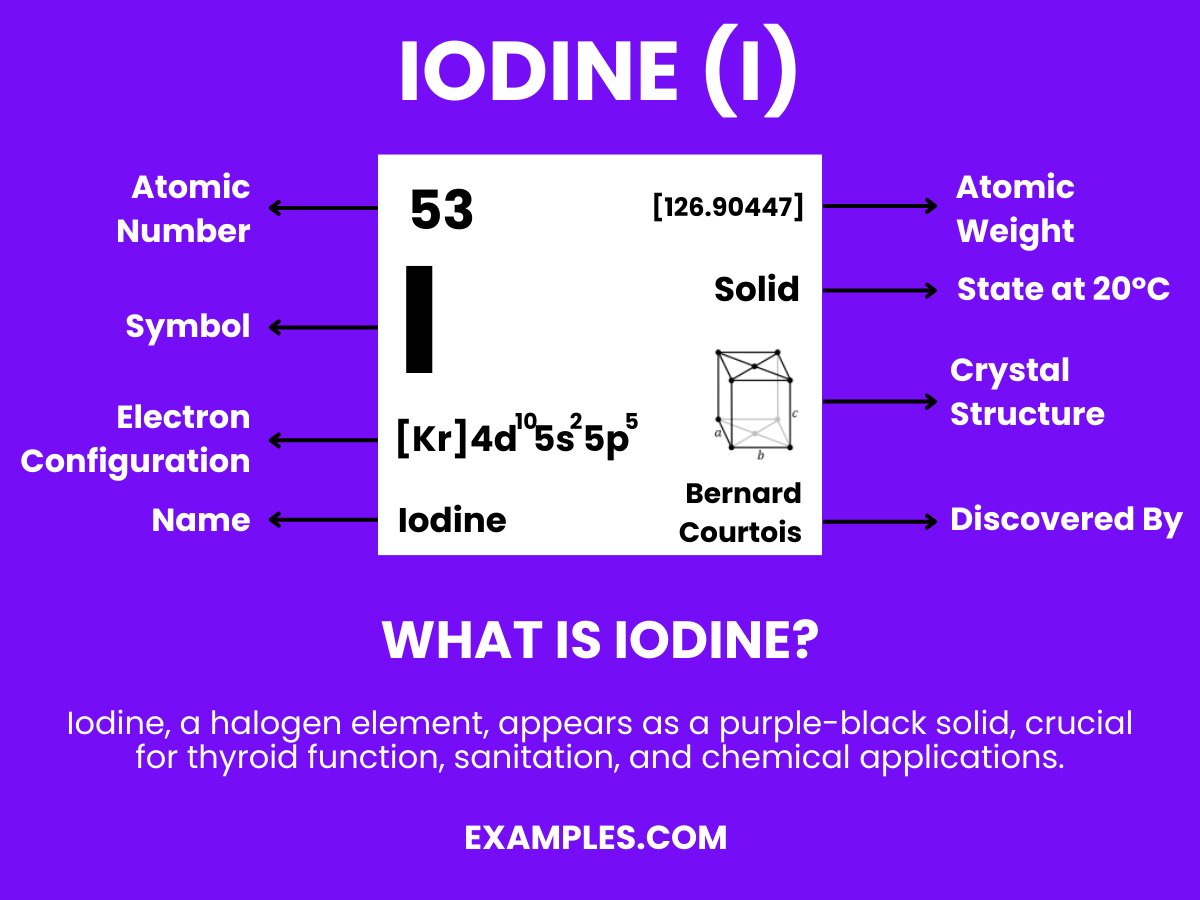
Iodine (I) – Definition, Preparation, Properties, Uses, Compounds, Reactivity
Discover the intriguing world of iodine, an essential element in both nature and the classroom. This guide dives deep into its properties, uses, and significance, providing you with well-rounded content to enrich your lessons. As you teach about the role of iodine in human health and the environment, remember it’s as fundamental as hydrogen in many chemical reactions. Engage your students with captivating examples and clear explanations, making chemistry relatable and exciting.
