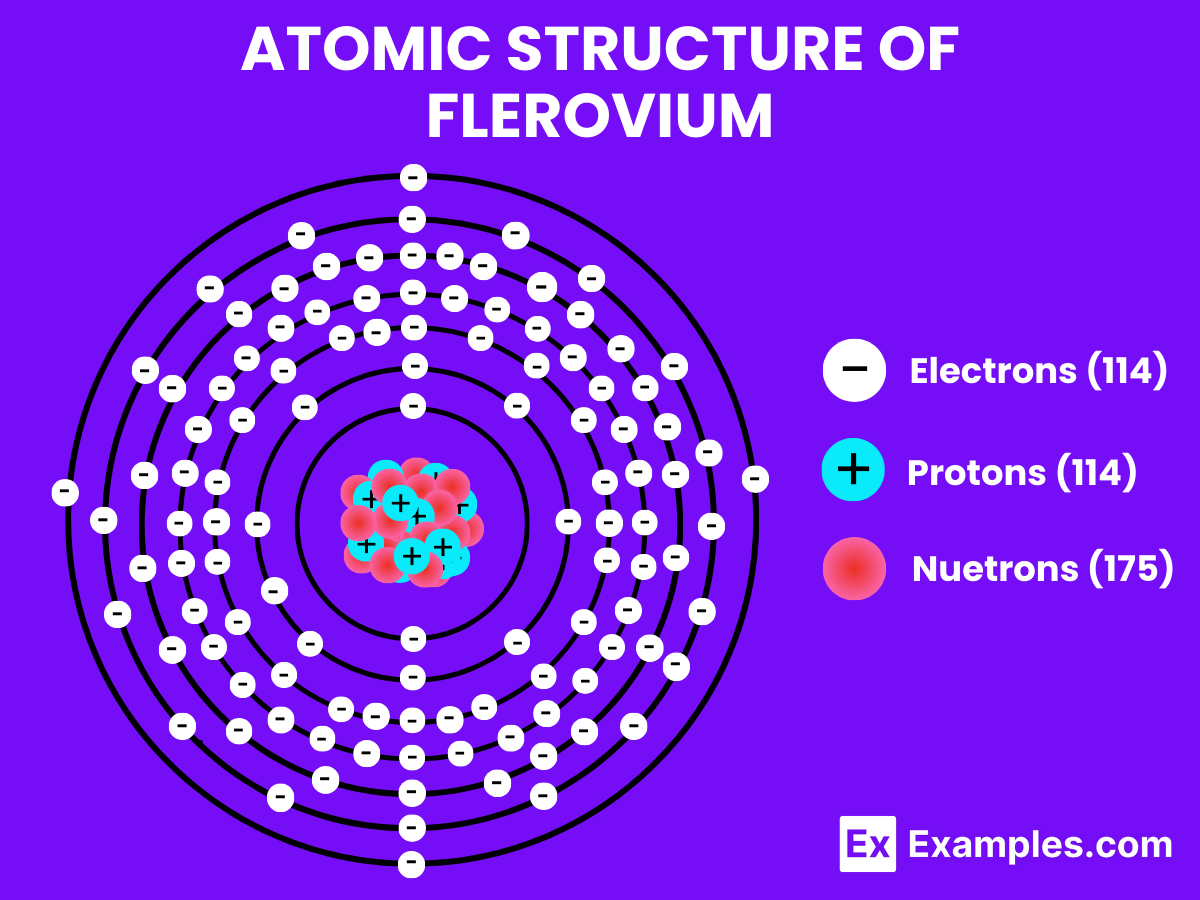
Atomic Structure of Flerovium
Flerovium (Fl) is a superheavy synthetic element with the atomic number 114 on the periodic table. Named after the Flerov Laboratory of Nuclear Reactions where it was first synthesized, Flerovium’s atomic structure exhibits some fascinating characteristics that distinguish it from lighter elements. Understanding its atomic structure requires delving into its electronic configuration, nuclear composition, and predicted chemical properties, which are all influenced by its position in the periodic table and the effects of relativistic mechanics on its electrons.
