5+ Endothermic Reaction Examples
Heat is a pivotal part of a chemical reaction, as high temperatures excite the molecular structure of an object allowing it to create a chemical reaction. Endothermic reactions are one of the most pivotal interactions with heat in the natural world.
1. Endothermic & Exothermic Reactions
2. Chemical Reactions Object Endothermic Reactions

augusta.edu
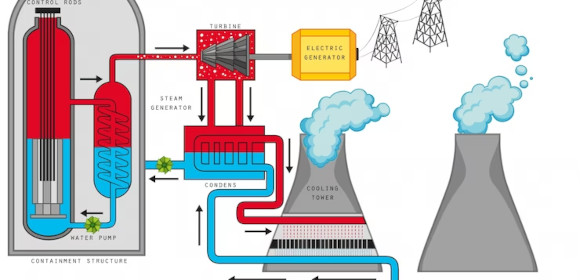
3. Applications of Endothermic Reaction

ntrs.nasa.gov
4. Endothermic & Exothermic Reactions Lab

eips.ca
5. Exothermic and Endothermic Reactions

data.testprepkart.com
What Is an Endothermic Reaction?
An endothermic reaction is the chemical property of an object or organism to absorb heat from its surroundings or environment. These reactions do not exert a lot of energy in the form of heat unlike their counterparts the exothermic reaction.
How to Identify an Endothermic Reaction
There are many ways to identify how the reaction will interact with heat due to the elements or factors that will surround said reaction. It is important to know whether the reaction is endothermic or exothermic as this will indicate how much heat is required in the solution.
Step 1A: Obtain a Thermometer
One of the easiest ways to identify and check if the reaction is endothermic is to monitor and calculate how the temperature in the environment and the object changes during the reaction. Begin by obtaining a thermometer to use as the main measuring tool in this reaction, you may use a normal thermometer or a digital thermometer for this effort.
Step 1B: Use the Formula of Enthalpy of Reaction
Another way to identify whether or not heat is absorbed or emitted is through the formula of enthalpy of reaction. This formula is ?H = energy used in reactant bond breaking + energy released in product bond making, where ?H is the change or alteration of heat or the enthalpy change.
Step 2A: Monitor the Temperature Change in the Solution or Reaction
Using the thermometer you must check whether the temperature increases or decreases in the environment or the solution. If the overall temperature decreases then the reaction is endothermic, but if the temperature increases then it is exothermic.
Step 2B: Solve for Enthalpy of Reaction
Using the formula, you will calculate the ?H. If the result is a positive integer then the reaction is endothermic because there is the act of breaking apart the bond does not require more energy.
FAQs
Exothermic vs. endothermic reaction; what is the difference between exothermic and endothermic reactions?
There are two ways the reaction will interact with heat, which are endothermic and exothermic. In an exothermic reaction outcome or the output will release heat as chemical energy or thermal energy. An endothermic reaction is the complete juxtaposition of the exothermic reaction, as the output will instead absorb heat from its surroundings.
What are common examples of endothermic reactions we can easily observe in the real world?
There are many real-life phenomena we can set for the observation that will help our understanding of the concept of an endothermic reaction. A real-life example of an endothermic reaction is the melting of ice and the evaporation of water. Another example of an endothermic reaction is nuclear fission, which also uses endothermic reactions to create nuclear energy and convert it to electrical energy.
Is the act of sweating an endothermic or exothermic reaction?
When a person does aerobic exercises or simple physical activities, the body converts complex carbohydrates and fats into energy. This creates heat and raises the internal body temperature of the person. To cool down, the body creates moisture and produces sweat through specific glands in the body. The sweat will try and absorb the heat from the body, which makes it an endothermic process.
A lot of processes and chemical reactions either produce or absorb heat as their primary consumption. The process of endothermic reactions is very pivotal in the survival of the human race because a lot of basic phenomena use endothermic reactions. For example, plants rely on endothermic processes to absorb the heat from the sunlight to create glucose and sugars, which allows these organisms to act as producers in the food web or food chain. Therefore, it is important to understand the role and phenomenon of endothermic reactions.