What is Iodine?
Iodine is a chemical element with the symbol “I” and atomic number 53. It’s a non-metallic, dark-gray or purple-black, lustrous solid at room temperature. Iodine is essential in biology, notably in the synthesis of thyroid hormones in the human body, which regulate metabolism and other important functions. It’s also used widely in medicine, nutrition, and various industrial applications. Teachers can explain iodine’s significance by highlighting its critical role in maintaining human health and its diverse uses in our daily lives.
Other Reactive Nonmetals
| Hydrogen | Phosphorus |
| Carbon | Sulfur |
| Nitrogen | Chlorine |
| Oxygen | Selenium |
| Fluorine | Bromine |
Iodine Formula
Formula: I₂
Composition: Two iodine atoms.
Bond Type: A single covalent bond connecting the atoms.
Molecular Structure: Diatomic molecule.
Electron Configuration: Seven valence electrons per atom, fourteen in total for I₂.
Significance: Essential nutrient for thyroid function and widely used as a disinfectant and antiseptic.
Role in Chemistry: Used in the synthesis of certain organic compounds and in iodometry. Appears as a solid crystal at room temperature with a distinctive purple vapor.
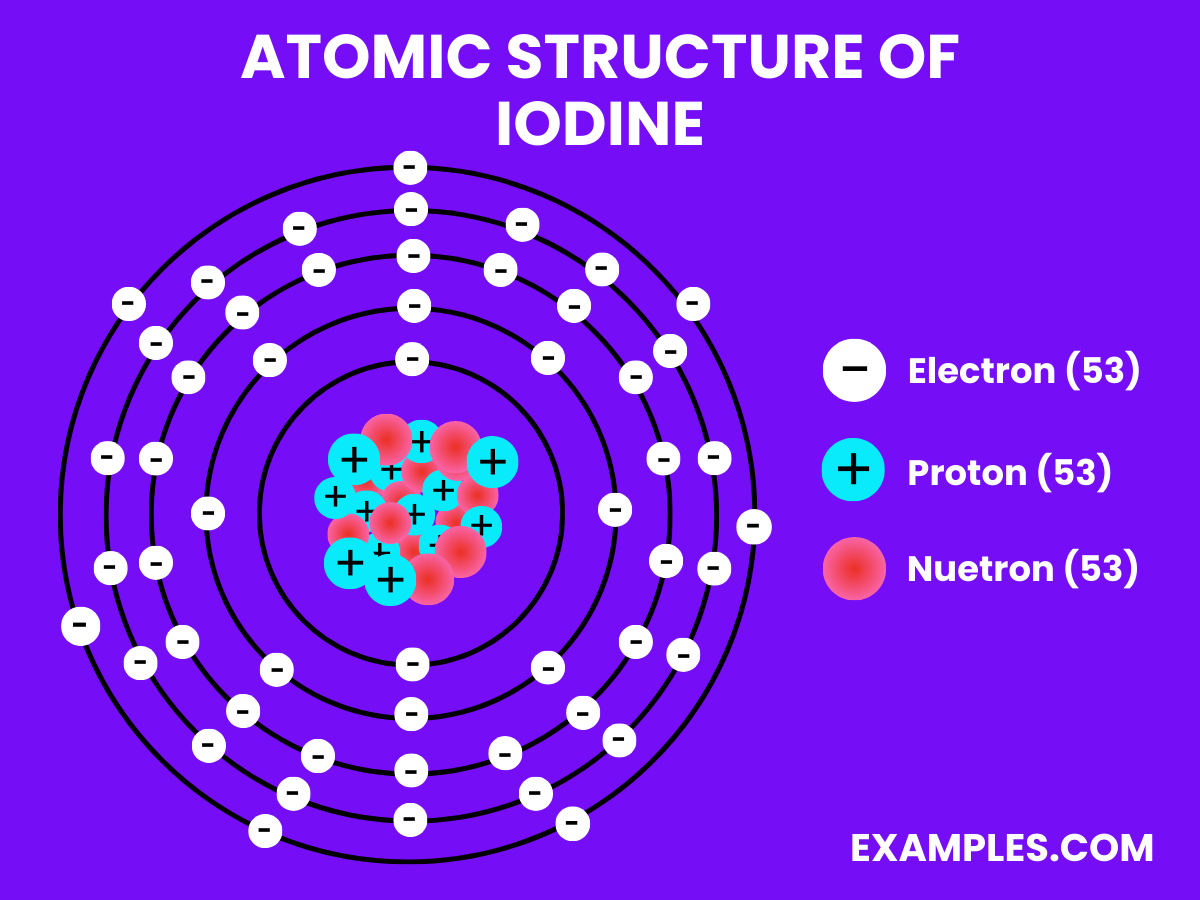
Atomic Structure of Iodine
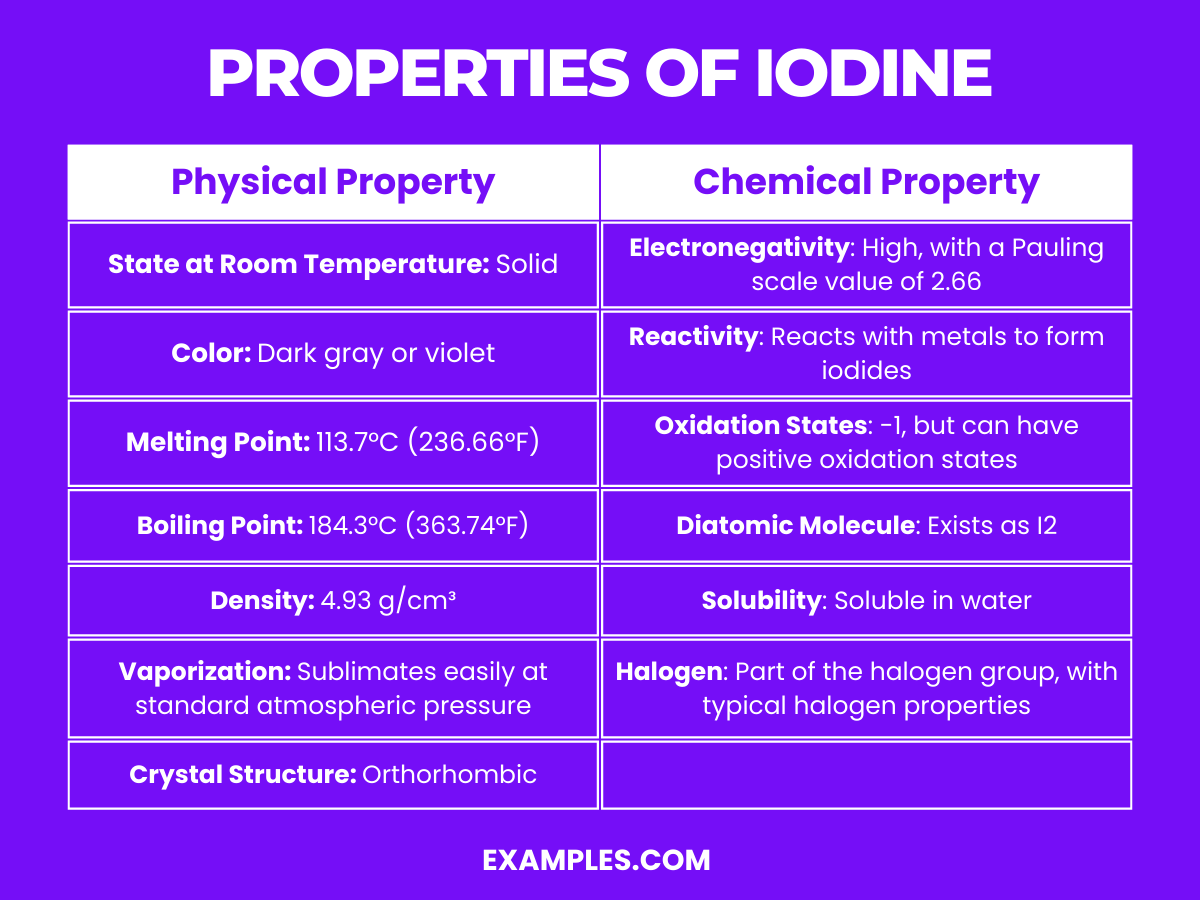
Properties of Iodine
Physical Properties of Iodine