Electronic Configuration
Flerovium’s electronic configuration is theorized to be [Rn] 5f¹⁴ 6d¹⁰ 7s² 7p² placing it in the p-block of the periodic table. This configuration suggests that Flerovium may exhibit some properties similar to lead (Pb), which is directly above it in Group 14. However, due to relativistic effects, the actual chemistry of Flerovium may deviate significantly from these predictions.
Nuclear Composition
The nucleus of Flerovium atoms contains 114 protons, which defines its position as the 114th element in the periodic table. The number of neutrons in Flerovium can vary, leading to the creation of different isotopes. The most stable isotope known to date is Flerovium-289, with 175 neutrons. These isotopes are characterized by extremely short half-lives, with Flerovium-289 having a half-life of about 2.6 seconds, indicating the inherent instability of superheavy elements.
Relativistic Effects
The atomic structure of Flerovium is significantly influenced by relativistic effects. As electrons in heavy elements move at speeds approaching a significant fraction of the speed of light, their mass increases, which in turn affects their interaction with the nucleus and with each other. These effects are particularly pronounced in superheavy elements like Flerovium, leading to changes in electron orbital shapes, energy levels, and chemical reactivity.
Predicted Properties
Based on its electronic configuration and the anticipated impact of relativistic effects, Flerovium is expected to have some unique properties:
- It might display a relatively high degree of inertness, similar to the noble gases, due to the stabilization of its 7p electrons.
- Its metallic properties are predicted to be quite distinct from those of lighter group 14 elements, possibly exhibiting a lower melting point and a higher density.
- The chemical reactivity of Flerovium is largely unknown and a subject of ongoing research. Predictions suggest it might form compounds with oxygen, hydrogen, and halogens, but the stability and characteristics of such compounds remain speculative.