| Physical Property | Description |
|---|---|
| State at Room Temperature | Solid, usually appears as shiny, dark gray or violet crystals. |
| Color | Dark gray or violet, depending on the form and purity. |
| Melting Point | 113.7°C (236.66°F), where solid iodine begins to transition into a liquid form. |
| Boiling Point | 184.3°C (363.74°F), the temperature at which it converts from a liquid to a gaseous state. |
| Density | 4.93 g/cm³ at 20°C, relatively high density for a non-metallic element. |
| Vaporization | Sublimates easily, meaning it transitions directly from a solid to a gaseous state at standard atmospheric pressure. |
| Crystal Structure | Orthorhombic, forming lattice structures in its solid form. |
| Appearance | Iodine crystals are shiny and lustrous with a metallic sheen, signifying its crystalline solid state. |
Chemical Properties of Iodine
- Electronegativity: Iodine has a relatively high electronegativity with a Pauling value of 2.66. This makes it a good oxidizing agent and reactive towards many elements and compounds.
- Reactivity: Iodine reacts with many metals to form iodides M+I₂→MI, which are generally ionic salts. For example, 2Na+I₂→2NaI.
- Oxidation States: Iodine commonly exhibits a -1 oxidation state, particularly in iodides. However, it can also form compounds with positive oxidation states such as I₂O7, and various organic iodine compounds.
- Diatomic Molecule: In standard conditions, iodine exists as a diatomic molecule I₂. This molecular form is significant as it influences its physical and chemical behavior.
- Solubility: Iodine is moderately soluble in water, forming a brown solution. It is much more soluble in organic solvents like chloroform, carbon tetrachloride, or carbon disulfide, forming a purple solution due to the formation of charge-transfer complexes.
- Halogen Characteristics: As a member of the halogen family, iodine shares characteristics like forming diatomic molecules and exhibiting various oxidation states. It’s less reactive than chlorine and bromine but more reactive than astatine.
- Role in Organic Chemistry: Iodine is used in various organic synthesis reactions. For example, in the addition of iodine across the double bonds of alkenes or in iodination reactions where iodine is introduced into organic molecules.
- Biological Role: Iodine is vital for life; it’s a key component of thyroid hormones, such as thyroxine (T₄) and triiodothyronine (T3). These hormones are crucial for regulating metabolism, growth, and development.
By understanding both the physical and chemical properties of iodine, scientists and manufacturers can use it effectively in various applications, from medical treatments and dietary supplements to chemical synthesis and industrial processes.