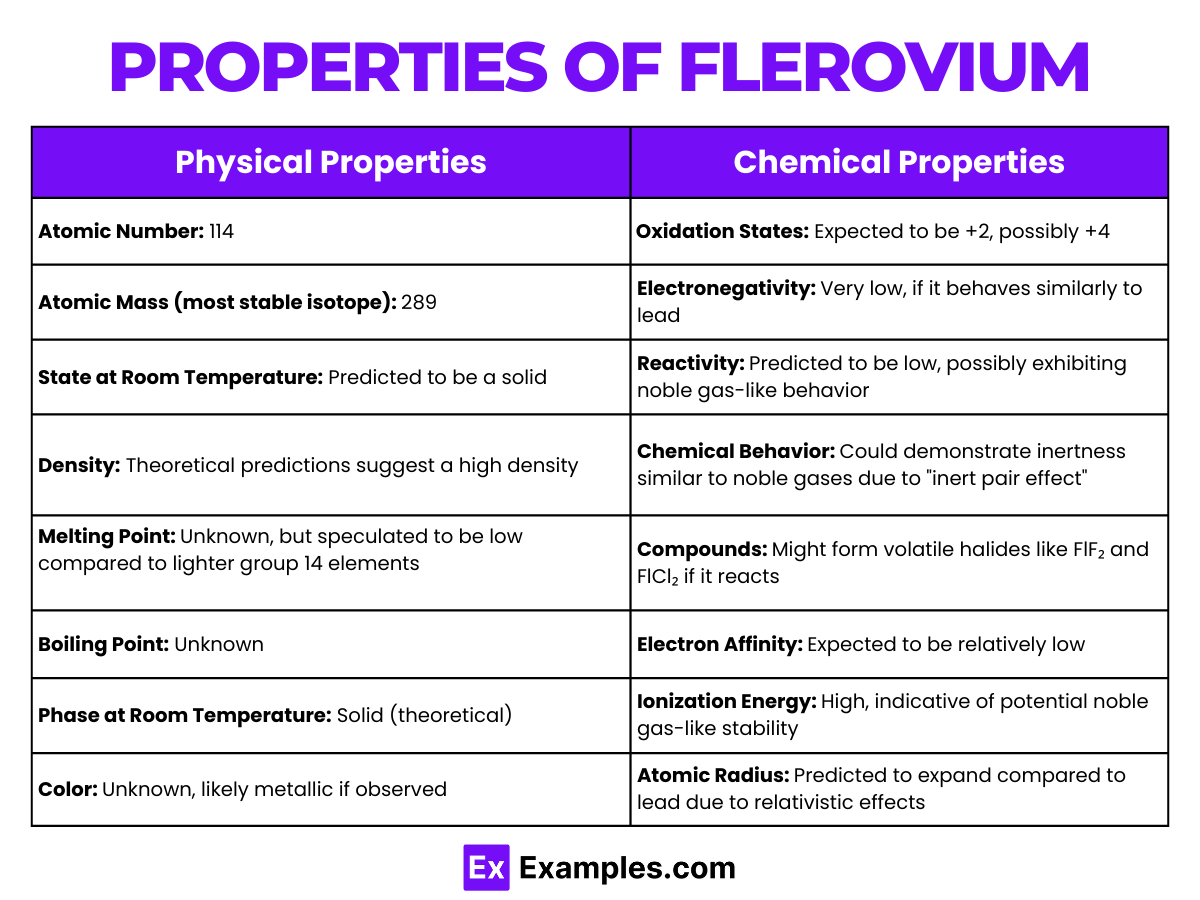
Properties of Flerovium
Physical Properties of Flerovium
| Property | Value |
|---|---|
| Atomic Number | 114 |
| Relative Atomic Mass | (289) |
| Density | Predicted to be high, potentially similar to or greater than lead |
| Melting Point | Unknown, but predicted to be low for a metal |
| Boiling Point | Unknown |
| State at Room Temperature | Presumed to be solid |
| Color | Unknown, potentially metallic |
| Half-life of Most Stable Isotope (Fl-289) | ~2.6 seconds |
Chemical Properties of Flerovium
Flerovium (Fl), with atomic number 114, resides in the p-block of the periodic table. Its chemical properties are inferred from theoretical predictions and relativistic calculations due to its extremely short half-life and the complexities associated with its production.
Electronic Configuration and Reactivity
- Electronic Configuration:[Rn] 5f¹⁴ 6d¹⁰ 7s² 7p².This suggests potential chemical similarities with other group 14 elements, modified by relativistic effects.
- Predicted Reactivity: The stabilization of 7p orbitals due to relativistic effects might make Flerovium less reactive than expected for its group.
Oxidation States
- Expected Oxidation States: +2 and +4, with the latter possibly more stable in Flerovium, deviating from lighter group 14 trends.
Predicted Compounds
- Hydrides: Flerovium hydride (FlH₂) could mimic PbH₂, indicating covalent bonding capabilities.
- Halides: Flerovium’s halides, such as Flerovium tetrafluoride (FlF₄) and Flerovium dichloride (FlCl₂), would showcase its versatility in oxidation states.
- Fl + 2Cl₂ → FlCl₄ (Predicted reaction for Flerovium tetrachloride, assuming it follows lead’s chemical behavior.)
- Oxides: Flerovium dioxide (FlO₂) aligns with a +4 oxidation state, analogous to PbO₂.
- Fl + O₂ → FlO₂ (Hypothetical reaction, suggesting reactivity with oxygen.)
- Organometallic Compounds: The possibility of Flerovium forming organometallic compounds, like Flerovium hexacarbonyl (Fl(CO)₆), is speculative.
Thermodynamic Properties of Flerovium
| Property | Value |
|---|---|
| Melting Point | Predicted to be low, possibly around the melting point of Lead or slightly higher |
| Boiling Point | Theoretical; expected to be similar to or slightly higher than that of Lead due to relativistic effects |
| Density | Predicted to be significantly high, potentially higher than Lead due to relativistic effects |
Material Properties of Flerovium
| Property | Value |
|---|---|
| State at STP | Predicted to possibly be a solid at room temperature |
| Crystal Structure | Unknown; theoretical predictions suggest a structure that could be influenced by strong relativistic effects |
| Thermal Conductivity | Expected to be low, in line with heavy metals and influenced by its electronic structure |
Electromagnetic Properties of Flerovium
| Property | Value |
|---|---|
| Electrical Conductivity | Expected to be relatively poor, influenced by its electronic configuration |
| Electronegativity | Predicted based on its position; however, exact values are unknown due to lack of experimental data |
Nuclear Properties of Flerovium
| Property | Value |
|---|---|
| Half-life of Most Stable Isotope (Fl-289) | About 2.6 seconds, showcasing the element’s instability |
| Decay Modes | Predominantly alpha decay; some isotopes may undergo spontaneous fission |
| Atomic Mass | Isotopes range observed: 284-289 amu, with a focus on nuclear stability and decay patterns |
Preparation of Flerovium
The preparation of Flerovium, a superheavy synthetic element with the atomic number 114, involves highly sophisticated nuclear reactions conducted in particle accelerators. Flerovium does not occur naturally and can only be created artificially through the collision of lighter nuclei. The process to prepare Flerovium primarily involves bombarding a target material with accelerated ions of another element.
Key Steps in the Preparation:
- Selection of Target and Projectile: The preparation process begins with the selection of suitable target and projectile materials. For Flerovium, Plutonium (Pu) or Californium (Cf) targets have been used, with Calcium (Ca) ions acting as the projectiles.
- Acceleration: The Calcium ions are accelerated to high velocities using a particle accelerator. This step is crucial for ensuring that the ions have sufficient kinetic energy to overcome the electrostatic repulsion of the target nucleus upon collision.
- Collision and Fusion: The high-speed Calcium ions collide with the target atoms. When these ions successfully fuse with the target nuclei, new, heavier nuclei are formed.
- Detection and Identification: The atoms of Flerovium produced are highly unstable and undergo rapid radioactive decay. Detecting and identifying Flerovium involves tracking these decay events, which are characterized by the emission of alpha particles (helium nuclei). The decay patterns help scientists confirm the creation of Flerovium and study its properties.
Chemical Compounds of Flerovium
- Flerovium Dioxide (FlO₂)
- A stable oxide expected to form when Flerovium reacts with oxygen.
- Equation: Fl+O₂→ FlO₂
- Flerovium Tetrafluoride (FlF₄)
- A compound resulting from the reaction of Flerovium with fluorine, highlighting its ability to form halides.
- Equation: Fl+4F₂→ FlF₄
- Flerovium Heptoxide (Fl₂O₇)
- A volatile, oxidizing agent predicted at high oxidation states of Flerovium.
- Equation: 2FlF₄+2H⁺→ Fl₂O₇+4HF
- Flerovium Sulfide (Fl₂S)
- A compound indicating Flerovium’s potential to form sulfides.
- Equation: 2Fl+2S→ Fl₂S
- Flerovium Hexacarbonyl (Fl(CO)₆)
- Demonstrates Flerovium’s capacity to engage in complex organometallic chemistry.
- Equation: Fl+6CO→ Fl(CO)₆
- Flerovium Chloride (FlCl₃)
- A compound showcasing Flerovium’s reactivity with halogens, specifically chlorine, to form trihalides.
- Equation: Fl+3Cl₂→ FlCl₃
Isotopes of Flerovium
| Isotope | Half-Life | Decay Mode |
|---|---|---|
| Flerovium-284 | 2.5 seconds | Alpha decay to Copernicium-280 |
| Flerovium-285 | 0.89 milliseconds | Alpha decay to Copernicium-281 |
| Flerovium-286 | 0.13 seconds | Alpha decay to Copernicium-282 |
| Flerovium-287 | 0.48 seconds | Alpha decay to Copernicium-283 |
| Flerovium-288 | 0.8 seconds | Alpha decay to Copernicium-284 |
| Flerovium-289 | 1.9 seconds | Alpha decay to Copernicium-285 |
| Flerovium-290 | 19 seconds | Alpha decay to Copernicium-286 |
| Flerovium-291 | Unknown | Predicted alpha decay to Copernicium-287 |
Uses of Flerovium
Currently, Flerovium (Fl) does not have any practical uses outside of scientific research due to its extremely short half-life and the difficulty in producing it in significant quantities. Flerovium is a superheavy synthetic element that was first discovered in 1998, and its properties are not fully understood due to its instability and rarity. Here are the primary areas where Flerovium is of interest:
- Scientific Research: The main use of Flerovium is in scientific experiments designed to understand more about the properties of superheavy elements. Research on Flerovium helps scientists explore the limits of the periodic table, nuclear physics, and the theoretical “island of stability,” where it’s believed that superheavy elements might have longer half-lives.