Thermodynamic Properties of Iodine
| Property | Description / Value |
|---|---|
| Melting Point | 113.7°C (236.66°F) |
| Boiling Point | 184.3°C (363.74°F) |
| Thermal Conductivity | 0.449 W/(m·K) at 25°C |
| Specific Heat | 0.214 J/(g·K) at 25°C |
| Heat of Vaporization | 41.57 kJ/mol at boiling point |
| Heat of Fusion | 15.52 kJ/mol at melting point |
Material Properties of Iodine
| Property | Description / Value |
|---|---|
| Phase at STP | Solid |
| Density | 4.933 g/cm³ at 20°C |
| Color | Shiny, metallic-gray solid or violet vapor |
| Solubility in Water | 0.03 g/100 mL at 20°C |
| Crystal Structure | Orthorhombic |
Electromagnetic Properties of Iodine
| Property | Description / Value |
|---|---|
| Magnetic Susceptibility | Diamagnetic |
| Electrical Conductivity | Poor conductor of electricity |
Nuclear Properties of Iodine
| Property | Description / Value |
|---|---|
| Atomic Number | 53 |
| Atomic Mass | 126.90447 u |
| Neutron Cross Section | 6.15 barns (for ^127I) |
| Isotopes | Predominantly ^127I; Numerous radioactive isotopes, notably ^131I with a half-life of 8 days |
| Radioactivity | ^131I is commonly used in medical diagnostics and treatment, especially in thyroid conditions |
Chemical Compounds of Iodine
Iodine is a halogen element with a rich chemistry, primarily known for its roles in iodized salt and thyroid health. Here are some of the key compounds of iodine:
- Hydrogen Iodide (HI): This compound is a diatomic molecule and forms hydroiodic acid when dissolved in water. It’s used in organic chemistry as a reducing agent.
Equation: H₂+I₂→2HI - Iodine Monochloride (ICl): It’s an interhalogen compound where iodine is bonded to chlorine. It’s used in some analytical chemistry procedures.
Equation: I₂+Cl₂→2ICl - Iodine Pentafluoride (IF5): This is a fluorinating agent used to introduce iodine in certain organic compounds.
Equation: I₂+5F₂→2IF5 - Potassium Iodate (KIO3): Often used in iodized salt, it serves as a source of dietary iodine.
Equation: 3I₂+6KOH→5KI+KIO3+3H₂O - Sodium Iodide (NaI): It is used in photographic films and as a nutritional supplement.
Equation: Na+I→NaI
Isotopes of Iodine
Iodine has several isotopes, but here are the most noteworthy, particularly Iodine-127 (the only stable isotope) and Iodine-131 (known for its medical applications):
| Isotope | Mass Number | Half-Life | Application or Note |
|---|---|---|---|
| I-123 | 123 | 13.22 hours | Used in nuclear medicine imaging, particularly for thyroid scans. |
| I-124 | 124 | 4.18 days | Useful in positron emission tomography (PET) imaging. |
| I-125 | 125 | 59.4 days | Widely used in medical diagnostics and treatment, especially in brachytherapy. |
| I-127 | 127 | Stable | The only stable isotope, natural iodine is composed of I-127. |
| I-129 | 129 | 15.7 million years | Long-lived radioactive isotope, used in geological dating. |
| I-131 | 131 | 8.02 days | Widely known for its use in treating thyroid cancer and hyperthyroidism. |
Uses of Iodine
Iodine, a chemical element with the symbol I and atomic number 53, plays a vital role in various applications due to its unique properties. Here are some of its primary uses:
Medical Applications
- Disinfectant: Iodine is widely used as a disinfectant in medical settings due to its effectiveness in killing bacteria, viruses, and fungi. It’s often applied to wounds or used for sanitizing surgical equipment.
- Dietary Supplement: As an essential nutrient, iodine is crucial for thyroid function. It’s used in iodized salt to prevent iodine deficiency, which can lead to goiter and other health issues.
- Radiocontrast Agent: In radiology, iodine compounds are used as contrast agents to improve the visibility of internal bodily structures in imaging techniques like X-rays and CT scans.
Industrial Applications
- Catalyst: Iodine is used as a catalyst in the production of certain polymers and pharmaceuticals.
- Inks and Dyes: Due to its vibrant color, it’s utilized in inks, dyes, and colorants for printing and photography.
Scientific Research
- Laboratory Reagent: In chemistry, iodine is a common reagent used in various analytical techniques and synthetic processes.