- Nuclear Chemistry: Studies involving Flerovium contribute to the field of nuclear chemistry, particularly in understanding the synthesis of superheavy elements and their decay patterns. This can provide insights into the forces that hold atomic nuclei together and might lead to discoveries of new elements.
- Astrophysics and Cosmology: While not directly used in astrophysics, the study of superheavy elements like Flerovium can offer insights into processes that might occur in extreme astrophysical environments, contributing to our understanding of the universe’s chemical composition.
- Radiation Shielding Materials: In the distant future, materials incorporating superheavy elements like Flerovium might be explored for their potential in radiation shielding, especially if they exhibit unique properties that make them more effective at absorbing or deflecting high-energy particles.
- Technological Innovation: Although currently speculative, the study of Flerovium and other superheavy elements could eventually lead to technological innovations, particularly in materials science. Understanding the properties of these elements may unlock new possibilities in the development of materials with unique electromagnetic, optical, or catalytic properties, potentially useful in electronics, energy, and nanotechnology sectors in the far future.
Production of Flerovium
Flerovium, a superheavy synthetic element, is produced in highly specialized nuclear research facilities rather than being a byproduct of nuclear reactors like Technetium. Here’s an overview of its production process:
Direct Fusion with Calcium Ions
- Source: Flerovium (Fl) is produced by bombarding target atoms like Plutonium (Pu) or Californium (Cf) with Calcium (Ca) ions in particle accelerators.
- Process: When the target atoms are bombarded with high-speed Calcium ions, the nuclei can fuse, resulting in the creation of Flerovium.
- Extraction: Unlike Technetium, Flerovium cannot be “extracted” through chemical processes due to its rapid decay. Instead, its presence is confirmed by detecting the decay products characteristic of Flerovium isotopes.
Cold Fusion Reactions
- Source: An alternative method for producing Flerovium involves using lighter targets and projectiles in cold fusion reactions, though this method is less commonly used for Flerovium than for other superheavy elements.
- Process: In these reactions, a beam of lighter ions is directed at a heavy target material, aiming to achieve fusion at relatively low energies compared to hot fusion methods.
- Extraction: The identification of Flerovium follows its synthesis, relying on the detection of specific decay chains that confirm the production of Flerovium isotopes.
Applications of Flerovium
Currently, Flerovium (element 114) does not have practical applications due to its extremely short half-life and the challenges associated with producing it in sufficient quantities for practical use. As one of the superheavy elements, its existence is primarily of interest to scientific research aimed at understanding the properties of elements at the extreme end of the periodic table. Here are the potential areas of interest related to Flerovium:
- Scientific Research: The synthesis and study of Flerovium are significant for advancing our knowledge of nuclear physics and chemistry. Research into its properties can help scientists explore the theoretical “island of stability” where certain superheavy elements might have longer half-lives.
- Periodic Table and Elemental Behavior: The discovery and investigation of Flerovium contribute to the broader understanding of the periodic table’s structure and the behaviors of elements within it, especially those in the superheavy category.
Flerovium represents a frontier in the exploration of superheavy elements, offering insights into the limits of the periodic table and nuclear stability. While practical applications are yet to be realized due to its short half-life and production challenges, ongoing research on Flerovium could pave the way for future scientific breakthroughs and a deeper understanding of elemental behavior.