Photography
- In the form of silver iodide, iodine is used in photography for the preparation of photographic films and papers.
Agriculture
- Iodine is utilized in agriculture for the treatment of iodine deficiency in plants, enhancing plant growth, and quality of the produce. It is also used as a fungicide and bactericide for fruits and vegetables.
Commercial Production of Iodine
The commercial production of iodine involves several methods, primarily sourced from underground brines associated with natural gas and oil deposits. Here’s how iodine is typically produced:
Extraction from Brine
- Underground Brines: The primary source of iodine is brine water, typically found in oil fields. The brine contains iodine in the form of iodide ions.
- Oxidation Process: The iodide is extracted by adding an oxidizing agent to convert it into iodine.
- Precipitation: The iodine is then precipitated out of the solution, often as iodine crystals.
Processing from Caliche Ore
- Caliche Ore: In certain regions, especially Chile, iodine is extracted from caliche ore. The ore is processed to release iodate ions, which are then reduced to iodine.
Recycling
- Waste Streams: Iodine is also reclaimed from waste streams of certain industries, like the production of acetic acid.
Purification
- Refining: The crude iodine obtained from extraction is further purified through sublimation or other refining processes to get the desired purity for various applications.
In summary, iodine’s multifaceted uses in medical, industrial, and scientific domains highlight its importance. Its commercial production, primarily from brine sources, underscores a sophisticated process of extraction and purification to meet global demands.
Health Effects of Iodine
Iodine is a vital micronutrient required for normal thyroid function, growth, and development. It’s found naturally in various foods and is also added to salt in many countries.
Positive Health Effects
- Thyroid Regulation: Iodine is crucial for the synthesis of thyroid hormones, which regulate metabolism, energy production, and neurological development.
- Cognitive Function: Adequate iodine during pregnancy and infancy leads to better cognitive outcomes in children.
- Growth and Development: It ensures proper bone and brain development during pregnancy and infancy.
Negative Health Effects
- Iodine Deficiency Disorders: Lack of iodine can lead to goiter, cretinism, and varied cognitive impairments.
- Excessive Intake: Excessive iodine can cause thyroiditis, goiter, or hypothyroidism, especially in individuals with pre-existing thyroid disease.
Environmental Effects of Iodine
While iodine is essential for life, its distribution and availability in the environment can significantly impact ecosystems and human health.
Positive Environmental Effects
- Nutrient Cycling: Iodine plays a role in the biogeochemical cycles of oceans, aiding in the production and decay of biological matter.
- Air Quality: Iodine from the ocean contributes to atmospheric chemistry, including the formation of clouds which can influence climate patterns.
Negative Environmental Effects
- Bioaccumulation: Excessive iodine can accumulate in the food chain, particularly affecting marine life and those who consume seafood.
- Radioactive Iodine: Iodine has a radioactive isotope, I-131, which can be released from nuclear accidents or medical treatments, posing a significant health risk and environmental contamination.
What is Iodine Used For?
Iodine is essential for thyroid hormone production, regulating metabolism, and supporting cognitive functions.
What Foods are Rich in Iodine?
Seafood, dairy products, grains, and iodized salt are rich in iodine.
What are the Main Benefits of Iodine?
Iodine benefits include thyroid health, cognitive development, and preventing iodine deficiency disorders.
Is it OK to Take Iodine Daily?
Taking iodine daily is safe in controlled amounts; consult healthcare providers for personal recommendations.
What Group is Iodine in Chemistry?
Iodine is in Group 17, the halogens, of the periodic table.
Iodine is an essential nutrient with significant health impacts. Regular consumption through iodine-rich foods ensures thyroid health and cognitive development. While daily intake is generally safe, it’s vital to adhere to recommended amounts. Understanding its role in chemistry as a halogen further highlights its importance. Always consult healthcare advice for personalized guidance, ensuring a healthy balance of this crucial element.